Samsung HS40 Service manual

HS40
Service Manual

Version 1.00
HS40
Service Manual
English
SM-HS40-ENG-01
(Empty page)

Safety Requirements
Classification
–Type of protection against electric shocks: Class I
–Degree of protection against electric shocks (when the patient is in physical contact): Type BF or type CF mounting
–Degree of protection against the ingress of harmful liquids: General equipment
–Degree of safety of use in the presence of flammable anesthetic agent mixed with air,
oxygen, or nitrous oxide: Not suitable for use near flammable anesthetic agent mixed with air, oxygen, or nitrous oxide
–Mode of operation: Continuous operation
Safety standards the device conforms to
–Medical Electrical Equipment - Part 1: General Requirements for Basic Safety and Essential Performance [IEC 60601-1:2005/A1:2012]
–Medical Electrical Equipment - Part 1-2: General Requirements for Basic Safety and Essential Performance - Collateral Standard: Electromagnetic Compatibility – Requirements and Tests [IEC 60601-1-2:2007]
–Medical Electrical Equipment - Part 1-6: General Requirements for Basic Safety and Essential Performance - Collateral Standard: Usability [IEC 60601-1-6:2010]
–Medical Electrical Equipment - Part 2-37: Particular Requirements for the Basic Safety
and Essential Performance of Ultrasonic Medical Diagnostic and Monitoring Equipment
[IEC 60601-2-37:2007]
–Medical Electrical Equipment - Part 1: General Requirements for Safety [IEC 60601- 1:1988, A1:1991, A2:1995]
–Medical Electrical Equipment - Part 1-1: General Requirements for Safety – Collateral Standards: General Requirements for Medical Electrical Systems [IEC 60601-1-1:2000]
–Medical Electrical Equipment - Part 1-2: General Requirements for Safety – Collateral Standards: Electromagnetic Compatibility – Requirements and Tests [IEC 60601-1-
2:2001, A1:2004]
–Medical Electrical Equipment - Part 1-4: General Requirements for Safety – Collateral
Standards: Programmable Electrical Medical Systems [IEC 60601-1-4:1996, A1:1999]
–Medical Electrical Equipment - Part 2-37: Particular Requirements for the Basic Safety
and Essential Performance of Ultrasonic Medical Diagnostic and Monitoring Equipment
[IEC 60601-2-37:2001, A1:2004, A2:2005]
– Medical Devices – Application of Risk Management to Medical Devices [ISO 14971:2007]
–Medical Electrical Equipment - Part 1: General Requirements for Safety [UL 60601-
1:2003]
–Medical Electrical Equipment – Part 1: General Requirements for Basic Safety and Essential Performance[CAN/CSA C22.2 No. 60601-1:14]

–Medical Electrical Equipment – Part 1: General Requirements for Basic Safety and
Essential Performance [ANSI /AAMI ES60601-1:2005/(R)2012, AND C1:2009 AND A2:2010(R)2012]
–Biological Evaluation of Medical Devices – Part 1: evaluation and testing [ISO 10993-1:
2009]
–Standard Means for Reporting the Acoustic Output of Medical Diagnostic Ultrasonic Equipment [IEC 61157:2007]
Statements
Nemko-CCL mark with the indicators “C” and “US” means that the product is certified for both the U.S. and Canadian markets, to the applicable U.S. and Canadian standards.
This mark certifies that the product conforms to applicable EEC standards and has been certified by the European certification
agency.
This mark certifies that the product conforms to applicable EEC standards.
The GMP symbol indicates the Good Manufacturing Practice in accordance with the Korean quality management regulations.

Precautions for Use
Be sure to read this Service Manual thoroughly to familiarize yourself with the operation of the product and the relevant safety information before attempting to use the product.
•Keep this service manual near the product and refer to it when using the product.
•Please familiarize yourself with the safety precautions in “Chapter 2. Safety” and “Chapter 10. Maintenance” in particular.
•This service manual does not include diagnosis results or opinions. Also, check the reference information for the measured area of the body before using the application’s measurement results in any diagnosis.
•This product is an ultrasound diagnosis device and cannot be used from the user’s PC. The
manufacturer is not responsible for any problems that may be caused by such attempts.
•This product must only be used by persons who have sufficient knowledge of and/or a
qualification in clinical pathology. Unqualified persons are prohibited from using the product.
•The manufacturer is not responsible for any damage to this product caused by user carelessness and/or neglect.
•Product orders are based on individually agreed specifications and may not include all
functions specified in this service manual.
• Some functions or options, probes, and the like may not be used in certain countries.
•All reference material on standards, regulations, and related revisions are valid at the time of the publication of this service manual.
•Screen images in this service manual are examples and may differ from the actual screen or system.
• The content of this service manual is subject to change without prior notice.
•Products that are not manufactured by Samsung Medison are indicated with the trademarks of their respective owners.
•The following terms are used to highlight precautions that the user must pay particular attention to:

Disregarding this instruction mayresult in death, serious injury, or other dangerous situations.
DANGER
Follow these instructions to prevent a serious accident or damage to property.
WARNING
Follow these instructions to prevent minor accidents and damage to property.
CAUTION
The accompanying information covers installation, operation, and maintenance procedures that require careful attention from the user, but have little chance of leading
directly to a dangerous situation.
NOTE

Revision History
The revision history of this service manual is as follows:
VERSION |
DATE |
REASON FOR CHANGE |
|
|
|
|
|
|
1.00.00 |
2017.04.01 |
Initial Release |
|
|
|
If You Need Assistance
If you need a service manual or any assistance with the product, please contact the Samsung Medison customer service department or your local vendor.

Patient Privacy Policy
Treatment of Patients’ Personal Information
One of Samsung Medison’s social responsibilities is to recognize the importance of patient privacy and to handle and protect the personal information provided by our patients in an appropriate manner.
Patient information stored in repaired and demo products should be handled as follows:
•Patient Information: Refers to information, such as IDs, names, DOBs, and images, which can be used to identify specific patients.
•Patient Information Management: Personal information provided by patients must be managed carefully and solely for the purposes of repairs and demonstrations of Samsung Medison products, and they must not be used for unauthorized purposes, be lost, be revised, or be shared with a third party.
Management of Patient Information on Demo and Trade Products
•Patient information stored on a product during the course of its use must be handled as the patient desires. (delete or backup)
•When completing a demonstration and/or collecting a trade product, patient information must be deleted if so requested by the client, to prevent its unintended distribution to outside parties.
Management of Patient Information on Repaired Hospital Products
•Before removing the product from hospital premises for repairs, all patient information should be backed up to a storage device such as external HDD or memory, and then handed over to the hospital.
•Before removing the product from hospital premises, each patient should sign the patient privacy agreement.

Request for Repairs and Patient Privacy Agreement
Date: YYYY/MM/DD
Customer Information
Hospital
Handler
Product Information
|
Product name |
|
|
|
|
|
|
|
|
|
|
|
Software version |
|
|
|
|
|
|
|
|
|
|
|
S/N |
|
|
|
|
|
|
|
|
|
|
|
Symptom |
|
|
|
|
|
|
|
|
|
|
|
Stored item |
□ None |
□ Main unit |
□ HDD |
□ DVD/CD |
|
□ USB |
□ Photo |
□ Other ( |
) |
|
|
|
||||
|
|
|
|
|
|
|
Outsourced product |
□ None |
|
|
|
|
□ Provide |
details if applicable ( |
) |
||
|
|
||||
|
|
|
|
|
|
Administration |
|
|
|
|
|
|
|
|
|
|
|
|
Handling date |
YY |
MM DD |
|
|
|
|
|
|
|
|
|
Customer service |
|
|
|
|
|
representative |
|
|
|
|
|
|
|
|
|
|
Please sign inside the box below if you agree to the following:
□Data saved on memory devices of products being repaired (e.g., hard disks) may become lost during tests and repairs.
□Data saved on a product being repaired must be backed up by the customer.
□Samsung will not be held responsible for any loss of data not backed up.
□Samsung Medison does not use patient information for unauthorized purposes, modify such information, or provide it to third parties.
Customer Signature
I entrust my product to ( |
) for repair and agree to the policies above. |
||
Date: |
YY/MM/DD |
Name: |
(Signature) |
Table of Contents |
1 |
Table of Contents
Chapter 1 |
Introduction |
1 |
|
1.1. |
Product Specifications............................................................................................. |
2 |
|
1.2. |
Product Configuration ............................................................................................. |
5 |
|
|
1.2.1. |
Monitor ........................................................................................................ |
7 |
|
1.2.2. |
Control Panel ............................................................................................... |
9 |
|
1.2.3. |
Console ..................................................................................................... |
16 |
|
1.2.4. |
Peripheral Devices ..................................................................................... |
18 |
|
1.2.5. |
Probes....................................................................................................... |
20 |
|
1.2.6. |
Accessories ............................................................................................... |
22 |
|
1.2.7. |
Optional Accessories .................................................................................. |
23 |
|
1.2.8. |
Optional Functions ..................................................................................... |
24 |
Chapter 2 Safety |
1 |
||
2.1. |
Purpose of Use ........................................................................................................ |
2 |
|
|
2.1.1. |
Contraindications ......................................................................................... |
2 |
2.2. |
Safety Information.................................................................................................... |
3 |
|
|
2.2.1. |
Safety Symbols ............................................................................................ |
3 |
|
2.2.2. |
Symbols ...................................................................................................... |
7 |
|
2.2.3. |
Labels ......................................................................................................... |
7 |
2.3. |
Electrical Safety ....................................................................................................... |
8 |
|
|
2.3.1. |
Prevention of Electric Shocks........................................................................ |
8 |
|
2.3.2. |
ECG-Related Information............................................................................ |
10 |
|
2.3.3. |
ESD .......................................................................................................... |
10 |
|
2.3.4. |
EMI ............................................................................................................ |
11 |
|
2.3.5. |
EMC........................................................................................................... |
11 |
2.4. |
Mechanical Safety................................................................................................... |
18 |
|
|
2.4.1. |
Moving the Equipment ................................................................................ |
18 |
|
2.4.2. |
Precautions for Use .................................................................................... |
19 |
2.5. |
Biological Safety..................................................................................................... |
22 |
|
|
2.5.1. |
ALARA Principle......................................................................................... |
22 |
2.6. |
Protecting the Environment .................................................................................... |
35 |
|
|
2.6.1. |
EU WEEE Directive .................................................................................... |
35 |
|
2.6.2. |
EU REACH Regulation ............................................................................... |
35 |
|
2.6.3. |
State of California Proposition 65 Warning (US Only).................................... |
35 |

2 HS40 Service Manual
Chapter 3. Installing Product |
1 |
||
3.1. |
Transporting ............................................................................................................. |
3 |
|
|
3.1.1. |
Precautions During Transport ....................................................................... |
3 |
|
3.1.2. |
Brakes ........................................................................................................ |
3 |
|
3.1.3. |
Precautions on Ramps ................................................................................. |
3 |
|
3.1.4. |
Humidity and Temperature ............................................................................ |
4 |
3.2. |
Unpacking the Product ............................................................................................. |
5 |
|
|
3.2.1. |
Dismantling the Product's Box ...................................................................... |
5 |
|
3.2.2. |
Accessories................................................................................................. |
6 |
|
3.2.3. Release of the Locking Mechanism ............................................................... |
6 |
|
3.3. |
Installation Environment........................................................................................... |
7 |
|
|
3.3.1. |
Caution ....................................................................................................... |
7 |
3.4. |
Installing the Product................................................................................................ |
8 |
|
|
3.4.1. |
Installation Safety......................................................................................... |
8 |
|
3.4.2. |
Connecting Peripherals ............................................................................... |
10 |
3.5. |
System Power......................................................................................................... |
13 |
|
|
3.5.1. Turning the Power On ................................................................................. |
13 |
|
|
3.5.2. Shutting Down the System........................................................................... |
13 |
|
3.6. |
System Settings...................................................................................................... |
14 |
|
|
3.6.1. |
General System Settings ............................................................................. |
15 |
|
3.6.2. |
General ...................................................................................................... |
15 |
|
3.6.3. |
Display ....................................................................................................... |
19 |
|
3.6.4. |
Patient ....................................................................................................... |
20 |
|
3.6.5. |
Quick Preset............................................................................................... |
23 |
|
3.6.6. |
EzExam+.................................................................................................... |
24 |
|
3.6.7. |
Imaging ...................................................................................................... |
27 |
|
3.6.8. |
Application/Preset ....................................................................................... |
30 |
|
3.6.9. |
Application.................................................................................................. |
32 |
|
3.6.10. |
Measurement Settings................................................................................. |
33 |
|
3.6.11. |
Report ........................................................................................................ |
49 |
|
3.6.12. |
Annotation .................................................................................................. |
52 |
|
3.6.13. |
Body Marker ............................................................................................... |
57 |
|
3.6.14. |
Customize .................................................................................................. |
60 |
|
3.6.15. |
Peripherals ................................................................................................. |
64 |
|
3.6.16. |
Connectivity................................................................................................ |
67 |
|
3.6.17. |
Service ....................................................................................................... |
86 |
|
3.6.18. |
Help ........................................................................................................... |
86 |
Table of Contents |
3 |
Chapter 4. Product Inspection |
1 |
|
4.1. Inspecting Functions ............................................................................................... |
2 |
|
4.1.1. |
Basic Inspections ......................................................................................... |
2 |
4.1.2. |
Detailed Inspections ..................................................................................... |
3 |
Chapter 5. Product Structure |
1 |
||
5.1. |
Overview .................................................................................................................. |
3 |
|
5.2. |
System Block Diagram............................................................................................. |
5 |
|
|
5.2.1. |
System Block Diagram ................................................................................. |
5 |
|
5.2.2. |
System Rack Design .................................................................................... |
6 |
5.3. |
Basic Structure of the Product................................................................................. |
7 |
|
|
5.3.1. |
Electronic Structure ...................................................................................... |
7 |
|
5.3.2. |
Ultrasound System Part ................................................................................ |
7 |
|
5.3.3. |
PC Part ....................................................................................................... |
8 |
|
5.3.4. |
User Interface Part ....................................................................................... |
8 |
|
5.3.5. |
Power Part ................................................................................................... |
8 |
5.4. |
Ultrasound System Part ........................................................................................... |
9 |
|
|
5.4.1. |
Main Functions of PSA (Probe Select Assembly) ............................................ |
9 |
|
5.4.2. |
Beam Former Board .................................................................................... |
11 |
|
5.4.3. |
Back End Board ......................................................................................... |
15 |
5.5. |
PC Part |
.................................................................................................................... |
20 |
|
5.5.1. ................................................................................................. |
PC Module |
20 |
|
5.5.2. ............................................................................................ |
Rear IO Board |
21 |
|
5.5.3. ............................................................................................ |
Software DSC |
23 |
5.6. |
User Interface ..................................................................................................Part |
24 |
|
|
5.6.1. ............................................................................................. |
Control Panel |
24 |
|
5.6.2. .............................................................................................. |
Main Monitor |
26 |
|
5.6.3. ............................................................................................. |
Touch - Screen |
27 |
|
5.6.4. ........................................................................................... |
Display Layout |
28 |
5.7. |
Power Part .............................................................................................................. |
29 |
|
|
5.7.1. .......................................................................................................... |
ADM |
29 |
|
5.7.2. ................................................................................. |
Power Block Diagram |
30 |
|
5.7.3. .............................................................................................. |
Specification |
30 |
5.8. |
Interconnect ..............................................................................................Diagram |
31 |
|
|
5.8.1. ........................................................................ |
HS40 Interconnect Diagram |
31 |
|
5.8.2. ....................................................................................... |
Cable Description |
32 |

4 HS40 Service Manual
Chapter 6. Service Mode |
1 |
||
6.1. |
Service Mode ............................................................................................................ |
3 |
|
|
6.1.1. How to Enter the Service Mode..................................................................... |
3 |
|
6.2. |
Service Information .................................................................................................. |
4 |
|
6.3. |
Configuration............................................................................................................ |
5 |
|
|
6.3.1. |
Clock ........................................................................................................... |
5 |
|
6.3.2. |
Keyboard/Region ......................................................................................... |
5 |
|
6.3.3. |
TCP/IP ........................................................................................................ |
6 |
|
6.3.4. |
Option ......................................................................................................... |
7 |
|
6.3.5. |
User Account and User Account Management ............................................... |
8 |
|
6.3.6. |
Printer ......................................................................................................... |
9 |
6.4. |
System |
.................................................................................................................... |
10 |
|
6.4.1. ....................................................................................... |
Upgrade Software |
10 |
|
6.4.2. ................................................................................. |
System Serial Number |
11 |
|
6.4.3. ......................................................................................... |
Window Explorer |
12 |
|
6.4.4. .............................................................................. |
Install Recovery System |
13 |
6.5. |
Diagnostics............................................................................................................. |
14 |
|
|
6.5.1. ....................................................................................... |
Control Panel Test |
14 |
|
6.5.2. ............................................................................................. |
Keyboard Test |
14 |
|
6.5.3. ..................................................................................... |
Power On Self Test |
14 |
|
6.5.4. .......................................................................................... |
Built in Self Test |
14 |
|
6.5.5. ................................................................................................ |
Monitor Test |
14 |
6.6. |
Backup ...............................................................................................and Restore |
15 |
|
|
6.6.1. ....................................................................................................... |
Backup |
15 |
|
6.6.2. ...................................................................................................... |
Restore |
15 |
|
6.6.3. ................................................................................................ |
Log Backup |
15 |
6.7. |
Report ..................................................................................................................... |
|
16 |
|
6.7.1. ........................................................................................................ |
Report |
16 |
6.8. |
Demo Play............................................................................................................... |
17 |
|
|
6.8.1. .................................................................................................. |
Demo Play |
17 |
6.9. |
Enter Installation ..............................................................................................Key |
18 |
|
Table of Contents |
5 |
Chapter 7. Troubleshooting |
1 |
||
7.1. |
Power Issues............................................................................................................ |
2 |
|
|
7.1.1. |
Power Does Not Turn On .............................................................................. |
2 |
|
7.1.2. |
Power Does Not Turn Off .............................................................................. |
2 |
|
7.1.3. |
Power Turns Off by Itself ............................................................................... |
2 |
7.2. |
Monitor |
..................................................................................................................... |
3 |
|
7.2.1. ............................................................... |
Nothing Is Displayed on the Screen |
3 |
|
7.2.2. ..................................................................................... |
Screen is Discolored |
3 |
7.3. |
Error Messages........................................................................................................ |
4 |
|
|
7.3.1. ......................................................................... |
Error Occurs During Booting |
4 |
7.4. |
Image ....................................................................................................................... |
|
4 |
|
7.4.1. ..............................2D Mode: There is No IMAGE ECHO or IMAGE FORMAT |
4 |
|
|
7.4.2. ........................................................ |
Lines (Noise) Appear in 2D Mode Image |
4 |
|
7.4.3. ........................................................................M, C, PW, CW Mode Trouble |
4 |
|
7.5. |
USB Drive ..........................................................................................Unrecognized |
5 |
|
|
7.5.1. ................................ |
Failed to recognize the USB (Removable device) device |
5 |
7.6. |
Trouble .............................................................................................Shooting Tree |
6 |
|
Chapter 8. Disassembly and Assembly |
1 |
||
8.1. |
Preparation .............................................................................................................. |
2 |
|
|
8.1.1. |
Preparation .................................................................................................. |
2 |
8.2. |
Product Disassembly ............................................................................................... |
3 |
|
|
8.2.1. |
Front Side Disassembly ................................................................................ |
3 |
|
8.2.2. |
Rear Side Disassembly ................................................................................ |
6 |
|
8.2.3. |
Control Panel Disassembly ........................................................................... |
7 |
|
8.2.4. |
Touch Panel Disassembly ............................................................................. |
8 |
|
8.2.5. |
Track Ball .................................................................................................... |
9 |
|
8.2.6. |
Speaker Disassembly ................................................................................. |
10 |
|
8.2.7. |
Monitor Disassembly ................................................................................... |
11 |
|
8.2.8. |
Monitor ARM Disassembly .......................................................................... |
12 |
8.3. |
Installation of Optional Parts .................................................................................. |
13 |
|
|
8.3.1. |
Overview of Optional Parts ......................................................................... |
13 |
|
8.3.2. |
Option Parts............................................................................................... |
14 |
|
8.3.3. |
Installation of External Physical Keyboard .................................................... |
16 |
|
8.3.4. |
Installation of Gel Warmer........................................................................... |
16 |
|
8.3.5. |
Installation of Endo Cavity Holder ................................................................ |
17 |
|
8.3.6. |
Installation of Holder Chart.......................................................................... |
18 |
|
8.3.7. |
Installation of Rear Tray .............................................................................. |
18 |

6 HS40 Service Manual
|
8.3.8. |
Installation of Printer Tray (1/2) .................................................................... |
19 |
|
8.3.9. |
Installation of Printer Tray (2/2) .................................................................... |
19 |
Chapter 9 |
Probes |
1 |
|
9.1. |
Probes |
...................................................................................................................... |
2 |
|
9.1.1. .................................................................................................... |
Probe List |
2 |
|
9.1.2. ........................................................................ |
Ultrasound Transmission Gel |
9 |
|
9.1.3. ...................................................................................................... |
Sheaths |
10 |
|
9.1.4. ............................................................................ |
Probe Safety Precautions |
11 |
|
9.1.5. .............................................................Cleaning and Disinfecting the Probe |
13 |
|
9.2. |
Biopsy..................................................................................................................... |
|
22 |
|
9.2.1. ............................................................................... |
Biopsy Kit Components |
22 |
|
9.2.2. .................................................................................... |
Using the Biopsy Kit |
23 |
|
9.2.3. ........................................................................... |
Assembling the Biopsy Kit |
26 |
|
9.2.4. ....................................................... |
Cleaning and Disinfecting the Biopsy Kit |
30 |
10. |
Maintenance |
1 |
|
10.1. |
Operational Environment.......................................................................................... |
2 |
|
10.2. |
Product Maintenance ................................................................................................ |
3 |
|
|
10.2.1. |
Cleaning and Disinfecting ............................................................................. |
3 |
|
10.2.2. |
Cleaning...................................................................................................... |
3 |
|
10.2.3. |
Cleaning Air Filters ....................................................................................... |
5 |
|
10.2.4. |
Accuracy Checks ......................................................................................... |
6 |
10.3. |
Data Maintenance ..................................................................................................... |
7 |
|
|
10.3.1. |
User Settings Backup ................................................................................... |
7 |
|
10.3.2. |
Backing Up Patient Information ..................................................................... |
7 |
|
10.3.3. |
Software...................................................................................................... |
7 |
Chapter 11. Service Part List |
1 |
|
11.1. |
Body Cover Parts...................................................................................................... |
2 |
11.2. |
Control Panel Parts................................................................................................... |
5 |

Chapter 1
Introduction
1.1. |
Product Specifications .............................................................................................. |
2 |
|
1.2. |
Product Configuration ............................................................................................... |
5 |
|
|
1.2.1. |
Monitor........................................................................................................... |
7 |
|
1.2.2. |
Control Panel ................................................................................................. |
9 |
|
1.2.3. |
Console ....................................................................................................... |
16 |
|
1.2.4. |
Peripheral Devices........................................................................................ |
18 |
|
1.2.5. |
Probes ......................................................................................................... |
20 |
|
1.2.6. |
Accessories.................................................................................................. |
22 |
|
1.2.7. |
Optional Accessories .................................................................................... |
23 |
|
1.2.8. |
Optional Functions........................................................................................ |
24 |
1 – 2 HS40 Service Manual
1.1. Product Specifications
Physical
Dimensions
Imaging Modes
Gray Scale
Focusing
Probes
(Type BF/IPX7)
Height: 1,620 mm (with Monitor)
Width: 520 mm
Depth: 730 mm (with Keyboard)
Weight: 54 kg (without accessories)
Weight: Approx. 57 kg (with Safe Working Load)
2D Mode
M Mode Color M Mode
Anatomical Mode Color Doppler Mode
Pulsed Wave (PW) Spectral Doppler Mode Continuous Wave (CW) Doppler Mode Tissue Doppler Imaging (TDI) Mode Tissue Doppler Wave (TDW) Mode
Power Doppler (PD) Mode ElastoScan Mode
3D/4D/XI STIC imaging Mode Dual Mode
Quad Mode Combined Mode Simultaneous Mode Zoom Mode
256 (8 bits)
Transmit focusing, maximum of eight points (four points simultaneously selectable)
Digital dynamic receive focusing (continuous)
Linear Array LA3-16AD
Curved Array CA2-8AD, CF4-9
Phased Array
PN2-4
Endo Cavity
EVN4-9
3D
VN4-8, V5-9
Chapter 1. Introduction 1 – 3
Probe
Connections
Monitor
ECG
Rear Panel
Input/Output
Connections
Image Storage
Application
Electrical
Parameters
Measurement
Packages
Signal
Processing (Pre-processing)
Pencil
DP2B
3 Probe Connectors, 4 Probe Connectors for option CW Probe Connector
Main Monitor
Number of Pixel: 1920 x 1080
21.5 inch LCD Monitor (LED Backlight unit, hereafter referred to as “LCD monitor”)
Touch Screen Monitor Number of Pixel: 1280 x 800
10.1 inch LCD Monitor (LED Backlight unit, hereafter referred to as “LCD monitor”)
USB Type (Type CF)
Audio Output Port (Right/Left)
VGA monitor
LAN
USB Port
CW
HDMI output
HDMI Input (HDMI Input is currently not supported.)
Maximum 45,000 frames for Cine memory
Maximum 14,000 Lines for Loop memory
Image filing system
Obstetrics, Gynecology, Urology, Abdomen, Cardiac, Vascular, Small Parts, TCD, MSK, Pediatric
100 - 240VAC, 620VA, 50/60Hz
Abdomen, Obstetrics, Gynecology, MSK, Pediatric, Small Parts, Urology, Vascular, Cardiac
* Refer to the ‘Chapter 8. Measurements and Calculations’ for additional information.
Acoustic Power Control
Analog TGC Control
Dynamic Aperture Control
Dynamic Apodization Control
1 – 4 HS40 Service Manual
Signal
Processing (Post-
processing)
Measurement
Auxiliary
User Interface
Pressure Limits
Humidity Limits
Temperature
Limits
Dynamic LPF Control
Digital TGC Control
Slider TGC Control Mode-Independent Gain Control Black Hole/Noise Spike Filtering 1D Lateral/Axial Filtering
2D Edge/Blurring Filtering Frame average
M/D Mode Sweep Speed Control Zoom
Image View Area Control
Image Orientation (left/right and up/down)
Trackball operation of multiple cursors
•2D mode: Linear measurements and area measurements using elliptical approximation or trace
• |
M mode: Continuous readout of distance, time, and slope rate |
• |
Doppler mode: Velocity and trace |
DVD Multi-Drive
Digital B/W Video Printer
Digital Color Video Printer
USB Printer
DVD Recorder
Foot switch (IPX8)
USB Flash Memory Media
USB HDD
USB ECG
Monitor
English, German, French, Spanish, Italian, Russian, Chinese, Portuguese
Operating: 700 – 1060hPa
Storage: 700 – 1060hPa
Operating: 30 – 75%
Storage & Shipping: 20 – 90%
Operating: 10 – 35°C
Storage & Shipping: -25 – 60°C

Chapter 1. Introduction 1 – 5
1.2. Product Configuration
This product consists of monitor, control panel, console, peripheral devices and probes.
Monitor
Monitor armTouch PanelSpeaker
Control Panel & HandleUSB port
Probe holderLift & Lift LeverECG port
Outlet for internal peripheral devices
Probe portCW PortWheel
Brake
Filter
[Figure 1.1 Front of the Product]
1 – 6 HS40 Service Manual
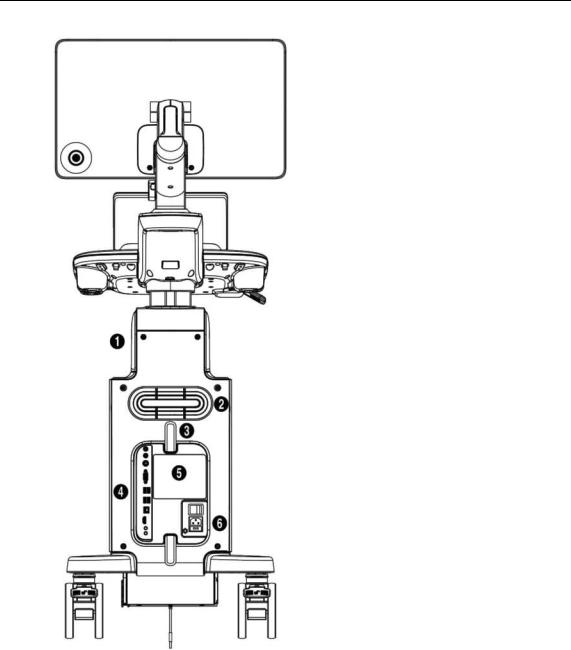
Storage compartmentsVentilation
Cable holderRear panelID label
Power connection
[Figure 1.2 Back of the Product]
Chapter 1. Introduction 1 – 7
1.2.1. Monitor
Ultrasound images and other information are displayed on the color LCD monitor.
1.2.1.1. Screen Layout
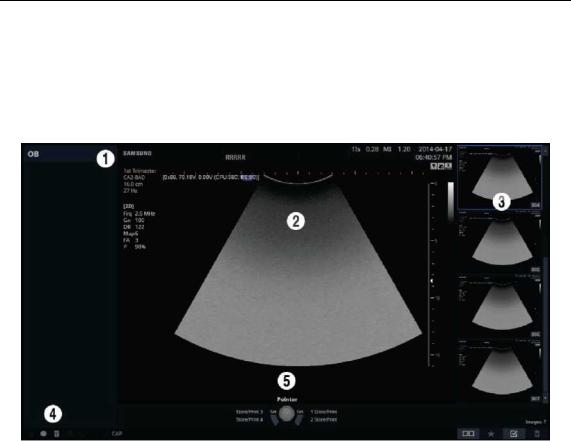
The monitor displays ultrasound images, operation menus and a variety of other information.
[Figure 1.3 Monitor Display]
Title Area
Displays patient information, hospital name, application, frame rate, depth, probe information, acoustic output information, and the current date and time.
Image Area
Displays ultrasound images. Image information, annotation, and measurement information are also displayed.
Thumbnail Area
Images saved by pressing the preset Store button are shown in the thumbnails. When you save Single screens, up to 4 images are shown in a list; for Quad screens, up to 12 images are shown. Click it with the pointer to enlarge the preview image.
User Information and Status Information Area
Information that is useful to the user, such as current system status, image information, selectable items, etc., is displayed.
User Key (User Defined Key) Area
Settings for User Defined Keys, including the positions of Set and Exit buttons, are displayed. You can change the setting of each button in Setup > Customize > Buttons.

1 – 8 HS40 Service Manual
For information on User Key Setup, please refer to ‘Chapter 3. Utilities’.
NOTE
TIP
Principles of Operation of the Diagnostic Ultrasound System
Medical ultrasound images are created by digital memory and computer when they
convert the high-frequency wave signals that are transmitted and received by the probe.
As ultrasound waves propagate through the human body, they generate reflected signals whenever they encounter a change in density. For example, reflected signals are generated when signals pass from fatty tissues to muscle tissues.
Reflected signals return to the probe, where they are converted into electronic signals. The reflected signals are amplified and processed by analog and digital circuits that have filters for various frequencies and response time options. Then
they are again converted into high-frequency electronic signals, and saved as a series of digital image signals. The monitor displays the image signals stored on the storage device in real time. The entire process of transmitting, receiving, and
processing signals is controlled by the computer.

Chapter 1. Introduction 1 – 9
1.2.2. Control Panel
The system can be controlled by using the control panel.
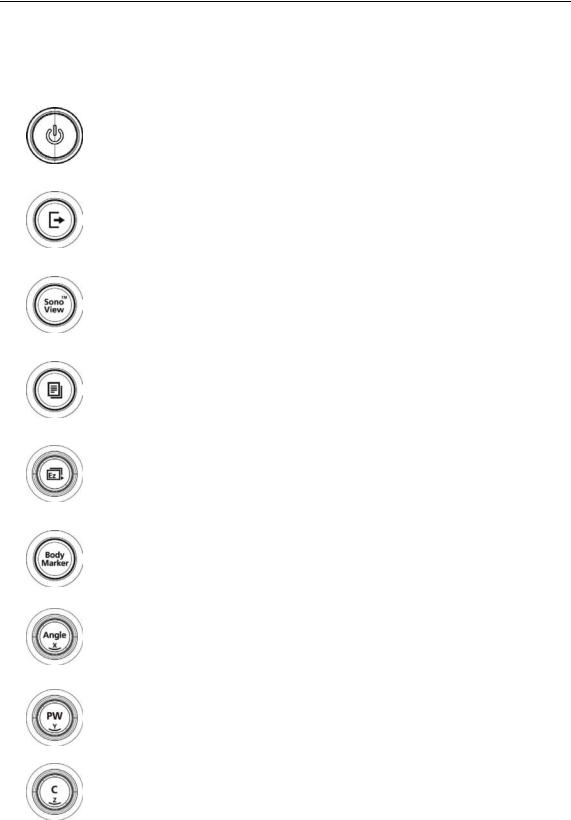
[Figure 1.4 Control Panel]
The control panel consists of a soft menus, buttons, dials, dial-buttons, a slider, and a trackball. The dial-button can be used both as a dial and a button.
1 – 10 HS40 Service Manual
1.2.2.1. Functions of the Control Panel
The following are the descriptions and instructions for the controls on the control panel. For
more information on controls with multiple functions, see ‘Chapter 3. Utilities’ and later in this manual.
|
Button |
Turns the system on/off. |
On/Off |
|
|
|
|
|
|
Button |
Finishes the exam of the currently selected patient and resets |
|
the related data. |
|
|
|
|
End Exam |
|
|
|
|
|
|
Button |
Displays the screen for viewing and managing stored images. |
SonoView |
|
|
|
|
|
|
Button |
Displays the Report screen that shows the measurement |
|
results of the current application and other information. |
|
|
|
|
Report |
|
|
|
|
|
|
|
When you press the dial-button, the menu items that are |
|
|
available in the current scan mode are shown on screen. |
|
Dial-button |
Rotating the EzExam dial-button to the right selects the menu |
|
|
one row above the current menu selected, and rotating it to the |
EzExam |
|
left selects the menu one row below. |
|
|
|
|
Button |
Allows the user to enter a Body Marker over an image. |
|
|
|
|
|
Adjusts the angle of the sample volume in Spectral Doppler |
|
|
mode. It is also used to adjust the Arrow’s angle or the probe |
|
Dial-button |
angle for a Body Marker. |
|
|
In 3D View, turning this dial-button will rotate the image around |
|
|
the x-axis. |
|
|
|
|
|
Enter or exit PW Spectral Doppler Mode. Rotate this dial- |
|
Dial-button |
button to adjust the Gain value. |
|
In 3D View, turning this dial-button will rotate the image around |
|
|
|
|
|
|
the y-axis. |
|
|
|
|
|
Enter or exit Color Doppler Mode. Rotate this dial-button to |
|
Dial-button |
adjust the Gain value. |
|
In 3D View, turning this dial-button will rotate the image around |
|
|
|
|
|
|
the z-axis. |
|
|
|

Chapter 1. Introduction 1 – 11
|
Dial-button |
Press this button to enter 2D Mode. Rotate this dial-button to |
|
|
adjust the Gain value. |
|
|
|
|
|
|
|
|
|
|
|
Button |
Enter or exit M Mode. Rotate this dial-button to adjust the Gain |
|
|
value. |
|
|
|
|
|
|
|
|
|
|
|
Button |
Enter or exit CW Spectral Doppler Mode. Available only with |
|
|
the phased array probe. |
|
|
|
|
|
|
|
|
|
|
|
Button |
Enter or exit Power Doppler Mode. |
|
|
|
|
|
|
Button |
Use this button to enter or exit 3D or 4D Mode. |
|
|
|
|
|
|
|
Press this button to turn the Quick Scan function on. The ‘Q |
|
|
Button |
scan’ mark will appear at the top of the image. This can be |
|
|
|
used in applications for all probes. |
|
|
|
|
|
|
Button |
Compares two independent |
images. The active image mode is |
|
shown on the left. |
|
|
|
|
|
|
|
|
|
|
|
Button |
Compares two independent |
images. The activated image |
|
mode is shown on the right. |
|
|
|
|
|
|
|
|
|
|
|
Switch |
Adjusts the scanning depth of the image. |
|
|
|
|
|
|
Switch |
Moves the focus to the target area for observation. |
|
|
|
|
|
|
Dial-button |
Makes the Zoom Box appear. In order to exit Zoom Mode, |
|
|
press the Exit button. |
|
|
|
|
|
|
|
|
|
|

1 – 12 HS40 Service Manual
|
Button |
Pauses an image being scanned or reactivates a paused |
||
|
image. |
|||
|
|
|||
|
|
|
||
|
|
Stands for User Key; the user can assign functions they need |
||
|
Button |
to each button. The function for each button can be assigned |
||
|
|
in |
Setup > Customize > Buttons. |
|
|
|
|
||
|
|
The user can select, set up, and use Set/Set, Set/Exit, or |
||
|
|
Exit/Set. The function for each button can be assigned in |
||
|
|
Setup > Customize > Buttons. |
||
|
Button |
– |
Set: Selects an item or value using the trackball. |
|
|
|
Alternatively, this can be used to change the function of the |
||
Set/Exit |
|
|
||
|
|
trackball. |
||
|
|
|
||
|
|
– |
Exit: Exits the function currently being used and returns to |
|
|
|
|
the previous screen. |
|
|
|
|
||
|
Button |
Deletes text, Arrows, Body Markers, measurement results, |
||
Clear |
etc., displayed on an image. |
|||
|
||||
|
|
|
||
|
|
|
||
|
Button |
In Scan Mode, an arrow-shaped pointer appears on the |
||
Pointer |
screen. |
|||
|
||||
|
|
|
||
|
Button |
Changes to a different function that can be supported by the |
||
Change |
current trackball setting. |
|||
|
||||
|
|
|
||
|
|
|
||
|
Button |
Allows the user to place text over an image. |
||
|
|
|
||
Calculator |
Button |
Starts measurements by application. |
||
|
|
|
||
|
|
|
||
|
Trackball |
Moves the cursor on the screen. Also, scrolls through Cine |
||
|
images. |
|||
|
|
|||
Trackball |
|
|
|
|
|
|
|
|
|
Chapter 1. Introduction 1 – 13
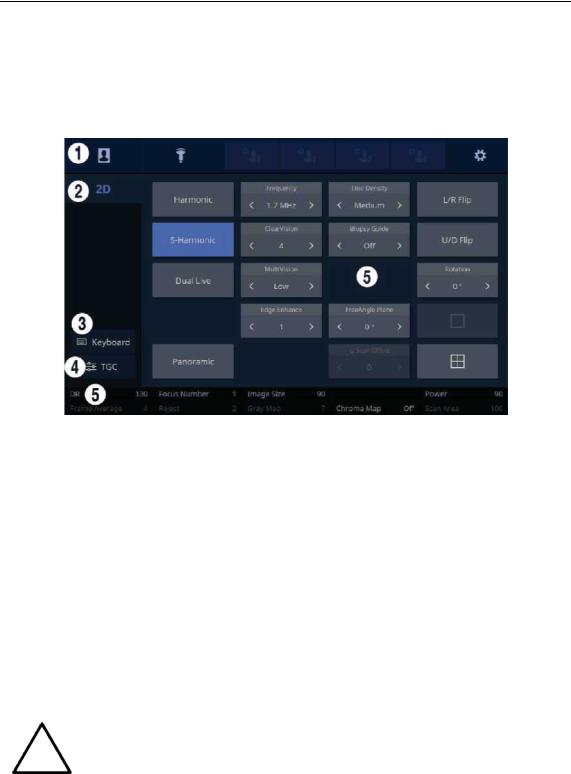
1.2.2.2. Touch Screen
The touch screen is an operating tool that can be touched by the user to input data. The functio ns that are available in the current mode are shown in the form of buttons or a dial-button.
■ Touch Screen Layout
[Figure 1.5 Touch Screen Display]
These buttons are always displayed on the touch screen. Buttons that are in use are shown in blue and buttons that cannot be used are deactivated.
Patient |
The Patient Information screen will appear, |
where you can select a |
|
Patient ID in the list or enter new patient information. |
|||
|
|||
|
|
||
Probe |
It displays the Probe Selection screen where you can select and |
||
modify the probe and application. |
|
||
|
|
||
|
|
|
|
Quick Preset |
It shows up to 4 Preset buttons configured |
at Setup. |
|
1–4 |
|||
|
|
||
|
|
||
Setup |
The general system settings that do not have a direct bearing on |
||
imaging are explained below. |
|
||
|
|
||
|
|
|
|
For further details about setting up Quick Preset, please refer to ‘Setup > System > Quick Preset’ in ‘Chapter 3. Utilities’.
NOTE
 Loading...
Loading...