GE Logiq 200 PRO User manual

e i s e s
!
"
G
B M !
C E c c C

Operating Documentation
2 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |
Regulatory Requirement
This product complies with regulatory requirements of the following European Directive 93/42/EEC concerning medical devices
This manual is a reference for the LOGIQt200 PRO Series. It applies to all version of the 3.1X software for the LOGIQt200 PRO Series.
GE Medical Systems
GE Medical Systems: Telex 3797371
P.O. Box 414, Milwaukee, Wisconsin 53201 U.S.A.
(Asia, Pacific, Latin America, North America)
GE Ultrasound Europe: Tel: +49(0)212 2802 0
Kranzbühler GmbH & Co KG
Beethovenstraûe 239, Postfach 110560, D±42655
Solingen
GERMANY


|
|
Revision History |
|
|
|
% " "# !& |
|
|
|
|
|
REV |
DATE |
REASON FOR CHANGE |
|
|
|
|
|
|
|
|
|
0 |
August 1, 1999 |
Initial Release |
|
|
|
|
|
1 |
January 15, 2000 |
Software Version 3.02 Release |
|
|
|
|
|
2 |
June 15, 2000 |
Software Version 3.15 Release |
|
|
|
|
|
3 |
November 21, 2000 |
Software Version 3.16 Release |
|
|
|
|
|
L P
P |
|
P |
|
|
M |
M |
M |
M |
|
|
|
|
|
|
|
|
|
|
|
# |
|
|
|
|
% " "# !& A |
|
|
|
|
$# !& $! #" |
|
|
|
|
# #" # !$ |
|
|
|
|
# !$ |
|
|
|
|
# !$ |
|
|
|
|
# !$ 6 |
|
|
|
|
# !$ |
|
|
|
|
# !$ |
|
|
|
|
6 # !$ 6 |
|
|
|
|
7 # !$ 7 6 |
|
|
|
|
# !$ |
|
|
|
|
# !$ 66 |
|
|
|
|
# !$ |
|
|
|
|
# !$ |
|
|
|
|
6 |
|
|
|
|
# !$ |
|
|
|
|
# !$ 6 |
|
|
|
|
# !$ |
|
|
|
|
6 # !$ 6 |
|
|
|
|
LOGIQt200 PRO Series Basic Users Manual |
1 |
|
|
2234813±100 Rev 1 |
|

Revision History
Index 1 thru 14 |
1 |
2 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

Revision History
Please verify that you are using the latest revision of this document. Information pertaining to this document is maintained on GPC (GE Medical Systems Global Product Configuration). If you need to know the latest revision, contact your distributor, local GE Sales Representative or in the USA call the GE Ultrasound Clinical Answer Center at 1-800-682±5327 or 414-524-5698.
LOGIQt200 PRO Series Basic Users Manual |
3 |
|
|
2234813±100 Rev 1 |
|


Regulatory Requirements
Regulatory Requirements
.ForOnlyUSA
This product complies with the regulatory requirements of the following:
SCouncil Directive 93/42/EEC concerning medical devices: the 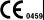 label affixed to the product testifies compliance to the Directive.
label affixed to the product testifies compliance to the Directive.
The location of the CE marking is shown on Safety chapter of this manual.
European registered place of business:
GE Medical Systems Europe
Quality Assurance Manager BP 34
F 78533 BUC CEDEX France Tel: +33 (0)1 30 70 40 40
SMedical Device Good Manufacturing Practice Manual issued by the FDA (Food and Drug Administration, Department of Health, USA).
SUnderwriters' Laboratories, Inc. (UL), an independent testing laboratory.
SCanadian Standards Association (CSA).
SInternational Electrotechnical Commission (IEC), international standards organizations, when applicable.
Caution: United States law restricts this device to sale or use by or on the order of a physician.
SGeneral Electric Medical Systems is ISO 9001 and EN 46001 certified.
SThe original document was written in English.
LOGIQt200 PRO Series Basic Users Manual |
1 |
|
|
2234813±100 Rev 1 |
|

Regulatory Requirements
NOTE: This equipment generates, uses and can radiate radio frequency energy. The equipment may cause radio frequency interference to other medical and non-medical devices and radio communications. To provide reasonable protection against such interference, this product complies with emissions limits for a Group 1, Class A Medical Devices Directive as stated in EN 60601±1±2. However, there is no guarantee that interference will not occur in a particular installation.
NOTE: If this equipment is found to cause interference (which may be determined by turning the equipment on and off), the user (or qualified service personnel) should attempt to correct the problem by one or more of the following measure(s):
±reorient or relocate the affected device(s)
±increase the separation between the equipment and the affected device
±power the equipment from a source different from that of the affected device
±consult the point of purchase or service representative for further suggestions
NOTE: The manufacturer is not responsible for any interference caused by using other than recommended interconnect cables or by unauthorized changes or modifications to this equipment. Unauthorized changes or modifications could void the users' authority to operate the equipment.
NOTE: To comply with the regulations on electromagnetic interference for a Class A FCC Device, all interconnect cables to peripheral devices must be shielded and properly grounded. Use of cables not properly shielded and grounded may result in the equipment causing radio frequency interference in violation of the FCC regulations.
NOTE: Do not use devices which intentionally transmit RF Signals (cellular phones, transceivers, or radio controlled products) in the vicinity of the equipment as it may cause performance outside the published specifications. Keep the power to these type devices turned off when near this equipment.
The medical staff in charge of this equipment is required to instruct technicians, patients, and other people who may be around this equipment to fully comply with the above requirement.
2 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

Table of Contents
Table of Contents
Front Matter
Title Page |
A |
Revision History . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
|
Regulatory Requirements . . . . . . . . . . . . . . . . . . . . . . |
Regulatory Req 1 |
Table of Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
Table of Contents 1 |
Chapter 1 ± Introduction
System Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±2 |
Attention . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±2 |
Prescription Device . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±2 |
System Components . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±2 |
Documentation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±3 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±4 |
General Indications for Use . . . . . . . . . . . . . . . . . . . . . . . . . |
1±5 |
Contraindications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±5 |
Who to Contact . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±6 |
Who To Contact . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±6 |
How This Book is Organized . . . . . . . . . . . . . . . . . . |
1±9 |
Manual Content . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±9 |
Manual Format . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
1±11 |
Chapter 2 ± Safety
Safety Precautions . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±2 |
Icon Description . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±2 |
Hazard Symbols . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±3 |
Patient Safety . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±4 |
Equipment and Personnel Safety . . . . . . . . . . . . . . . . . . . . |
2±6 |
Related Hazards . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±6 |
Device Labels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±8 |
Acoustic Output . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±16 |
Controls Affecting Output . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±16 |
Warning Label Locations . . . . . . . . . . . . . . . . . . . . . . . . . . . |
2±18 |
LOGIQt200 PRO Series Basic Users Manual |
1 |
|
|
2234813±100 Rev 1 |
|

Table of Contents
Chapter 3 ± Preparing the System for Use
Site Requirements . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±2 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±2 |
Before the system arrives . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±3 |
Environmental Requirements . . . . . . . . . . . . . . . . . . . . . . . |
3±4 |
Console Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±5 |
Console graphics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±5 |
Storage areas . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±7 |
Peripheral/Accessory Connector Panel . . . . . . . . . . . . . . |
3±8 |
Foot Switch (option) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±10 |
System Positioning/Transporting . . . . . . . . . . . . . |
3±11 |
Moving the System . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±11 |
Transporting the System . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±13 |
Wheels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±14 |
Powering On the System . . . . . . . . . . . . . . . . . . . . . |
3±15 |
Connecting and Using the System . . . . . . . . . . . . . . . . . . . |
3±15 |
Power Up Sequence . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±17 |
Adjusting the Display Monitor . . . . . . . . . . . . . . . . |
3±18 |
Rotate, tilt, raise and lower the monitor . . . . . . . . . . . . . . |
3±18 |
Brightness and Contrast . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±19 |
Probes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±21 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±21 |
Connecting the Probe . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±21 |
Activating the Probe . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±23 |
Storing the Probe . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±23 |
Deactivating the Probe . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±24 |
Operator Controls . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±25 |
Control Panel Map . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±25 |
Preset Keys . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±26 |
Measurement and Annotation . . . . . . . . . . . . . . . . . . . . . . |
3±27 |
Mode, Display and Record . . . . . . . . . . . . . . . . . . . . . . . . . |
3±29 |
Keyboard . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±31 |
Special Key Function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
3±32 |
2 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

Table of Contents
Chapter 4 ± Preparing for an Exam
Beginning an Exam . . . . . . . . . . . . . . . . . . . . . . . . . . |
4±2 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
4±2 |
Beginning a New Patient . . . . . . . . . . . . . . . . . . . . . . . . . . . |
4±2 |
OB/GYN Exam Category . . . . . . . . . . . . . . . . . . . . . . . . . . . |
4±6 |
ID/Name . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
4±6 |
Chapter 5 ± Modes
B±Mode . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
5±2 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
5±2 |
Optimizing the Image . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
5±6 |
M-Mode . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
5±17 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
5±17 |
M-Mode Display . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
5±17 |
Optimizing the Timeline . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
5±18 |
Mixed Mode Display Formats . . . . . . . . . . . . . . . . . |
5±23 |
Chapter 6 ± Scanning/Display Functions
Zooming an Image . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±2 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±2 |
Zooming an Image . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±2 |
Freezing an Image . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±6 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±6 |
Foot Switch option . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±6 |
Using Cine . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±7 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±7 |
Accessing Cine . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±8 |
Cine Gauge . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±8 |
Exiting Cine . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±8 |
Annotating an Image . . . . . . . . . . . . . . . . . . . . . . . . . |
6±9 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±9 |
Annotation Library . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±11 |
Adding Comments to an Image . . . . . . . . . . . . . . . . . . . . . |
6±12 |
Special Annotation Keys . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±13 |
Editing Annotations . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±14 |
Body Patterns . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
6±15 |
LOGIQt200 PRO Series Basic Users Manual |
3 |
|
|
2234813±100 Rev 1 |
|

Table of Contents
Chapter 7 ± General Measurements and Calculations
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±2 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±2 |
General Instructions . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±2 |
Erasing Measurements . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±3 |
Locating measurement controls . . . . . . . . . . . . . . . . . . . . . |
7±4 |
Measurement Key . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±5 |
Cursors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±5 |
B-Mode Measurements . . . . . . . . . . . . . . . . . . . . . . . |
7±6 |
Distance Measurement . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±6 |
Circumference/Area (Ellipse) Measurement . . . . . . . . . . |
7±8 |
Circumference/Area (Trace) Measurement . . . . . . . . . . . |
7±10 |
Echo Level Measurement . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±12 |
M-Mode Measurements . . . . . . . . . . . . . . . . . . . . . . . |
7±13 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±13 |
Tissue depth . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±14 |
Time . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±15 |
Slope . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
7±16 |
Chapter 8 ± Abdomen and Small Parts
General Calculations . . . . . . . . . . . . . . . . . . . . . . . . . |
8±2 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
8±2 |
Measuring Volume . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
8±5 |
Measuring Angle . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
8±8 |
Measuring Stenosis Area Ratio (% stenosis) . . . . . . . . . . |
8±9 |
Echo Level Histogram . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
8±15 |
Measuring heart rate (HR) . . . . . . . . . . . . . . . . . . . . . . . . . |
8±17 |
Helpful hints . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
8±19 |
General Calculation Formulas . . . . . . . . . . . . . . . . . . . . . . |
8±19 |
4 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

Table of Contents
Chapter 9 ± OB/GYN
Exam Preparation . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±3 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±3 |
OB Measurements and Formulas . . . . . . . . . . . . . |
9±4 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±4 |
OB Format Selection . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±4 |
OB Measurement Menus and Formulas . . . . . . . . . . . . . . |
9±4 |
OB Summary Reports . . . . . . . . . . . . . . . . . . . . . . . . |
9±17 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±17 |
Starting an Exam . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±19 |
Formulas . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±20 |
Editing the OB Summary Report . . . . . . . . . . . . . . . . . . . . |
9±21 |
Gestational Age Error Markers . . . . . . . . . . . . . . . . . . . . . . |
9±22 |
OB Report Layout . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±23 |
Measurement Averaging Page Layout . . . . . . . . . . . . . . . |
9±27 |
OB Anatomical Survey Page Layout . . . . . . . . . . . . . . . . . |
9±29 |
OB Trend Graph Page Layout . . . . . . . . . . . . . . . . . . . . . . |
9±30 |
OB Trend Graph Labeling . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±31 |
Changing OB Trend Graph Selection . . . . . . . . . . . . . . . . |
9±33 |
Input Previous Data Page Layout . . . . . . . . . . . . . . . . . . . |
9±34 |
Fetal Trend Management . . . . . . . . . . . . . . . . . . . . . |
9±36 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±36 |
Storing Patient Information . . . . . . . . . . . . . . . . . . . . . . . . . |
9±36 |
Growth Trending . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±41 |
Patient List Management . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±42 |
MGOB±Multigestational . . . . . . . . . . . . . . . . . . . . . . |
9±47 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±47 |
Patient Entry Menu . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±47 |
Distinguishing Each Fetus . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±48 |
Measurements/Calculations . . . . . . . . . . . . . . . . . . . . . . . . |
9±48 |
Change the Number of Fetuses . . . . . . . . . . . . . . . . . . . . . |
9±49 |
Report Page Layout . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±50 |
OB Graph . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±52 |
GYN Measurements . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±54 |
B-Mode . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±54 |
GYN Summary Report . . . . . . . . . . . . . . . . . . . . . . . . |
9±60 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±60 |
GYN Report Layout . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±62 |
GYN Calculation Formulas . . . . . . . . . . . . . . . . . . . . . . . . . |
9±63 |
Measurement Averaging Page Layout . . . . . . . . . . . . . . . |
9±64 |
IVF Report Page Layout . . . . . . . . . . . . . . . . . . . . . . . . . . . |
9±65 |
LOGIQt200 PRO Series Basic Users Manual |
5 |
|
|
2234813±100 Rev 1 |
|

Table of Contents
Chapter 10 ± Cardiology
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±2 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±2 |
Cardiology Calculations Measurement Menus . . . . . . . . |
10±5 |
General Guidelines . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±6 |
Auto Sequence Measurement . . . . . . . . . . . . . . . . . . . . . . |
10±6 |
BSA Calculation Methods . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±7 |
LV Analysis Measurements . . . . . . . . . . . . . . . . . . . |
10±8 |
Cubed, Teichholz, Gibson Methods . . . . . . . . . . . . . . . . . . |
10±8 |
Cubed Method Formulas . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±9 |
Teichholz Method Formulas . . . . . . . . . . . . . . . . . . . . . . . . |
10±10 |
Gibson Method Formulas . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±11 |
Bullet and Modified Simpson's Rule Methods . . . . . . . . . |
10±12 |
Bullet Method Formulas . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±13 |
Modified Simpson's Rule Method Formulas . . . . . . . . . . . |
10±14 |
Single Plane Ellipsoid Methods . . . . . . . . . . . . . . . . . . . . . |
10±15 |
Bi Plane Ellipsoid Methods . . . . . . . . . . . . . . . . . . . . . . . . . |
10±15 |
Single Plane Ellipsoid Method Formulas . . . . . . . . . . . . . |
10±16 |
Bi Plane Ellipsoid Method Formulas . . . . . . . . . . . . . . . . . |
10±17 |
M±Mode Analysis Measurement . . . . . . . . . . . . . . |
10±18 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±18 |
M-Mode Analysis ± Left/Right Ventricle (M-LV/RV) . . . . . |
10±19 |
M-Mode Analysis ± Left/Right Ventricle (M-LV/RV) Formulas . . . . . . . . . . . |
10±20 |
M-Mode Analysis ± Mitral Valve (M-MV) . . . . . . . . . . . . . . |
10±22 |
M-Mode Analysis ± Mitral Valve (M-MV) Formulas . . . . . |
10±23 |
M-Mode Analysis ± Aortic Valve (M-AV) . . . . . . . . . . . . . . |
10±24 |
M-Mode Analysis ± Aortic Valve (M-AV) Formulas . . . . . |
10±25 |
M-Mode Analysis ± Pulmonic Valve (M-PV) . . . . . . . . . . . |
10±26 |
M-Mode Analysis ± Pulmonic Valve (M-PV) Formulas . . |
10±27 |
M-Mode Analysis ± Tricuspid Valve (M-TV) . . . . . . . . . . . |
10±28 |
M-Mode Analysis ± Tricuspid Valve (M-TV) Formulas . . |
10±29 |
Additional Cardiology Calculations . . . . . . . . . . . |
10±30 |
ECG Option . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±31 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±31 |
ECG Lead Placement . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±32 |
ECG Default Preset . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±33 |
ECG Controls . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
10±33 |
6 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

Table of Contents
Chapter 11 ± Urology
Urology Calculations . . . . . . . . . . . . . . . . . . . . . . . . . |
11±2 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
11±2 |
Urology Summary Report Page . . . . . . . . . . . . . . . . . . . . . |
11±2 |
Using the Transaxial Probe . . . . . . . . . . . . . . . . . . . |
11±8 |
Transaxial Probe Preparation . . . . . . . . . . . . . . . . . . . . . . . |
11±8 |
Use of Transaxial Probe with Volume Stepper Device . . |
11±9 |
Stepper Volume Calculation . . . . . . . . . . . . . . . . . . |
11±13 |
Stepper Volume Formula . . . . . . . . . . . . . . . . . . . . . . . . . . . |
11±13 |
Description . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
11±14 |
Prerequisite . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
11±14 |
Area Measurement for Each Slice (Step Increment) . . . |
11±16 |
Chapter 12 ± Recording Images
Archiving Images . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
12±2 |
Image Memory . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
12±2 |
Recall . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
12±4 |
Helpful hints . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
12±4 |
Peripheral Devices . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
12±5 |
Black /White Video Page Printer Operations . . . . . . . . . . |
12±6 |
Multi-Image Camera (MIC) (IIE Model 460) . . . . . . . . . . . |
12±8 |
Video cassette recorder (VCR) . . . . . . . . . . . . . . . . . . . . . . |
12±9 |
MOD Image Archive (option) . . . . . . . . . . . . . . . . . . . . . . . |
12±10 |
Chapter 13 ± Customizing Your System
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
13±2 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
13±2 |
Setup Menu Command Lines . . . . . . . . . . . . . . . . . . . . . . . |
13±3 |
Basic Operation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
13±5 |
Menu Structure . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
13±8 |
General System 1 Setup (page 1 of 11) . . . . . . . . . . . . . . |
13±8 |
General System 2 Setup (page 2 of 11) . . . . . . . . . . . . . . |
13±12 |
Probe Parameter 1 Setup (page 3 of 11) . . . . . . . . . . . . . |
13±19 |
Probe Parameter 2 Setup (page 4 of 11) . . . . . . . . . . . . . |
13±22 |
Image Display & Application Setup (page 5 of 11) . . . . . |
13±24 |
Body Pattern Setup (page 6 of 11) . . . . . . . . . . . . . . . . . . . |
13±28 |
Comment Setup (page 7 of 11) . . . . . . . . . . . . . . . . . . . . . |
13±31 |
Measurement Setup (page 8 of 11) . . . . . . . . . . . . . . . . . . |
13±32 |
Patient Entry Setup (page 9 of 11) . . . . . . . . . . . . . . . . . . . |
13±45 |
User Define Category and Key Setup (page 10 of 11) . . |
13±47 |
User Utility (page 11 of 11) . . . . . . . . . . . . . . . . . . . . . . . . . |
13±48 |
LOGIQt200 PRO Series Basic Users Manual |
7 |
|
|
2234813±100 Rev 1 |
|

Table of Contents
Chapter 14 ± Probes and Biopsy
Probe Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±2 |
Ergonomics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±2 |
Applications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±5 |
Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±6 |
Connecting and Disconnecting a Probe . . . . . . . . . . . . . . |
14±7 |
Care and Maintenance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±8 |
Probe Safety . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±9 |
Probe handling and infection control . . . . . . . . . . . . . . . . . |
14±11 |
Coupling gels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±17 |
Planned Maintenance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±17 |
Probe Discussion . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±18 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±18 |
Curved Array (Convex) Probes . . . . . . . . . . . . . . . . . . . . . |
14±19 |
Linear Array Probes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±22 |
Sector Array Probes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±24 |
Biopsy Special Concerns . . . . . . . . . . . . . . . . . . . . . |
14±25 |
Precautions Concerning the Use of Biopsy Procedures |
14±25 |
Guidezones . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±27 |
Biopsy Procedure . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±27 |
Needle Guide Type Selection . . . . . . . . . . . . . . . . . . . . . . . |
14±28 |
Biopsy Guide Attachment . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±30 |
MTZ Probe Biopsy Guide . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±33 |
Biopsy Probes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
14±36 |
8 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

Table of Contents
Chapter 15 ± User Maintenance
System Data |
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±2 |
Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±2 |
|
LOGIQ 200 |
PRO Series Clinical Measurement Accuracy |
15±4 |
LOGIQ 200 |
PRO Series Clinical Calculation Accuracy . |
15±6 |
Warranties . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±7 |
Scope and Duration of Warranties . . . . . . . . . . . . . . . . . . . |
15±7 |
Warranty Exclusions . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±9 |
Exclusive Warranty Remedies . . . . . . . . . . . . . . . . . . . . . . |
15±10 |
System Care and Maintenance . . . . . . . . . . . . . . . . |
15±11 |
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±11 |
Inspecting the System . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±11 |
Weekly Maintenance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±12 |
Planned Maintenance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±16 |
Troubleshooting . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±17 |
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±17 |
Loose cables . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±17 |
Display Messages . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±18 |
System Error Message Description . . . . . . . . . . . . . . . . . . |
15±19 |
Operation Error Message Description . . . . . . . . . . . . . . . . |
15±20 |
Operation Guide Message Description . . . . . . . . . . . . . . . |
15±21 |
Assistance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±22 |
Clinical Questions . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±22 |
Service Questions . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±22 |
Literature . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±22 |
Accessories . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±22 |
Supplies/Accessories . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
15±23 |
LOGIQt200 PRO Series Basic Users Manual |
9 |
|
|
2234813±100 Rev 1 |
|

Table of Contents
Chapter 16 ± Acoustic Output
Bioeffects . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . |
16±2 |
Concerns Surrounding the Use of Diagnostic Ultrasound |
16±2 |
Operator Awareness and Actions to Minimize Bioeffect |
16±5 |
Implementing ALARA Methods . . . . . . . . . . . . . . . . . . . . . |
16±8 |
Training and User Assistance . . . . . . . . . . . . . . . . . . . . . . . |
16±9 |
IEC Acoustic Output Tables . . . . . . . . . . . . . . . . . . |
16±10 |
IEC Acoustic Output Tables . . . . . . . . . . . . . . . . . . . . . . . . |
16±10 |
Index
10 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

System Overview
System Overview
Attention
This manual contains enough information to operate the system safely. Advanced equipment training will be provided by a factory trained Applications Specialist for the agreed upon time period.
Read and understand all instructions in this manual before attempting to use the LOGIQ 200 PRO Series system.
Keep this User's Manual with the equipment at all times. Periodically review the procedures for operation and safety precautions.
Prescription Device
. For USA Caution: United States law restricts this device to sale or use by Only or on the order of a physician.
System Components
. |
Refer to the Service Manual (2235374) for the LOGIQ 200 PRO |
|
Series system components. |
2 |
LOGIQ 200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

System Overview
Documentation
LOGIQ 200 PRO Series Documentation consists of three manuals:
SThe Quick Start Guide (TRANSLATED) provides a step-by-step description of the basic features and operation of the LOGIQ 200 PRO Series. It is intended to be used in conjunction with the Basic User Manual in order to provide the information necessary to operate the system safely.
SThe Basic User Manual (TRANSLATED) provides information needed by the user to operate the system safely. It describes basic functions of the system, safety features, operating modes, basic measurements/calculations, probes, user care and maintenance.
SThe Advanced Reference Manual (ENGLISH ONLY) is intended for the trained, professional user. It contains all the information found in the Quick Start Guide and Basic User Manual, as well as information on options, advanced customization techniques and data tables.
The LOGIQ 200 PRO Series manuals are written for users who are familiar with basic ultrasound principals and techniques. They do not include sonography training or clinical procedures.
LOGIQ 200 PRO Series Basic Users Manual |
3 |
|
|
2234813±100 Rev 1 |
|

System Overview
Introduction
The LOGIQt200 PRO Series Ultrasound System is a high performance and compact size ultrasound imaging system, intended for general purpose applications.
The system provides image generation in B-Mode, M-Mode and B/M-Mode with all transducer types. Digital architecture allows maximum flexibility of all scanning modes and transducer types, throughout the full spectrum of operating frequencies.
All transducers are precise solid state array devices, allowing electronically controlled imaging with Convex, Micro-convex, Sector and Linear probes. Use of solid state designs allows a wide variety of scan parameters to be optimized including focusing, scan control, spatial resolution, temporal resolution and contrast resolution. The result is consistent generation of finely detailed anatomical resolution with excellent dynamic contrast tissue range and penetration.
The system display processor is highly versatile to produce the optimal set of imaging parameters and display formats without compromising important diagnostic information.
Versatile, yet easy to use, the LOGIQt200 PRO Series system combines a wide variety of state-of-the-art operator features without complicating operation. The operator can customize all set-up parameters for a given mode, probe or clinical application. Operator controls have been placed in a logical clinical format. Two simultaneous probe connections allow rapid switching electronically between probes without delaying the examination.
The LOGIQt200 PRO Series System provides a total imaging solution for today's diverse ultrasound department needs, with investment security through reliable upgrades, application enhancements, and complete product support from GE.
4 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

System Overview
General Indications for Use
The LOGIQ 200 PRO Series diagnostic ultrasound system is intended for use in diagnostic ultrasound imaging using B, M, B/M combination modes in the following areas:
SFetal
SAbdomen
SIntraoperative
SPediatric
SSmall organs including breast, neck, chest, male and female reproductive organs, limbs, and extremities
SAdult cephalic
SNeonatal cephalic
SAdult cardiac
SPediatric cardiac
STrans-vaginal
STrans-rectal
SUrology
Contraindications
The System is NOT intended for use in the following areas: Ophthalmic use (or any use causing the acoustic beam to pass through the eye).
LOGIQ 200 PRO Series Basic Users Manual |
5 |
|
|
2234813±100 Rev 1 |
|

Who to Contact
Who to Contact
Who To Contact
For additional information or assistance, please contact your local distributor or the appropriate support resource listed below:
USA
GE Medical Systems |
TEL: (1) 800±437±1171 |
Ultrasound Service Engineering |
FAX: (1) 414±647±4090 |
4855 W. Electric Avenue |
|
Milwaukee, WI 53219 |
|
Customer Answer Center |
TEL: (1) 800±682±5327 or |
|
(1) 414±524±5698 |
CANADA |
|
GE Medical Systems |
TEL: (1) 800±664±0732 |
Ultrasound Service Engineering |
|
4855 W. Electric Avenue |
|
Milwaukee, WI 53219 |
|
Customer Answer Center |
TEL: (1) 800±682±5327 or |
|
(1) 414±524±5698 |
LATIN & SOUTH AMERICA |
|
GE Medical Systems |
TEL: (1) 305±735±2304 |
Ultrasound Service Engineering |
|
4855 W. Electric Avenue |
|
Milwaukee, WI 53219 |
|
Customer Answer Center |
TEL: (1) 800±682±5327 or |
|
(1) 414±524±5255 |
EUROPE |
|
GE Ultraschall |
TEL: 0130 81 6370 toll free |
Deutschland GmbH & Co. KG |
TEL: (49)(0) 212.28.02.208 |
Beethovenstra e 239 |
FAX: (49)(0) 212.28.02.28 |
Postfach 11 05 60 |
|
D±42655 Solingen |
|
ASIA |
|
GE Medical Systems Asia |
TEL: (81) 426±56±0033 |
Asia Support Center |
FAX: (81) 426±56±0053 |
67±4 Takakura cho, Hachiouji±shi |
|
Tokyo, 192 JAPAN |
|
6 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

|
Who to Contact |
Who To Contact (cont'd) |
|
AUSTRIA |
|
GE GesmbH Medical Systems Austria |
TEL: 0660 8651 toll free |
Prinz Eugen Strasse 8/8 |
FAX: +43 1 505 38 74 |
A±1040 WIEN |
TLX: 136314 |
BELGIUM |
|
GE Medical Systems Benelux |
TEL: +32 0 3 320 12 11 |
Gulkenrodestraat 3 |
FAX: +32 0 3 320 12 59 |
B±2160 WOMMELGEM |
TLX: 72722 |
DENMARK |
|
GE Medical Systems Danmark |
TEL: +45 45 51 00 55 |
Skovlytoften 4 |
FAX: +45 42 42 59 89 |
DK±2840 HOLTE |
|
FRANCE |
|
GE Medical Systems |
TEL: +33 1 46 10 01 30 |
738 rue Yves Carmen |
FAX: +33 1 46 10 01 20 |
F±92658 BOULOGNE CEDEX |
|
GERMANY |
|
GE Ultraschall |
TEL: 0130 81 6370 toll free |
Deutschland GmbH & Co. KG |
TEL: (49)(0) 212.28.02.208 |
Beethovenstra e 239 |
FAX: (49)(0) 212.28.02.28 |
Postfach 11 05 60 |
|
D±42655 Solingen |
|
GREECE |
|
GE Medical Systems Hellas |
TEL: +30 1 93 24 582 |
41, Nikolaou Plastira Street |
FAX: +30 1 93 58 414 |
G±171 21 NEA SMYRNI |
|
ITALY |
|
GE Medical Systems Italia |
TEL: +39 39 20 881 |
Via Monte Albenza 9 |
FAX: +39 39 73 37 86 |
I±20052 MONZA |
TLX: 3333 28 |
NETHERLANDS |
|
GE Medical Systems Nederland B.V. |
TEL: +31 304 79711 |
Atoomweg 512 |
FAX: +31 304 11702 |
NL±3542 AB UTRECHT |
|
POLAND |
|
GE Medical Systems Polska |
TEL: +48 2 625 59 62 |
Krzywickiego 34 |
FAX: +48 2 615 59 66 |
P±02±078 WARSZAWA |
|
LOGIQt200 PRO Series Basic Users Manual |
7 |
|
|
2234813±100 Rev 1 |
|

Who to Contact
Who to Contact (cont'd)
PORTUGAL
GE Medical Systems Portuguesa S.A. |
TEL: |
+351 2 2007696/97 |
Rua Sa da Bandeira, 585 |
FAX: +351 2 2084494 |
|
Apartado 4094 |
TLX: |
22804 |
P±4002 PORTO CODEX |
|
|
RUSSIA
GE VNIIEM |
TEL: +7 095 956 7037 |
Mantulinskaya UI. 5A |
FAX: +7 502 220 32 59 |
123100 MOSCOW |
TLX: 613020 GEMED SU |
SPAIN
GE Medical Systems España |
TEL: +34 1 676 4012 |
Hierro 1 Arturo Gimeno |
+34 1 676 4047 |
Poligono Industrial I |
FAX: +34 1 675 3364 |
E±28850 TORREJON DE ARDOZ |
TLX: 22384 A/B GEMDE |
SWEDEN
GE Medical Systems |
TEL: +46 87 50 57 00 |
PO±BOX 1243 |
FAX: +46 87 51 30 90 |
S±16428 KISTA |
TLX: 12228 CGRSWES |
SWITZERLAND
GE Medical Systems (Schweiz) AG |
TEL: +41 41 425577 |
Sternmattweg 1 |
FAX: +41 41 421859 |
CH±6010 KRIENS |
|
TURKEY
GE Medical Systems Turkiye A.S. |
TEL: +90 212 75 5552 |
Mevluk Pehliran Sodak |
FAX: +90 212 211 2571 |
Yilmaz Han, No 24 Kat 1 |
|
Gayretteppe |
|
ISTANBUL |
|
UNITED KINGDOM
IGE Medical Systems |
TEL: +44 753 874000 |
Coolidge House |
FAX: +44 753 696067 |
352 Buckingham Avenue |
|
SLOUGH |
|
Berkshire SL1 4ER |
|
Manufacturer
SAMSUNG GE MEDICAL SYSTEMS
65±1, Sangdaewon±Dong, Chungwon±Ku, TEL: (82) 342±740±6114
Sungnam±Si,Kyunggi±Do FAX: (82) 342±746±9634
KOREA
8 |
LOGIQt200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

How This Book Is Organized
How This Book is Organized
The LOGIQ 200 PRO Series User Manual is organized to provide the information needed to start scanning right away. Detailed information is also provided for more time-intensive studies.
Manual Content
SGetting started. These sections give an overview of the system to help the operator start scanning as soon as possible.
S Introduction. Information concerning indications/ contraindications for use, who to contact and how this documentation is organized.
S Safety. Important information concerning the safe operation of the LOGIQ 200 PRO Series system.
S Preparing the System for Use. How to prepare the system for use and a map of the control layout.
S Preparing for an Exam. How to enter patient information, select an exam category.
SImage optimization. These sections detail how to improve image, trace, or spectral information.
S Modes. How to adjust and optimize B-Mode and M-Mode imaging.
S Scanning and Display Functions. Information concerning Zoom, Freeze, Cine and Annotation functions.
LOGIQ 200 PRO Series Basic Users Manual |
9 |
|
|
2234813±100 Rev 1 |
|

How This Book Is Organized
Manual Content (cont'd)
SMeasurements and Reports. Shows how to do general and exam category specific measurements and calculations.
SGeneral Measurements and Calculations. Emphasis on basic measurements for each mode.
SExam Categories.
SAbdomen and Small Parts.
SOB/GYN.
SCardiology.
SUrology.
SRecording Images. Explains the use of image archive and peripheral options.
SCustomizing your system. Shows how to customize the system for your particular institution, clinic, or exam type.
SProbes and Biopsy. Provides intended uses, specifications, care and maintenance, and biopsy capability instructions for each probe.
SUser Maintenance. Provides information concerning system specifications, warranties, error messages, user diagnostics, bioeffects, quality assurance, system care and assistance.
SData Tables. Provides necessary data for reference.
SAcoustic Output.
10 |
LOGIQ 200 PRO Series Basic Users Manual |
|
|
|
2234813±100 Rev 1 |

How This Book Is Organized
Manual Format
Information has been arranged and provided to help find information easily and quickly.
Finding information
Tables of Contents |
Locate topics in the main table of contents. |
||||
Tabs |
Chapter tabs are provided. |
||||
Headers/Footers |
The section name and page number appear on the outer |
||||
|
|
|
|
|
corners of every page. |
References |
See also page references that are noted. |
||||
Index |
Meant for frequent and easy reference. Extensive tool that |
||||
|
|
|
|
|
presents ideas, topics, terms, titles, headings, and cross |
|
|
|
|
|
references. Also, use it to find all entries of a like topic |
|
|
|
|
|
throughout the manual. |
2-Column Layout |
The right column contains text; the left column contains headers |
||||
|
|
|
|
|
and graphics to highlight the text. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Graphics |
Graphics provide a visual guide to the text when possible. |
Turn rotary knobs to the left (counterclockwise) and right (clockwise).
LOGIQt200 PRO Series Basic Users Manual |
11 |
|
|
2234813±100 Rev 1 |
|
 Loading...
Loading...