Samsung UGEO H60 Service manual

UGEO H60
Service Manual
Version 1.00.00
Copyright SAMSUNG MEDISON Co., LTD.

Safety Classifications
Classifications:
Type of protection against electrical shock: Class I
- Degree of protection against electrical shock (Patient connection):Type BF equipment Degree of protection against harmful ingress of water: Ordinary equipment
Degree of safety of application in the presence of a flammable anesthetic material with air or with oxygen or nitrous oxide: Equipment not suitable for use in the presence of a flammable anesthetic mixture with air or with oxygen or nitrous oxide.
Mode of operation: Continuous operation
Electromechanical safety standards met:
-IEC/EN 60601-1 Medical Electrical Equipment, Part 1General Requirements for Safety.
-IEC/EN 60601-1-1 Safety requirements for medical electrical systems.
-IEC/EN 60601-1-2 Electromagnetic compatibility -Requirements and tests.
-IEC/EN 60601-2-37 Particular requirements for the safety of ultrasonic medical diagnostic and monitoring equipment.
-IEC 61157 Declaration of acoustic output parameters.
-ISO 10993-1 Biological evaluation of medical devices.
-UL 60601-1 Medical Electrical Equipment, Part 1 General Requirements for Safety.
-CSA 22.2, 601.1 Medical Electrical Equipment, Part 1 General Requirements for Safety.
Declarations:
This is the CSA symbol for Canada and United States of
America
This is the manufacturer’s declaration of product
0123 compliance with applicable EEC directive(s) and the European notified body.
This is the manufacturer’s declaration of product compliance with applicable EEC directive(s).
This is the GMP symbol that shows that the product
complies with the Korean Good Manufacturing Practice
quality regulation system.

Attention
Read this service manual to familiarize yourself thoroughly with repair procedures and important safety information before attempting to service the product.
Failure to follow this information may cause an accident such as electric shock, as well as mechanical or other hazards to the service engineer, product operator, and/or patient.
1)Refer to the service manual when you service the product.
2)You are strongly urged to familiarize yourself with the operational safety information contained in ‘Chapter 2 Safety’.
3)This product is an ultrasound diagnosis device and cannot be used from the user's PC.
We are not responsible for errors that occur when the system is run on the user’s PC.
4)This product may only be serviced by the Global Service Team of Samsung Medison or an authorized engineer.
5)Samsung Medison is not responsible for any problems caused by an unauthorized person servicing the product.
6)The manufacturer is not responsible for any damage to this product caused by user carelessness and/or neglect.
7)The content of this manual may be changed without prior notice.
8)The following terms are used to highlight safety precautions that the user must be aware of:
DANGER
WARNING
CAUTION
NOTE
Disregarding this instruction may result in death, serious injury, or other dangerous situations.
Follow this information to prevent a serious accident or damage to property.
Hazards or unsafe practices that may result in minor personal injury or property damage.
The accompanying information covers an installation, operation, or maintenance procedure that requires careful attention from the user, but has little chance of leading directly to a dangerous situation.
If You Need Help
If you need help regarding the product, please contact the Samsung Medison Global Service Team in
charge of servicing this product.
|
|
|
UGEO H60 Service Manual |
Table of Contents |
|
||
Chapter 1 |
Introduction to Products .................................................................... |
1-1 |
|
1.1 |
Overview............................................................................................................... |
1-1 |
|
1.2 |
Main Features of UGEO H60............................................................................. |
1-2 |
|
1.3 |
Components......................................................................................................... |
1-3 |
|
|
1.3.1 |
Console .................................................................................................... |
1-3 |
|
1.3.2 |
Probe ........................................................................................................ |
1-4 |
|
1.3.3 |
Product Specification .............................................................................. |
1-5 |
|
1.3.4 |
Product Components .............................................................................. |
1-8 |
Chapter 2 |
Safety |
..................................................................................................... |
2-1 |
2.1 |
Overview............................................................................................................... |
2-1 |
|
2.2 |
Safety ..............................................................................................Precautions |
2-2 |
|
|
2.2.1 ....................................................................................... |
Safety Symbols |
2-2 |
|
2.2.2 ..................................................................................... |
Location of Label |
2-5 |
2.3 |
Electrical ...................................................................................................Safety |
2-6 |
|
|
2.3.1 ..................................................................Prevention of Electric Shock |
2-6 |
|
|
2.3.2 .......................................................................................................... |
ESD |
2-7 |
|
2.3.3 ........................................................................................................... |
EMI |
2-7 |
|
2.3.4 .......................................................................................................... |
EMC |
2-8 |
2.4 |
Mechanical .............................................................................................Safety |
2-13 |
|
|
2.4.1 .............................................................. |
Precautions during Operation |
2-13 |
|
2.4.2 ............................................................ |
Precautions during Movement |
2-13 |
|
2.4.3 ................................................Precautions during Monitor Operation |
2-14 |
|
2.5 |
Biological ................................................................................................Safety |
2-15 |
|
|
2.5.1 .................................................................................. |
ALARA Principles |
2-15 |
2.6 |
Protecting ....................................................................................Environment |
2-29 |
|
Chapter 3 |
Installing ................................................................................Product |
3-1 |
|
3.1 |
Overview............................................................................................................... |
3-1 |
|
3.2 |
Delivery................................................................................................................. |
3-3 |
|
|
3.2.1 ................................................................. |
Precautions when Delivering |
3-3 |
|
3.2.2 ..................................................................... |
Temperature and Humidity |
3-3 |
3.3 |
Unpacking ............................................................................................................ |
3-4 |
|
|
3.3.1 ................................................................... |
Disassembling Product Box |
3-4 |
3.4 |
Installation ...........................................................................................Condition |
3-5 |
|
|
3.4.1 .............................................................................................. |
Precautions |
3-5 |
3.5 |
How to ........................................................................................................Install |
3-6 |
|
Table of Contents 1
|
|
|
|
|
|
3.5.1 |
....................................................................................Installation Safety |
3-6 |
|
|
3.5.2 |
Connecting the Power Cable................................................................. |
3-6 |
|
|
3.5.3 |
Connecting Probe ................................................................................... |
3-7 |
|
3.6 |
Turning on the Product ....................................................................................... |
3-8 |
||
3.7 |
Turning off the Product........................................................................................ |
3-9 |
||
3.8 |
Connecting Peripheral Devices ....................................................................... |
3-10 |
||
3.9 |
Settings............................................................................................................... |
3-11 |
||
|
3.9.1 |
System General Settings ..................................................................... |
3-11 |
|
|
3.9.2 |
Monitor Display Settings ...................................................................... |
3-16 |
|
|
3.9.3 |
Measurement related Settings ............................................................ |
3-18 |
|
|
3.9.4 |
Report..................................................................................................... |
3-28 |
|
|
3.9.5 |
Annotation.............................................................................................. |
3-30 |
|
|
3.9.6 |
Body Marker .......................................................................................... |
3-33 |
|
|
3.9.7 |
Application ............................................................................................. |
3-36 |
|
|
3.9.8 |
Peripheral Devices Settings................................................................. |
3-37 |
|
|
3.9.9 |
Connectivity ........................................................................................... |
3-41 |
|
|
3.9.10 |
Service ................................................................................................... |
3-51 |
|
|
3.9.11 Help ........................................................................................................ |
3-52 |
||
3.10 Printer Installation .............................................................................................. |
3-53 |
|||
|
3.10.1 BW Printer Installation .......................................................................... |
3-53 |
||
|
3.10.2 Color Printer Installation ....................................................................... |
3-60 |
||
Chapter 4 |
Product Inspection ............................................................................. |
4-1 |
||
4.1 |
Overview............................................................................................................... |
4-1 |
||
4.2 |
Performance Inspection...................................................................................... |
4-2 |
||
|
4.2.1 |
Basic Inspection ...................................................................................... |
4-2 |
|
|
4.2.2 |
Detailed Inspection ................................................................................. |
4-3 |
|
Chapter 5 |
Product Structure................................................................................ |
5-1 |
||
5.1 |
Overview............................................................................................................... |
5-1 |
||
5.2 |
System Block Diagram ....................................................................................... |
5-4 |
||
5.3 |
TI (Transducer Interface) Board......................................................................... |
5-5 |
||
|
5.3.1 |
TI board layout......................................................................................... |
5-5 |
|
|
5.3.2 |
Description............................................................................................... |
5-5 |
|
|
5.3.3 |
Functional Specifications........................................................................ |
5-6 |
|
5.4 |
TR (Transmit and Receive) Board..................................................................... |
5-7 |
||
|
5.4.1 |
TR board layout....................................................................................... |
5-7 |
|
|
5.4.2 |
Description ............................................................................................. |
5-7 |
|
5.5 |
PI (PC Interface) Board .................................................................................... |
5-10 |
||
|
5.5.1 |
PI board Layout..................................................................................... |
5-10 |
|
|
5.5.2 |
PI Board Block Diagram....................................................................... |
5-11 |
|
Table of Contents 2
|
|
|
UGEO H60 Service Manual |
|
5.5.3 |
Description............................................................................................. |
5-11 |
|
5.5.4 |
Processor Module................................................................................. |
5-12 |
|
5.5.5 |
CPLD (SIC CPLD XC2C256-7VQG100C) ........................................ |
5-12 |
|
5.5.6 |
PIC (PC interface controller, PIC FPGA)............................................ |
5-12 |
|
5.5.7 |
FEC (PC interface controller, FEC FPGA)......................................... |
5-12 |
|
5.5.8 |
HDD........................................................................................................ |
5-12 |
|
5.5.9 |
Peripheral............................................................................................... |
5-12 |
|
5.5.10 |
PEX8311................................................................................................ |
5-13 |
|
5.5.11 |
HDMI Port Block.................................................................................... |
5-13 |
|
5.5.12 Sound Block .......................................................................................... |
5-14 |
|
5.6 |
Main Monitor ...................................................................................................... |
5-15 |
|
|
5.6.1 |
Main Monitor Specification ................................................................... |
5-15 |
5.7 |
Touch Screen..................................................................................................... |
5-16 |
|
|
5.7.1 |
Touch Screen Specification.................................................................. |
5-16 |
5.8 |
I /O Board ........................................................................................................... |
5-17 |
|
|
5.8.1 |
I/O Board cable Connection Diagram................................................. |
5-17 |
|
5.8.2 |
Description............................................................................................. |
5-17 |
|
5.8.3 |
System Cable Grappling...................................................................... |
5-18 |
5.9 |
DC POWER....................................................................................................... |
5-19 |
|
|
5.9.1 |
DC Power Layout.................................................................................. |
5-19 |
|
5.9.2 |
Description............................................................................................. |
5-19 |
5.10 AC POWER ....................................................................................................... |
5-20 |
||
|
5.10.1 AC Power Layout.................................................................................. |
5-20 |
|
|
5.10.2 Description............................................................................................. |
5-20 |
|
5.11 Software DSC .................................................................................................... |
5-21 |
||
|
5.11.1 Software DSC Block Diagram ............................................................. |
5-21 |
|
|
5.11.2 |
Description............................................................................................. |
5-21 |
|
5.11.3 |
Specification........................................................................................... |
5-21 |
5.12 Control Panel ..................................................................................................... |
5-23 |
||
|
5.12.1 Control Panel Block Diagram .............................................................. |
5-23 |
|
|
5.12.2 Description............................................................................................. |
5-23 |
|
|
5.12.3 Specification........................................................................................... |
5-23 |
|
Chapter 6 |
Basic Maintenance.............................................................................. |
6-1 |
|
6.1 |
Overview............................................................................................................... |
6-1 |
|
6.2 |
Service Mode....................................................................................................... |
6-2 |
|
6.3 |
System Information ............................................................................................. |
6-3 |
|
6.4 |
Adding and Deleting Options ............................................................................. |
6-4 |
|
|
6.4.1 |
Types of Option ....................................................................................... |
6-4 |
|
6.4.2 |
Registration of Option............................................................................. |
6-4 |
|
6.4.3 |
Deletion of Option ................................................................................... |
6-5 |
Table of Contents 3
|
|
|
|
6.5 |
..............................................................................................Back Up & Restore |
6-6 |
|
|
6.5.1 |
Back Up .................................................................................................... |
6-6 |
|
6.5.2 |
Restore ..................................................................................................... |
6-7 |
|
6.5.3 |
Log Backup .............................................................................................. |
6-7 |
|
6.5.4 |
Install Recovery System ......................................................................... |
6-7 |
|
6.5.5 |
Up - Grade Soft Ware ............................................................................... |
6-7 |
6.6 |
Diagnosis.............................................................................................................. |
6-8 |
|
|
6.6.1 |
Control Panel Test ................................................................................... |
6-8 |
|
6.6.2 Power On Self Test ................................................................................. |
6-8 |
|
|
6.6.3 |
Keyboard Test ......................................................................................... |
6-9 |
|
6.6.4 Built In Self Test....................................................................................... |
6-9 |
|
6.7 |
DICOM................................................................................................................ |
6-10 |
|
|
6.7.1 |
Network Settings ................................................................................... |
6-10 |
|
6.7.2 |
Network Status Notification .................................................................. |
6-11 |
|
6.7.3 |
Network /DICOM Test ........................................................................... |
6-12 |
Chapter 7 |
Diagnosis ............................................................................................. |
7-1 |
|
7.1 |
Overview............................................................................................................... |
7-1 |
|
7.2 |
Power |
|
7-2 |
|
7.2.1 .........................................................................Power does not Turn on |
7-2 |
|
|
7.2.2 .........................................................................Power does not Turn off |
7-2 |
|
|
7.2.3 ............................................................... |
Power Turns off Automatically |
7-2 |
7.3 |
Monitor.................................................................................................................. |
7-3 |
|
|
7.3.1 ...........................................................Nothing is shown on the Screen |
7-3 |
|
|
7.3.2 ............................................................................ |
Screen color Changes |
7-3 |
7.4 |
Error Message ..................................................................................................... |
7-4 |
|
|
7.4.1 .................................................Product Stops after error while booting |
7-4 |
|
|
7.4.2 ....................................................Product Operates while having Error |
7-4 |
|
7.5 |
Image.................................................................................................................... |
|
7-5 |
|
7.5.1 ................................................No BW Mode Image Echo and Format |
7-5 |
|
|
7.5.2 ..............................Lining Phenomenon on BW Mode Image (Noise) |
7-5 |
|
|
7.5.3 ....................................................................PW, Color, M Mode Failure |
7-5 |
|
7.6 |
Error Code............................................................................................................ |
7-6 |
|
Chapter 8 |
Assemble ...............................................................and Disassemble |
8-1 |
|
8.1 |
Overview............................................................................................................... |
8-1 |
|
8.2 |
Disassembling ................................................................................Front Panel |
8-3 |
|
|
8.2.1 .............................................................................................. |
Preparation |
8-3 |
|
8.2.2 ................................................................... |
Disassembling Front Cover |
8-3 |
|
8.2.3 ......................................... |
Disassembling Transducer Interface Board |
8-3 |
8.3 |
Disassembling ...................................................................................Rear Side |
8-5 |
|
Table of Contents 4
|
|
|
UGEO H60 Service Manual |
|
8.3.1 |
Preparation .............................................................................................. |
8-5 |
|
8.3.2 |
Disassembling Rear Side....................................................................... |
8-5 |
8.4 |
Disassembling Upper Panel............................................................................... |
8-6 |
|
|
8.4.1 |
Preparation .............................................................................................. |
8-6 |
|
8.4.2 |
Disassembling Upper Panel .................................................................. |
8-6 |
8.5 |
Disassembling Side Panel.................................................................................. |
8-7 |
|
|
8.5.1 |
Preparation .............................................................................................. |
8-7 |
|
8.5.2 |
Disassembling Side Panel..................................................................... |
8-7 |
8.6 |
Disassembling Control Panel............................................................................. |
8-8 |
|
|
8.6.1 |
Preparation .............................................................................................. |
8-8 |
|
8.6.2 |
Disassembling Control Panel ................................................................ |
8-8 |
8.7 |
Disassembling LCD Monitor............................................................................ |
8-9 |
|
|
8.7.1 |
Preparation .............................................................................................. |
8-9 |
|
8.7.2 |
Disassembling LCD Monitor.................................................................. |
8-9 |
8.8 |
Disassembling Monitor ARM............................................................................ |
8-10 |
|
|
8.8.1 |
Preparation ............................................................................................ |
8-10 |
|
8.8.2 |
Disassembling LCD Monitor................................................................ |
8-10 |
Chapter 9 |
Probe .................................................................................................... |
9-1 |
9.1 |
Overview............................................................................................................... |
9-1 |
9.2 |
Probe List ............................................................................................................. |
9-2 |
9.3 |
TI Table ................................................................................................................. |
9-5 |
9.4 |
Using Ultrasound Gel.......................................................................................... |
9-6 |
9.5 |
Probe Safety Precautions................................................................................... |
9-7 |
9.6 |
Using Sheaths ..................................................................................................... |
9-9 |
9.7 |
Cleansing and Disinfection of Probe ............................................................... |
9-10 |
Chapter 10 Maintenance and Management....................................................... |
10-1 |
10.1 Overview............................................................................................................. |
10-1 |
10.2 Operating Environment..................................................................................... |
10-2 |
10.2.1 Product Installation and Storage ......................................................... |
10-2 |
10.3 Product Management ....................................................................................... |
10-3 |
10.3.1 Cleaning................................................................................................. |
10-3 |
10.3.2 Disinfecting ............................................................................................ |
10-3 |
10.3.3 Replacing Fuse ..................................................................................... |
10-4 |
10.3.4 Managing Air Filter ................................................................................ |
10-5 |
10.3.5 Accuracy Inspection.............................................................................. |
10-6 |
10.4 Information Management ................................................................................. |
10-7 |
10.4.1 User Setting Back-up............................................................................ |
10-7 |
10.4.2 Patient Information Back-up ................................................................ |
10-7 |
10.4.3 Software................................................................................................. |
10-7 |
Table of Contents 5

|
|
|
Chapter 11 Service Component List |
...................................................................11-1 |
|
11.1 Overview............................................................................................................. |
11-1 |
|
11.2 Body Cover ........................................................................................................ |
11-2 |
|
11.3 Ultrasound System Parts.................................................................................. |
11-4 |
|
11.4 Control Panel Parts ........................................................................................... |
11-6 |
|
11.5 Probe ................................................................................................................ |
11-10 |
|
Table of Contents 6

1 Introduction to Products
1.1Overview
1.2Main Features of UGEO H60
1.3Components

UGEO H60 Service Manual
1Introduction to Products
1.1Overview
Chapter 1 describes the basic important information you need to know before repairing UGEO H60. It describes the main features and configuration and the product specification.
UGEO H60, a color ultrasound diagnostor with high resolution and deep penetration, is convenient and offers variety of measurement functions
Table of Contents |
|
||
1.1 |
Overview ..................................................................................................................... |
1-1 |
|
1.2 |
Main Features of UGEO H60................................................................................... |
1-2 |
|
1.3 |
Components............................................................................................................... |
1-3 |
|
|
1.3.1 |
Console............................................................................................................ |
1-3 |
|
1.3.2 |
Probe................................................................................................................ |
1-4 |
|
1.3.3 |
Product Specification ...................................................................................... |
1-5 |
|
1.3.4 |
Product Components...................................................................................... |
1-8 |
Chapter 1 Introduction to Products 1-1

1.2Main Features of UGEO H60
Digital Beam forming technology, self-developed at Samsung Medison Co., Ltd. Is applied.
Various Application: Can be applied to various fields such as General, Obstetrics, Gynecology, Abdomen, Vascular, Extremity, Cardiac, Urology, Breast
Various Diagnostic Modes: Various types of modes are available such as 2D mode, M mode, Color Doppler Mode (C mode), Power Doppler Mode (PD mode), PW Spectral Doppler Mode (D mode)
Stereoscopic Feature: Implements solid and detailed visuals with 3D and 4D imaging mode.
Measuring and Reporting Feature: Offers various measurement functions for each specific area besides measurement functions for distance, area, volume and perimeter. In addition, there is a report function using this measurement result.
Reviewing Scan Video Feature: Provides 2621 frames of Cine video and 4086 lines of Loop video at maximum.
SonoViewTM Feature: Available on storing, querying and compatible data with integrated video management system.
Digital Imaging and Communication in Medicine (DICOM) Feature: Can store, transfer, and print video using network.
Ease of Connection to Peripheral Devices: Can connect to various peripheral devices and use it.
Chapter 1 Introduction to Products 1-2

UGEO H60 Service Manual
1.3Components
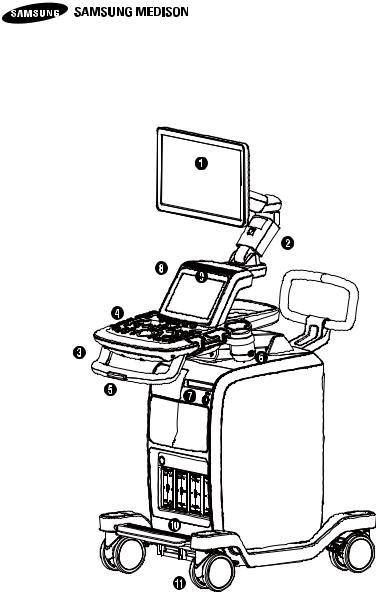
UGEO H60 is composed of console, probe and cart (option).
1.3.1 Console
The internal part of console is composed of devices mainly implementing ultrasound image, while the external part is composed of various connected devices and handle.
[Figure 1-1] H60 Front Figure
[Figure 1-2] H60 Side Figure
Chapter 1 Introduction to Products 1-3

[Figure 1-3] H60 Rear Figure
1.3.2 Probe
Probe is a device collecting data for ultrasound image by using ultrasound.
NOTE Refer to "Chapter 9 Probe" in service manual for more detail.
Chapter 1 Introduction to Products 1-4
UGEO H60 Service Manual
1.3.3 Product Specification
Physical Dimensions
Imaging modes
Gray Scale
Focusing
Probes
(Type BF / IPX7)
Probe connections
Monitor
Height: 1660 mm (with Monitor)
Width: 550 mm
Depth: 980 mm(with Keyboard)
Weight: 105kg (without accessories)
2D-Mode M-Mode Color Doppler
Pulsed Wave (PW) Spectral Doppler Power Doppler (PD)
3D/ 4D imaging mode Dual modes
Quad modes
Combined modes Simultaneous mode
Zoom Mode S-Flow
256 (8 bits)
Transmit focusing, maximum of eight points (four points simultaneously selectable)
Digital dynamic receive focusing (continuous)
Linear Array
L5-13
Curved Array
CS1-4, C2-8, CF4-9, ER4-9,EVN4-9
3D
3D2-6, 3D4-9, VE4-8
3 Probe Connectors
4 Probe Connectors for option
Main Monitor
Display area : 18.5 inch
Number of Pixel : 1366 x 768
Touch Screen Monitor
Display area : 10.1 inch
Number of Pixel : 1280 x 800
Chapter 1 Introduction to Products 1-5
Rear Panel
Input / Output
Connections
Image Storage
Application
Electrical Parameters
Measurement
Packages
Signal processing (Pre-processing)
Signal processing (Post-processing)
Audio Output Port( Right/Left ) VGA monitor
LAN USB Port
HDMI output
HDMI Input (HDMI Input is currently not supported.)
Maximum 45000 frames for CINE memory
Maximum 14000 Lines for LOOP memory
Image filing system
Obstetrics, Gynecology, Urology, Abdomen, Vascular,
Small Part, MSK Pediatric
100~240VAC, 620VA, 50/60Hz
Abdomen, Obstetrics, Fetal Echo, Gynecology, MSK, Pediatric Hips, Small Part, Urology, Vascular
* Refer the Chapter 5 for additional information
Acoustic Power Control
Analog TGC Control
Dynamic Aperture Control
Dynamic Apodization Control
Dynamic LPF Control
Digital TGC Control
Slider TGC Control Mode-Independent Gain Control Black Hole/Noise Spike Filtering 1D Lateral/Axial Filtering
2D Edge/Blurring Filtering Frame average
M/D Mode Sweep Speed Control Zoom
Image View Area Control
Image Orientation (left/right and up/down)
Chapter 1 Introduction to Products 1-6
UGEO H60 Service Manual
Measurement
Auxiliary
User Interface
Pressure Limits
Humidity Limits
Temperature Limits
Trackball operation of multiple cursors
2D mode: Linear measurements and area measurements using elliptical approximation or trace
M mode: Continuous readout of distance, time, and slope rate
Doppler mode: Velocity and trace
DVD Multi-Drive
Digital B/W Video Printer Digital Color Video Printer USB Printer
DVD Recorder Foot switch (IPX8)
e-Motion Marker (IPX 7) USB Flash Memory Media USB HDD
Monitor
English, German, French, Spanish, Italian, Russian,
Chinese
Operating: 700hPa to 1060hPa
Storage: 700hPa to 1060hPa
Operating: 30% to 75%
Storage & Shipping: 20% to 90%
Operating: 10 °C ~ 35°C
Storage & Shipping: -25°C ~ 60°C
Chapter 1 Introduction to Products 1-7
1.3.4 Product Components
This product is composed of monitor, control panel, console, peripheral devices and probe.
Monitor
Monitor Arm
Keyboard
Control Panel
Lift
Probe Holder
DVD Drive
USB Port
Speaker
Probe Port
Wheel
[Figure 1-4] The front of the product
1.3.4.1 Monitor
Color LCD flat monitors displays ultrasound image and extra information.
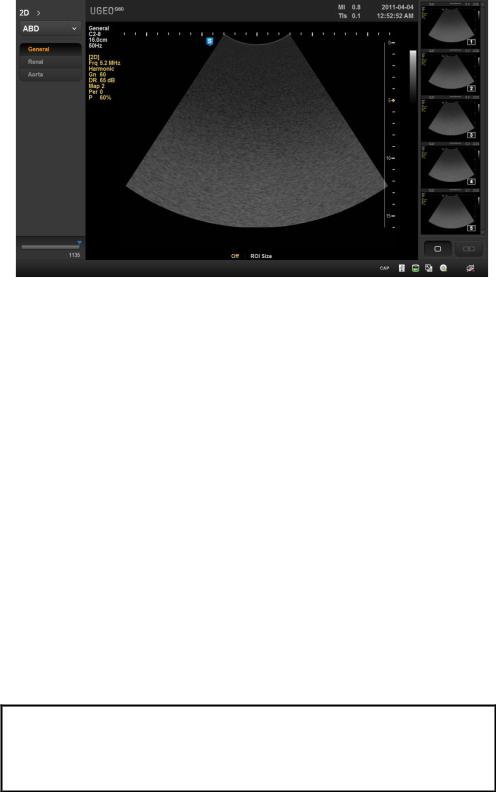
1) Screen Configuration
The screen of this product has contents such as ultrasound image and extra information, and necessary menu for users to operate the system. The screen configuration is composed of Title area, Measurement menu area, Imaging area, Thumbnail area, User Information as the figure below.
Chapter 1 Introduction to Products 1-8
UGEO H60 Service Manual
[Figure1-5] Screen Configuration
Title area
Displays patient information (name, ID, GA), name of hospital and operator, acoustic output information, date and time, etc.
Measurement menu area
Displays measurement menu while measuring each subject diagnosis.
Imaging area
Displays ultrasound image. Displays video information, annotation and various measuring information.
Thumbnail area
Displays stored video by clicking [Store] button. Clicking by pointer will display enlarge image on imaging area. Display up to 17 images.
Displays Body Marker in BodyMarker Mode.
User Information Status Information area
Displays various information needed for users while operating this system. Displays condition of the storage media (HDD, USB, CD), Caps Lock condition, DICOM, etc.
Tips! Displays of current condition of system.
 : Shows On condition of Caps Lock
: Shows On condition of Caps Lock
 : Shows that there is CD/DVD storage media
: Shows that there is CD/DVD storage media
Chapter 1 Introduction to Products 1-9
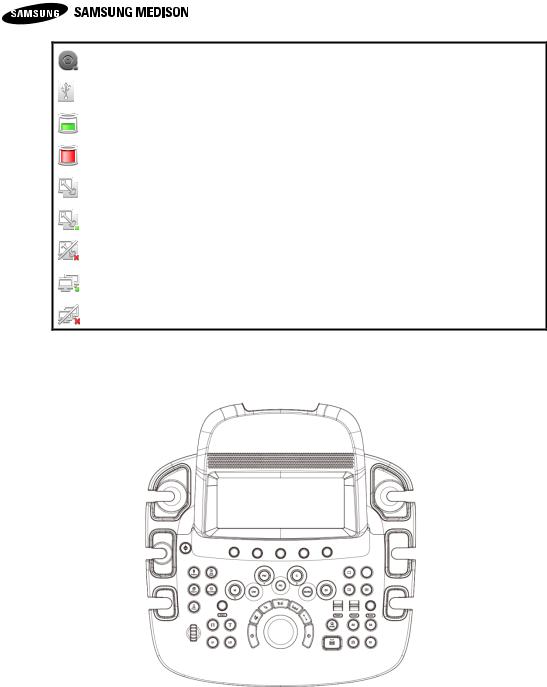
: Shows that there is no CD/DVD storage media. : Shows that there is no USB storage media.
: Shows that there is more than 10% of HDD storage. : Shows that there is less than 10% of HDD storage. : Shows that DICOM Spooler is empty.
: Shows that DICOM Spooler is in operation.
: Shows that DICOM Spooler has failed in its operation. : Shows that LAN is connected.
: Shows that LAN is not connected.
1.3.4.2 Control Panel
Users operate the system with control panel.
[Figure 1-6] Control Panel
Control panel is composed of keyboard, soft menu, button, dial, dial-button, slide and track.
Dial-button is a operating tool, combination of dial and button.
Chapter 1 Introduction to Products 1-10

UGEO H60 Service Manual
1) Detailed Features of Control Panel
Following are the description and the use of each control on control panel. Controls with various different features are described in more detail after Chapter 3 of this manual.
|
Button |
Turns On/Off the product |
|
On/Off |
|
|
|
|
|
|
|
Patient |
Button |
Displays Patient Information screen where you can select |
|
|
|||
|
|
the patient ID on the list or input new patient information |
|
|
|
|
|
Probe |
Button |
Displays Probe Selection screen where you can select and |
|
change the probe and diagnostic subject |
|||
|
|
||
|
|
|
|
SonoView |
Button |
Displays a screen where you can review or manage stored images |
|
|
|||
|
|
|
|
End Exam |
Button |
Reset the related data after finishing the examination on |
|
the diagnosed patients |
|||
|
|
||
|
|
|
|
Report |
Button |
Displays the report screen which shows measurement |
|
result of corresponding diagnostic subject |
|||
|
|
||
|
|
|
|
|
Button |
Inputs BodyMarker on the video |
|
|
|
|
|
|
Button |
Inputs texts on the video |
|
|
|
|
|
|
|
This button can be set to user's preference. Features of |
|
U1 |
Button |
each button can be set from Setup > Peripherals > |
|
|
|
Customize Key |
|
|
|
|
|
|
|
This button can be set to user's preference. Features of |
|
U2 |
Button |
each button can be set from Setup > Peripherals > |
|
|
|
Customize Key |
|
|
Button |
Displays exclusive videos on the screen |
|
|
|
||
|
|
|
|
|
Button |
Compares two separate videos |
|
|
|
||
|
|
|
Chapter 1 Introduction to Products 1-11

|
|
|
|
|
|
|
|
|
|
|
Button |
Compare four separate videos |
||
|
|
|||
|
|
|
|
|
Depth |
Dial-Button |
Adjusts the depth of the image which can be observed |
||
|
||||
|
|
|
|
|
Focus |
Dial-Button |
Moves the position of the focus to the desired target area |
||
|
||||
|
|
|
|
|
|
|
|
Zoom Box appears. |
|
Zoom |
Dial-Button |
Press [Exit] button to exit Zoom mode. |
||
|
|
|
||
|
|
|
|
|
|
|
|
Adjusts the angle of sample volume in spectral doppler |
|
|
|
|
mode. Also used for adjusting the angle of arrow and |
|
Angle |
Dial-Button |
adjusting probe angle of Body marker. |
||
|
|
|
Moves Reference Slice to the left and right using [Angle] |
|
|
|
|
Dial-Button in 3D View |
|
Q Scan |
Button |
Uses Quick Scan feature |
||
|
|
|
|
|
|
|
|
Stores, prints and sends to DICOM Server with having |
|
Store, |
Button |
settings of each feature for each. |
||
|
Features of each Button can be set from Setup > |
|||
S1, S2, |
|
|
||
|
|
Peripherals > Customize Keys |
||
S3 |
|
|
||
|
|
|
||
|
|
|
|
|
|
Button |
Stops the video which is being scanned or reactivates the |
||
Freeze |
stopped video |
|||
|
|
|||
|
|
|
|
|
|
|
|
Starts or exits the M mode. Adjusts gain by turning Dial- |
|
M |
Dial-Button |
Button. Rotates the image in the direction of the x-axis in |
||
|
|
|
3D View |
|
|
|
|
||
PD |
Button |
Starts or exits power doppler mode |
||
|
|
|
|
|
|
|
|
Starts or exits color doppler mode. Adjusts gain by turning |
|
Color |
Dial-Button |
Dial-Button. |
||
|
|
|
Rotates the image in the direction of the x-axis in 3D View |
|
|
|
|
||
2D |
Dial-Button |
Starts 2D mode. Adjusts gain by turning Dial-Button. |
||
|
|
|
|
|
Chapter 1 Introduction to Products 1-12
|
|
|
|
UGEO H60 Service Manual |
|
|
|
|
|
|
|
|
|
Starts or exits PW spectral doppler mode. Adjusts gain by |
PW |
|
Dial-Button |
turning Dial-Button. |
|
|
|
|
|
Rotates the image in the direction of the y-axis in 3D View |
|
|
|
|
|
CW |
|
|
Button |
This button is currently not supported |
|
|
|
|
|
3D / 4D |
|
|
Button |
Used for On / Off on 3D/4D mode. |
|
|
|
|
|
|
|
|
|
[Uses by setting the [Set] or [Exit] feature. Features of each |
|
|
|
|
Button can be set from Setup > Peripherals > Customize |
|
|
|
|
Keys |
Set / Exit |
|
|
Button |
- Set: Selects desireditem or value using trackball, or |
|
|
|
changes the feature of trackball. |
|
|
|
|
|
|
|
|
|
|
- Exit: Exists the current feature and returns to the previous |
|
|
|
|
state |
Pointer |
|
|
Button |
The arrow pointer appears on the screen in the scan mode |
|
|
|
|
|
|
|
|
|
|
|
|
|
Button |
Deletes text, arrow, body marker, measurement result, etc |
|
|
|
|
|
Clear |
|
|
|
displayed on the screen |
|
|
|
|
|
|
|
|
Button |
Changes to other feature which is supported in the current |
|
|
|
trackball feature |
|
Change |
|
|
|
|
|
|
|
|
|
Calculator |
|
|
Button |
Starts measuring on each diagnostic subject |
|
|
|
|
|
|
|
|
|
|
|
|
|
Button |
Starts basic measurement such as distance, volume, |
|
|
|
|
|
Caliper |
|
|
|
circumference and area |
|
|
|
|
|
Trackball |
|
Trackball |
Moves the cursor on the screen. There are video search |
|
|
feature in Cine footage. |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Bands might appear on the image if adjusting gain value of |
|
CAUTION |
|
adjacent TGC slide with large difference |
||
|
|
|
||
Chapter 1 Introduction to Products 1-13

2) Keybard
Used to input texts
|
[Figure 1-7] Keyboard |
|
|
Help |
Help Manual appears on the screen |
|
|
Patient |
General Information appears on the screen |
|
|
Patient info. |
Shows or hides patient information on the screen |
|
|
Image Info. |
Shows or hides Image Parameter on the screen |
|
|
DICOM Spooler |
DICOM Spooler appears on the screen |
|
|
Arrow |
Starts Arrow mode |
|
|
Home |
Moves cursor to the Home position in Annotation mode |
|
|
Set Home |
Sets the Home position in Annotation mode |
|
|
Delete Word |
Deletes last inputted text in Annotation mode |
|
|
Chapter 1 Introduction to Products 1-14
|
UGEO H60 Service Manual |
|
|
Delete All |
Deletes all inputted text in Annotation mode |
|
|
Setup |
Setup screen appears |
|
|
Insert |
Select the input method |
|
Decrease the brightness of the monitor by pressing the Fn key |
|
|
Delete |
Deletes Text |
|
Increase the brightness of the monitor by pressing the Fn key |
|
|
3) Touch-Screen
It is an operating tool which users directly touch. The available features are shown as Button or Dial Button in the current mode.
Screen configuration of touch-screen
Information area: shows the title of touch screen currently used.
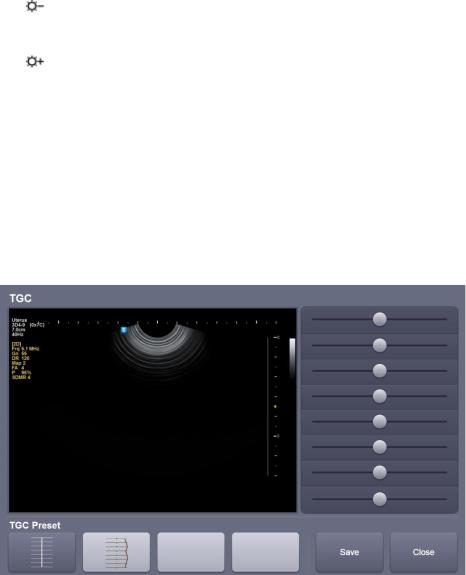
TGC: adjusts TGC slide when TGC curve settings on the touch screen is active.
[Figure 1-8] TGC
Menu area: The available menu in the current input mode is shown as Button. Use by pressing the Button itself, and menus which are being used are shown as yellow.
Chapter 1 Introduction to Products 1-15
Soft menu area: shows soft menu available in current input mode. Use by pressing Dial-Button right below the menu or turning it to left or right.
Tip! When there are two menu for touch screen
In case there are 2 menus on the top and bottom, press corresponding DialButton and select the desired menu. Use Dial-Button after pressing the Button on the menu that you wish to use on the touch screen.
[Figure 1-9] Display of touch-screen
4)Adjustment of the Control Panel
Do not move the control panel with excessive force.
Use the rear handle to move the product
CAUTION
Horizontal Adjustment
Move the product carefully side to side by holding the handle.
Vertical Adjustment
Move the product carefully up and down by pressing the lever on the handle of control panel.
Chapter 1 Introduction to Products 1-16

UGEO H60 Service Manual
1.3.4.3 Console
Console is divided into two main parts of the internal and external. The internal of the console is consisted of the devices for implementing ultrasound image. The external of the console is composed of various connected devices, probe holder, storage space, handle, wheel, etc.
1) Rear Panel
Located on the back of the product, it connects various peripheral devices such as monitor, printer, etc..
[Figure 1-10] Rear panel
2) Power connector
HDMI: delivers digital type image and audio signals in one cable simultaneously
USB port: connects peripheral devices for
USB
Audio Output (input): is used for output of audio signal
S-VHS port (I/O): connects VCR in S-VHS method
D-SUB port: outputs analog signals to monitor
Network port: connects network
Mic port (input): connects microphone
Chapter 1 Introduction to Products 1-17
 Loading...
Loading...