Page 1

Z Vent Ventilator Operator’s Guide
9650-002360-01 Rev. A
Page 2

The issue date for the ZOLL Ventilator Operator’s Guide (REF 9650-002360-01 Rev. A) is September, 2018.
Copyright © 2018 ZOLL Medical Corporation. All rights reserved. ZOLL, Z Vent, and Smart Help are
trademarks and/or registered trademarks of ZOLL Medical Corporation in the United States and/or other
countries. All other trademarks are the property of their respective owners.
Masimo Pulse Oximeter
This device uses Masimo SET® technology to provide continuous pulse oximeter and heart rate monitoring and
is covered under one or more of the following U.S.A. patents: 5,758,644, 5,823,950, 6,011,986, 6,157,850,
6,263,222, 6,501,975 and other applicable patents listed at www.masimo.com/patents.htm
.
Limited Copyright Release
Permission is hereby granted to any military/governmental agency to reproduce all materials furnished herein
for use in a military/governmental service training program and/or other technical training program.
Page 3

Contents
Chapter 1 General Information
Product Description ............................................................................................................ 1-1
How to Use this Guide ........................................................................................................ 1-1
Operator’s Guide Updates .................................................................................................. 1-2
Unpacking........................................................................................................................... 1-2
Assembly ............................................................................................................................1-2
Product Symbols.................................................................................................................1-2
Symbols on the Z Vent Ventilator Graphical User Interface................................................ 1-5
Conventions........................................................................................................................ 1-7
Abbreviations ...................................................................................................................... 1-8
Indications for Use .............................................................................................................. 1-9
Ventilation ...................................................................................................................1-9
Pulse Oximetry (SpO2) ............................................................................................... 1-9
Features ...................................................................................................................... 1-9
Warnings........................................................................................................................... 1-10
General .....................................................................................................................1-10
Ventilator ................................................................................................................... 1-10
Pulse Oximeter ......................................................................................................... 1-11
Batteries .................................................................................................................... 1-12
User Safety ............................................................................................................... 1-12
Patient Safety ........................................................................................................... 1-12
MR Conditional Equipment ....................................................................................... 1-13
Cautions ....................................................................................................................1-14
FDA Tracking Requirements............................................................................................. 1-14
Notification of Adverse Events .................................................................................. 1-15
Software License .............................................................................................................. 1-15
NO IMPLIED LICENSE ...................................................................................... 1-15
Limited Warranty ....................................................................................................... 1-15
Technical Support ............................................................................................................. 1-16
Returning a Ventilator for ZOLL Service ............................................................ 1-17
Chapter 2 Product Overview
Z Vent Ventilator Description .............................................................................................. 2-2
Main Features ............................................................................................................. 2-2
Controls and Indicators ............................................................................................... 2-3
Controls ...............................................................................................................2-3
Indicators ............................................................................................................. 2-4
Display Screen ............................................................................................................ 2-4
Message Area ...................................................................................................... 2-5
Parameter Windows ............................................................................................ 2-5
Shared Icon Area ................................................................................................. 2-5
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide i
Page 4

Contents
Auxiliary Parameter Boxes .................................................................................. 2-5
Fresh Gas/Emergency Air Intake and Attachments .................................................... 2-6
Oxygen Reservoir Bag Assembly ........................................................................ 2-6
Top Panel ................................................................................................................... 2-7
Pulse Oximeter Compatibility ...................................................................................... 2-8
Power Sources ........................................................................................................... 2-8
Operating Using External DC Power ................................................................... 2-8
Operating Using Battery Power ........................................................................... 2-8
Pneumatic Design ....................................................................................................... 2-9
Oxygen Input: High Pressure Gas Supply ........................................................................ 2-10
High Pressure Oxygen Supply Hose ................................................................. 2-10
Patient Circuits.................................................................................................................. 2-12
Intended Use ............................................................................................................ 2-13
Use of Heat and Moisture Exchangers ..................................................................... 2-13
Attaching a Patient Circuit to the Ventilator .............................................................. 2-14
Specifications ............................................................................................................2-15
Pediatric/Adult, 6 ft Patient Circuit ..................................................................... 2-15
Pediatric/Adult, 12 ft Patient Circuit ...................................................................2-15
Infant/Pediatric, 6 ft Patient Circuit .................................................................... 2-15
Infant/Pediatric, 12 ft Patient Circuit .................................................................. 2-16
Chapter 3 Setting Up the Z Vent Ventilator
1. Attach the Patient Circuit ................................................................................................ 3-2
2. Attach the High Pressure Oxygen Supply (Optional)...................................................... 3-3
3. Inspect Fresh Gas/Emergency Air Intake Filters ............................................................ 3-3
4. Connect Fresh Gas/Emergency Air Intake Attachments (Optional)................................ 3-4
3-Liter Reservoir Bag Assembly ................................................................................. 3-4
Bacterial/Viral (BV) Filter ............................................................................................ 3-5
Chemical/Biological C2A1 Filter ................................................................................. 3-5
5. Select the Ventilator’s Power Source.............................................................................. 3-6
Connecting the Power Supply .................................................................................... 3-7
6. Power On the Ventilator.................................................................................................. 3-8
7. Select Start Menu Option ................................................................................................ 3-8
8. Change Operating Mode (Optional)................................................................................ 3-9
9. Change Parameter Values ............................................................................................ 3-10
10. Perform Operational Test ............................................................................................ 3-11
11. Attach the Pulse Oximeter (Optional).......................................................................... 3-11
12. Attach Patient..............................................................................................................3-12
Chapter 4 Using the Z Vent Ventilator
Initial Operation with Default Parameter Settings ............................................................... 4-2
Default Parameter Settings for Adult, Pediatric and Mask CPAP ............................... 4-2
Adult Default Parameter Setting Values .............................................................. 4-2
Making a Choice From the Start Menu ....................................................................... 4-3
ii www.zoll.com 9650-002360-01 Rev. A
Page 5

Contents
Changing Parameter Settings............................................................................................. 4-4
Navigating the Parameter Windows Using Parameter Buttons .................................. 4-5
Changing a Parameter Setting ................................................................................... 4-6
Example 1 — Changing a Primary Parameter ..................................................... 4-6
Example 2 — Changing a Secondary Parameter ................................................ 4-6
Example 3— Changing a Context Menu Parameter ............................................ 4-6
Saving Changed Parameters for Future Use...................................................................... 4-7
Using the Last Settings Enabled on the Ventilator.............................................................. 4-7
Mode Parameter Window Options......................................................................................4-8
Primary Parameter ...................................................................................................... 4-8
Secondary Parameters and Alarm Thresholds/Limits ................................................ 4-8
Breath Target ....................................................................................................... 4-8
Leak Compensation ............................................................................................. 4-9
Context Menu ............................................................................................................. 4-9
Mode Parameter Window Reference ........................................................................ 4-10
BPM Parameter Window Options ..................................................................................... 4-11
Secondary Parameters and Alarm Thresholds/Limits .............................................. 4-11
Context Menu ........................................................................................................... 4-11
Control Parameter (I:E or Ti) ............................................................................. 4-12
Rise Time ........................................................................................................... 4-12
Cycle Off % Parameter ...................................................................................... 4-13
Spont Ti Limit Parameter ................................................................................... 4-13
BPM Parameter Window Reference ......................................................................... 4-14
Vt Parameter Window Options .........................................................................................4-15
Secondary Parameters and Alarm Thresholds/Limits .............................................. 4-15
Context Menu ........................................................................................................... 4-16
Vt Parameter Window Reference ............................................................................. 4-17
PIP Parameter Window Options ....................................................................................... 4-17
Secondary Parameters and Alarm Thresholds/Limits .............................................. 4-17
Context Menu ........................................................................................................... 4-18
PIP Parameter Window Reference ........................................................................... 4-19
FIO2 Parameter Window Options..................................................................................... 4-20
Secondary Parameters and Alarm Thresholds/Limits .............................................. 4-20
Context Menu ........................................................................................................... 4-20
FIO2 Parameter Window Reference ......................................................................... 4-21
SpO2 Parameter Window Options.................................................................................... 4-21
Secondary Parameters and Alarm Thresholds/Limits .............................................. 4-22
Context Menu ........................................................................................................... 4-22
SpO2 Parameter Window Reference ....................................................................... 4-23
HR (Heart Rate)................................................................................................................4-24
Secondary Parameters and Alarm Thresholds/Limits .............................................. 4-24
Context Menu ........................................................................................................... 4-24
HR Parameter Window Reference ........................................................................... 4-25
Manual Breath .................................................................................................................. 4-26
Plateau Pressure .............................................................................................................. 4-26
Plateau Pressure Maneuver ..................................................................................... 4-26
Popup Messages .............................................................................................................. 4-27
Popup Message List ................................................................................................. 4-29
Transitions ........................................................................................................................4-36
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide iii
Page 6

Contents
Using the Main Menu........................................................................................................4-36
Alarm Configuration .................................................................................................. 4-36
Powerup .................................................................................................................... 4-37
Specifying Powerup Settings ............................................................................. 4-37
Specifying Custom Settings ............................................................................... 4-37
Specifying a Language ...................................................................................... 4-38
LCD Contrast ............................................................................................................ 4-38
LCD Brightness ......................................................................................................... 4-38
UTC Offset ................................................................................................................ 4-39
Unit Info .................................................................................................................... 4-39
Alarm History ............................................................................................................ 4-40
Chapter 5 Alarms
Alarm Overview ..................................................................................................................5-1
Muting Alarms..................................................................................................................... 5-3
Alarm Priorities ................................................................................................................... 5-3
High Priority .........................................................................................................5-3
Medium Priority .................................................................................................... 5-3
Low Priority (Advisory) ......................................................................................... 5-3
Popup Messages ................................................................................................................ 5-4
Alarm Types........................................................................................................................ 5-4
Patient Safety Alarms ................................................................................................. 5-4
Environmental and Use Alarms .................................................................................. 5-7
Self-Check Alarms ...................................................................................................... 5-9
Managing Alarms.............................................................................................................. 5-11
Alarm Muting ............................................................................................................. 5-12
Start Up Muting .................................................................................................. 5-12
Preemptive Muting ............................................................................................. 5-12
Disabling Alarms from the Alarm Configuration Menu .............................................. 5-14
Patient Detect Mode ................................................................................................. 5-14
Backup Modes .......................................................................................................... 5-15
Apnea Backup Mode ......................................................................................... 5-15
Alarm Summary ................................................................................................................ 5-16
High Priority Alarms .................................................................................................. 5-16
Medium Priority Alarms ............................................................................................. 5-22
Low Priority Alarms ................................................................................................... 5-31
Chapter 6 Operating Environments
Using the Ventilator in Harsh Environments ....................................................................... 6-1
Airborne Particulates .................................................................................................. 6-1
Extreme Temperature Environments .......................................................................... 6-2
Operating at High Temperatures ......................................................................... 6-2
Operating at Low Temperatures .......................................................................... 6-2
Altitude ........................................................................................................................ 6-3
Rain and Snow ........................................................................................................... 6-3
iv www.zoll.com 9650-002360-01 Rev. A
Page 7

Using the Ventilator in Hazardous Environments................................................................ 6-3
Bacterial/Viral Filter Use ............................................................................................. 6-4
Chemical/Biological Filter Use .................................................................................... 6-4
Check Valve on Patient Circuit when in Hazardous Environments ............................ 6-5
Using the Ventilator in an MRI Environment ....................................................................... 6-6
Chapter 7 Maintenance
Inspecting the Ventilator ..................................................................................................... 7-1
Cleaning.............................................................................................................................. 7-2
Post-Contaminated Environment Cleaning ................................................................. 7-2
Fresh Gas/Emergency Air Intake Filters............................................................................. 7-3
Inspecting and Replacing the Foam Filter .................................................................. 7-3
Inspecting and Replacing the Disk Filter .................................................................... 7-3
Replacing Ventilator Filters .................................................................................................7-4
Replacing the Foam Filter ........................................................................................... 7-4
Replacing the Disk Filter ............................................................................................. 7-4
Battery Maintenance........................................................................................................... 7-5
Battery Storage ........................................................................................................... 7-7
Ventilator Storage ....................................................................................................... 7-7
Battery Replacement and Shipping Regulations ........................................................ 7-8
Calibration Checks..............................................................................................................7-8
Electrical Safety Check....................................................................................................... 7-8
Troubleshooting .................................................................................................................. 7-9
Contents
Appendix A Specifications
General ...............................................................................................................................A-1
Pulse Oximeter ...................................................................................................................A-3
Device Classification...........................................................................................................A-3
Appendix B Accessories
Appendix C Pulse Oximeter Principles
Appendix D Troubleshooting Patient Circuits
Troubleshooting Procedure.................................................................................................D-1
Index
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide v
Page 8

Page 9

This chapter provides general information about the ZOLL Z Vent™ ventilator and the
ZOLL Z Vent Ventilator Operator’s Guide, which we provide with this product. Specifically,
this chapter provides:
• A brief description of the Z Vent ventilator.
• Information about this manual (ZOLL Z Vent Ventilator Operator’s Guide).
• A table that describes the symbols that appear on the Z Vent ventilator and in this manual.
• The Z Vent ventilator Indications for Use.
• A list of Warnings and Cautions regarding the use of the ventilator.
• Information regarding FDA tracking requirements, and the product’s warranty and software
license.
• How to contact ZOLL Medical Corporation for service to this product.
Product Description
The ZOLL Z Vent ventilator is a small, extremely durable, full-featured portable mechanical
ventilator designed to operate in hospitals or severe and under-resourced environments. It can
be used in prehospital, aeromedical, field hospital and hospital settings.
Chapter 1
General Information
How to Use this Guide
The ZOLL Z Vent Ventilator Operator’s Guide provides information that users need for the safe
and effective use and care of the ventilator. It is important that all persons using this device read
and understand all the information contained within.
Please thoroughly read the warnings section.
Procedures for device care are located in Chapter 7, “Maintenance”.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-1
Page 10

General Information
Operator’s Guide Updates
An issue or revision date for this manual is shown on the front cover. If more than 3 years have
elapsed since this date, contact ZOLL Medical Corporation to determine if additional product
information updates are available.
All users should carefully review each manual update to understand its significance and then
file it in its appropriate section within this manual for subsequent reference.
Product documentation is available through the ZOLL web site at www.zoll.com. From the
Products menu, choose Product Manuals.
Unpacking
Carefully inspect each container for damage. If the shipping container or cushion material is
damaged, keep it until the contents have been checked for completeness and the device has
been checked for mechanical and electrical integrity. If the contents are incomplete, if there is
mechanical damage, or if the ventilator does not pass its Self-Check test when turned on,
U.S.A. customers should call ZOLL Medical Corporation (1-978-421-9655). Customers
outside of the U.S.A. should contact the nearest ZOLL authorized representative. If the
shipping container is damaged, also notify the carrier. If there is no apparent sign of mechanical
damage, read instructions contained within this manual before attempting operation.
Assembly
The device only requires that you attach the breathing circuit to begin ventilation using either
battery or external power. Both the ventilator and breathing circuit are supplied clean and are
ready for use on a patient.
Product Symbols
The following symbols appear on the ventilator or in this manual:
Symbol Description
Off
On
Direct Current: Identifies the location to connect external DC Power.
Mute / Cancel: Identifies button which mutes the active alarms or cancels
the parameter selection.
1-2 www.zoll.com 9650-002360-01 Rev. A
Page 11

Symbol Description
Accept /Select: Identifies button which accepts the parameter selection.
ESD: Warns that connector pins should not be touched.
Identifies the dial that allows the selection of parameter values.
General Information
Do Not Re-Use: This item should not be re-use
Do Not Discard: Follow all governing regulations regarding the disposal
of any part of this medical device.
Serial Number: Numbers following “SN” indicate the serial number.
Defibrillation Proof: Indicates the degree of protection against electrical
shock.
BF Symbol: Protection against electric shock, Type B with floating
(F-type) parts.
MR Symbol: Identifies the use of the device’s ability to perform in a MRI
environment.
d.
Power Input Orientation: Locates the DC input identifying its point of
insertion.
Manufacturer: This symbol identifies the name and address of the
manufacturer.
Manufacturer Date: Manufacturer Date Symbol identifies the device’s
date of manufacture.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-3
Page 12
General Information
280 - 600 kPa
(40 - 87 PSIG)
O
2
NOT OCC
Symbol Description
Consult Instruction: Consult the instructions for use or operation manual.
Refer to instruction manual.
Menu icon. This icon identifies the button that, when pressed, displays a
menu of options that you can select to configure the ventilator.
High Pressure O
Connector (top faceplate icon).
2
Exhalation Valve (top faceplate icon).
Exhaust Do Not Occlude (top faceplate icon).
Transducer (top faceplate icon).
Gas Output -- Patient Circuit Connector (top faceplate icon).
Contains Phthalates
Non-Sterile
1-4 www.zoll.com 9650-002360-01 Rev. A
Page 13

General Information
+
LC
LC
_ _ _
_ _
Symbols on the Z Vent Ventilator Graphical User Interface
The following symbols appear on the ventilator’s Graphical User Interface (GUI):
Symbol Description
Heart: Provides indication that the pulse oximeter is in use.
Alarm Bell: Identifies the number of off-screen alarms.
Alarm Bell Outline: Identifies alarm limit settings; Identifies the on-screen
alarms.
O
reservoir mode is in use.
2
Leak Compensation (LC) feature is ON.
Leak Compensation Feature is OFF.
Patient Detect Mode: Backup Ventilation Started.
Not receiving a reading from the pulse oximeter.
Attention: High Priority Alarm Active.
Caution: Medium Priority Alarm Active.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-5
Page 14

General Information
EXT
BATT
off
on
Symbol Description
Warning: Low Priority Alarm Active.
Mute: Active Alarm Audible Signal Muted.
Speaker: Active Alarm Audible Signal.
Oxygen Supply: Oxygen Supply Connected.
External Power: Indicates the device is operating using an external
power source.
No External Power: Indicates the device is operating without an external
power source.
Internal Battery: Provides indication of battery capacity and charging.
Indicates that an external battery is powering the ventilator.
No Internal Battery: Indicates when internal battery is not an available
power source.
Head with Mask: the device is in the non-invasive positive pressure
ventilation modes, CPAP or BL, with Leak Compensation turned on.
Feature OFF -- feature or alarm not selected.
Feature ON -- feature or alarm has been selected.
1-6 www.zoll.com 9650-002360-01 Rev. A
Page 15

General Information
srch
stby
Symbol Description
Search (Pulse oximeter searching for a patient signal.)
Standby (Pulse oximeter in standby.).
Conventions
This guide uses the following conventions:
• Within text, the names and labels for physical buttons and soft-keys appear in boldface type
(for example, “Press the Accept button”).
• This guide uses uppercase italics for text messages displayed on the screen
(for example, EXTERNAL POWER FAILURE).
Warning! Warning statements alert you to conditions or actions that can result in personal injury
or death.
Caution Caution statements alert you to conditions or actions that can result in damage to the device.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-7
Page 16

General Information
Abbreviations
A/C - Assist/Control ID - Internal Diameter
AEV - Automatic Electrical Ventilator L - Liters
ACLS - Advanced Cardiac Life Support LC - Leak Compensation
ALS - Advanced Life Support LCD - Liquid Crystal Display
ATL S - Advanced Trauma Life Support LED - Light Emitting Diode
ACV - Assist-Control Ventilation LPM - Liters Per Minute
AMC - Alarm Message Center ml - Milliliters
APOD - Advanced Probe Off Detection mm - Millimeter
ATP D - Ambient Temperature and Pressure Dry MRI - Magnetic Resonance Imaging
b/min - Beats Per Minute O
B/V - Bacterial/Viral Filter P
BL - Bilevel positive airway pressure PEEP - Positive End Expiratory Pressure
BPM - Breaths per Minute PIP - Peak Inspiratory Pressure
cm H2O - Centimeters of Water PPV - Positive-Pressure Ventilation
CPAP - Continuous Positive Airway Pressure PS - Pressure Support
CPR - Cardiopulmonary Resuscitation psig - Pounds per Square Inch Gage
CPU- Central Processor Unit RF - Radio Frequency
dBA - Decibel RGA # - Returned-Goods-Authorization number
DISS - Diameter Index Safety System RTC - Real Time Clock
EMC - Electromagnetic Compatibility SIMV - Synchronized Intermittent Mandatory
EMV - Emergency Medical Ventilator SPM - Smart Pneumatic Module
ESD - Electrostatic Discharge SpO
FIO
Fraction of Inspired Oxygen USP - United States Pharmacopoeia
2 -
HME - Heat and Moisture Exchanger VAC - Volts AC
- oxygen
2
- Airway Pressure
aw
Ventilation
- oxyhemoglobin saturation,%
2
HMEF - Heat and Moisture Exchanger/Bacterial Viral
filter combined
HP O2 - High Pressure Oxygen VT - Tidal Volume
Hz - Hertz (as in frequency, cycles per second) WOB – Work Of Breathing
I:E- Inspiratory/Expiratory Ratio
VDC - Volts DC
1-8 www.zoll.com 9650-002360-01 Rev. A
Page 17

Indications for Use
Ventilation
The ZOLL Z Vent ventilator is indicated for use in the management of infant through adult
patients weighing greater than or equal to 5 kg with acute or chronic respiratory failure or
during resuscitation by providing continuous positive-pressure ventilation. The ZOLL Z Vent
ventilator is appropriate for use in hospitals, outside the hospital, during transport and in severe
environments where they may be exposed to rain, dust, rough handling, and extremes in
temperature and humidity. With an appropriate third-party filter in place, they may be operated
in environments where chemical and/or biological toxins are present. When marked with an
“MRI conditional” label, the ZOLL Z Vent ventilator is suitable for use in an MRI environment
with appropriate precautions. The ZOLL Z Vent ventilator is intended for use by skilled care
providers with knowledge of mechanical ventilation, emergency medical services (EMS)
personnel with a basic knowledge of mechanical ventilation, and by first responders under the
direction of skilled medical care providers.
Pulse Oximetry (SpO2)
General Information
Features
The ZOLL Z Vent ventilator pulse oximeter with Masimo SET technology is intended for use
for continuous noninvasive monitoring of the oxygen saturation of arterial hemoglobin (SpO
and pulse rate. The pulse SpO
oximeter and accessories are indicated for use with adult and
2
),
2
pediatric patients during both no motion and motion conditions, and for patients who are well
or poorly perfused, in hospitals, hospital-type facilities, or in mobile environments.
• Portable ventilator that you can use in the hospital, aeromedical and ground transport, mass
casualty situations, and extreme environments.
• Multiple modes of ventilation for use with acute or chronic respiratory failure in both
intubated and non-intubated patients.
• Intuitive user interface minimizes user training and protects existing settings from
inadvertent contact and manipulation.
• Smart Help™ messages guide the user through on-screen commands when responding to
alarms.
• Lightweight < 10 lbs. (4.4 kg) — for easy transport.
• Rechargeable battery provides over 10 hours of operation (at factory default with pulse
oximeter operating).
• Operating temperature range for extreme conditions: -26 C to 55 C (-15 F to 131F).
• Altitude compensation from - 685.8 m to 7620 m (- 2,250 to 25,000 ft).
• Self-contained system able to operate with or without external oxygen.
• Gas manifold design allows operation with both high and low-pressure oxygen sources. All
oxygen is delivered to the patient breathing circuit.
• Sealed gas path with chemical/biological filter connected to assure safe breathing gas
supply.
• Case and control panel protects components from weather and fluids.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-9
Page 18

General Information
Warnings
General
• The Z Vent ventilator is intended for use by qualified personnel only. You should read this
• Before using the ventilator on a patient, you must test the device in its normal configuration
• Do not modify this equipment without authorization of the manufacturer.
• This operator’s guide is not meant to supersede any controlling operating procedure
• Follow all governing regulations regarding the disposal of any part of this medical device,
Ventilator
• The Z Vent ventilator can operate from its internal battery or from an external power source.
• The use of accessories and cables other than those sold by ZOLL may result in increased
• Portable and mobile RF communication equipment may affect the performance of this
• The ventilator may cause radio interference or may disrupt the operation of nearby
• Do not connect to an electrical outlet controlled by a wall switch or dimmer.
• The protection against defibrillator depends on the use of accessories (including Pulse
• Grounding:
• As with all medical equipment, carefully route the patient circuit, patient cabling, and
manual before using the device.
to ensure proper operation.
regarding the safe use of assisted ventilation.
the handling of materials contaminated by body fluids, and shipment of the Li-ION
batteries.
When using external power, position the power cord to avoid accidental disconnect.
emissions or decreased immunity of this device.
device. We describe the EMC performance for this device in Appendix A Specifications of
this guide.
equipment. It may be necessary to take mitigation measures, such as re-orienting or
relocating of the device or shielding the location.
Oximeter) that are specified by ZOLL.
• Do not under any circumstances remove the grounding conductor from the power
plug.
• Do not use extension cords or adapters of any type. The power cord and plug must
be intact and undamaged.
• If there is any doubt about the integrity of the protective earth conductor or power
supply, operate on internal battery power.
external power cords to reduce the possibility of patient entanglement or strangulation.
The product design includes materials with phthalates in the pressure lines of
both the manifold design and patient circuit. Patient mask accessories used
with the device also are made with materials containing phthalates.
Phthalates are NOT present in the inspiratory line (Gas Hose) of the patient
circuit.
1-10 www.zoll.com 9650-002360-01 Rev. A
Page 19

• Do not use in MRI environment unless MRI marking is present.
• Do not operate the ventilator on a patient when the USB port is connected to any other
device (the USB port is only for servicing the ventilator).
• The ZOLL-supplied patient circuit’s labeling provides the resistance and compliance values
for the circuits under normal operating conditions. If added accessories are used
(e.g. HME, filters etc.), you should assure they do not degrade the performance of the
device.
Pulse Oximeter
• Do not use the pulse oximeter as an apnea monitor.
• A pulse oximeter should be considered an early warning device. As a trend towards patient
deoxygenation is indicated, blood samples should be analyzed by a laboratory co-oximeter
to completely understand the patient’s condition.
• Measurements: if the accuracy of any measurement does not seem reasonable, first check
the patient’s vital signs by alternate means and then check the pulse oximeter for proper
functioning.
Inaccurate measurements may be caused by:
• Interfering Substances: carboxyhemoglobin may erroneously increase readings. The level of
increase is approximately equal to the amount of carboxyhemoglobin present. Dyes, or any
substance containing dyes, that change usual arterial pigmentation may cause erroneous
readings.
• Alarms: Check alarm limits each time the pulse oximeter is used to ensure that they are
appropriate for the patient being monitored.
• Loss of pulse signal can occur in any of the following situations:
General Information
• Incorrect sensor application or use.
• Significant levels of dysfunctional hemoglobin (e.g. carboxyhemoglobin or
methemoglobin).
• Intra-vascular dyes such as indocyanine green or methylene blue.
• Exposure to excessive illumination, such as surgical lamps (especially ones with a
xenon light source), bilirubin lamps, fluorescent lights, infrared heating lamps, or
direct sunlight (exposure to excessive illumination can be corrected by covering the
sensor with a dark or opaque material).
• Excessive patient movement.
• Venous pulsations.
• Placement of a sensor on an extremity with a blood pressure cuff, arterial catheter,
or intravascular line.
• The pulse oximeter is defibrillator-proof. The pulse oximeter can be used during
defibrillation, but the readings may be inaccurate for a short time.
• The sensor is too tight.
• Excessive illumination from light sources such as a surgical lamp, a bilirubin lamp,
or sunlight.
• A blood pressure cuff is inflated on the same extremity as the one with an SpO
2
sensor attached.
• The patient has hypotension, severe vascoconstriction, severe anemia, or
hypothermia.
• Arterial occlusion proximal to the sensor.
• The patient is in cardiac arrest or is in shock.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-11
Page 20

General Information
• Sensors:
• Do not use the pulse oximeter sensor during magnetic resonance imaging (MRI) scanning.
Batteries
• Before use, carefully read the Masimo LNCS
• Use only Masimo oximetry sensors for SpO
®
sensor directions for use.
measurements. Other oxygen
2
transducers (sensors) may cause improper performance.
• Tissue damage can be caused by incorrect application or use of an LNCS sensor for
example, by wrapping the sensor too tightly. Inspect the sensor site as directed in
the sensor Directions for Use to ensure skin integrity and correct positioning and
adhesion of the sensor.
• Do not damage LNCS sensors. Do not use an LNCS sensor with exposed optical
components. Do not immerse the sensor in water, solvents, or cleaning solutions
(The sensors and connectors are not waterproof). Do not sterilize by irradiation,
steam, or ethylene oxide. See the cleaning instructions in the directions for reusable
Masimo LNCS sensors.
• Do not use damaged patient cables. Do not immerse the patient cables in water,
solvents, or cleaning solutions (the patient cables are not waterproof). Do not
sterilize by irradiation, steam, or ethylene oxide. See the cleaning instructions in
the directions for reusable Masimo patient cables.
Inducing current could potentially cause burns. The pulse oximeter may affect the MRI
image and the MRI unit may affect the accuracy of the dosimetry measurements.
• Only use the power supply provided with the device. Use of any other power supply could
• If you witness a battery or the battery compartment starting to balloon, swell up, smoke, or
User Safety
• Electric shock hazard: Do not remove equipment covers. You may only perform
• Possible explosion hazard if used in the presence of flammable anesthetics or other
• This device is not intended for use in explosive atmospheres.
• Pins of connectors identified with the ESD warning symbol should not be touched. Always
Patient Safety
• To ensure patient electrical isolation, connect only to other equipment with electronically
• Do not place the device or external power supply in any position that might cause it to fall
• Never service the ventilator while in use with a patient.
cause damage or create a fire and/or destroy the battery and device.
feel excessively hot, turn off the device, disconnect external power, and observe it in a safe
place for approximately 15 minutes and send the device for service. Never puncture or
disassemble the battery packs or cells.
maintenance procedures specifically described in this manual. Refer all servicing to
ZOLL or a ZOLL-authorized service center.
flammable substances in combination with air, oxygen-enriched environments, or nitrous
oxide.
use precautionary procedures with ESD-sensitive connections.
isolated circuits.
on the patient. Do not lift the device by the power cord, patient circuit, or pulse oximeter
patient cable.
1-12 www.zoll.com 9650-002360-01 Rev. A
Page 21

MR Conditional Equipment
• Failure to follow all instructions can result in MRI artifacts, injury to the patient or
user, or malfunction of the device.
• You must follow all safety procedures that are in effect for the MRI Environment. Do
not use the ventilator in an MRI Environment with greater than 3T magnetic force.
• You must secure the device to a suitable MRI-compatible cart -- ZOLL MRI Roll
Stand; Optional IV Arm Assembly.
• You must place the ventilator behind the 2000 Gauss field line.
• The ventilator must be attended by a person with no other responsibility than
monitoring the device and patient while in the MRI Environment.
• You must visually monitor the ventilator for alarms at all times -- during imaging,
the alarms may not be audible beyond the area immediately adjacent to the MRI.
• Danger! Possible Missile Projection.
• DO NOT position any person between the bore entrance and an unsecured cart or
device.
• Lock the wheels when the rolling stand is in place.
• We recommend that you tether the rolling stand in place when in the MRI
Environment.
• Place the ventilator and stand in its position before the patient is positioned on the
scanner table and advanced into the bore.
• Remove the patient from the MRI Environment before removing the ventilator and
roll stand.
• Unapproved device apparatus shall NOT be allowed in the MRI Environment,
including:
• Pulse Oximeters sensors and cabling.
• External AC/DC Power Supply.
• Rolling Cart Breathing Circuit Arm.
• Active Humidification and associated support apparatus.
• Ensure proper configuration of the ventilator.
• DO NOT attach the pulse oximeter sensor to the patient and remove it from the
device.
• The ventilator should run only on battery power in the MRI Environment
-- DO NOT use an external AC/DC power supply.
• The ventilator’s battery should be fully charged before entering the MRI
Environment.
• Oxygen Supply -- an aluminum, non-magnetic cylinder and oxygen hose must
provide the oxygen supply.
• Ensure proper operation of the ventilator’s breathing system.
• 12 ft patient circuits are available for use with the ventilator -- the additional length
enables a suitable separation between the ventilator and the bore opening.
(Adult/Pediatric Wye Patient Circuit; Pediatric/Infant Wye Patient Circuit).
• The extended tubing length of a 12 ft patient circuit can result in loss of volume due
to additional tubing compressibility.
-- Set the Tubing Compliance (TC) to OFF and ensure that the patient is
receiving correct tidal volume.
-- Alternatively, calculate the TC as described by the patient circuit’s
Instructions For Use (IFU) and adjust the TC value to ensure that the patient is
receiving the correct tidal volume.
• DO NOT use the 12 ft circuit with PEEP settings below 5 (5 cm H
• Ensure that the ventilator is able to maintain PEEP -- for patients with short
expiratory times, the additional tubing length of the 12 ft circuit may affect system
behavior.
General Information
O).
2
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-13
Page 22

General Information
Cautions
• Inspect the circuit every day to ensure that there is no damage or wear that could affect its
performance. Remove fluid or other biological material from the circuit or replace the
circuit following the local standard of care.
• Federal law restricts this device to sale by or on the order of a physician.
• Only qualified biomedical equipment technicians should service the device.
• Internal components are susceptible to damage from static discharge. Do not remove device
covers.
• Possession or purchase of this device does not convey any expressed or implied license to
use the device with unauthorized sensors or cables which would, alone, or in combination
with this device fall within the scope of one or more of the patients related to this device.
ZOLL cannot ensure the proper functioning of this device if it is used with unauthorized
sensors, cables, or patient circuits.
FDA Tracking Requirements
U.S. Federal Law (21 CFR 821) requires the tracking of ventilators. Under this law, owners
of this ventilator must notify ZOLL Medical Corporation if this product is
• Received
• Lost, stolen, or destroyed
• Donated, resold, or otherwise distributed to a different organization
If any such event occurs, contact ZOLL Medical Corporation in writing with the following
information:
• Originator's organization – Company name, address, contact name, and contact phone
number
• Model number, and serial number of the ventilator
• Disposition of the ventilator (for example, received, lost, stolen, destroyed, distributed to
another organization), new location and/or organization (if known and different from
originator’s organization) – company name, address, contact name, and contact phone
number
• Date when the change took effect
Please address the information to:
ZOLL Medical Corporation
Attn: Tracking Coordinator
269 Mill Road
Chelmsford, MA 01824-04105
Fax: (978) 421-0007
Telephone: (978) 421-9655
1-14 www.zoll.com 9650-002360-01 Rev. A
Page 23

Notification of Adverse Events
As a health care provider, you may have responsibilities under the Safe Medical Devices Act
(SMDA), for reporting to ZOLL Medical Corporation, and possibly to the FDA, the occurrence
of certain events.
These events, described in 21 CFR Part 803, include device-related death and serious injury or
illness. In addition, as part of our Quality Assurance Program, ZOLL Medical Corporation
requests to be notified of device failures or malfunctions. This information is required to ensure
that ZOLL Medical Corporation provides only the highest quality products.
Software License
Note: Read this Operator’s Guide and License agreement carefully before operating the
Z Vent ventilator product.
Software incorporated into the system is protected by copyright laws and international
copyright treaties as well as other intellectual property laws and treaties. This software is
licensed, not sold. By taking delivery of and using this system, the Purchaser signifies
agreement to and acceptance of the following terms and conditions:
1. Grant of License: In consideration of payment of the software license fee which is part of
the price paid for this product, ZOLL Medical Corporation grants the Purchaser a
nonexclusive license, without right to sublicense, to use the system software in object-code
form only.
2. Ownership of Software/Firmware: Title to, ownership of, and all rights and interests in the
system software and all copies thereof remain at all times vested in the manufacturer, and
Licensors to ZOLL Medical Corporation and they do not pass to purchaser.
3. Assignment: Purchaser agrees not to assign, sublicense, or otherwise transfer or share its
rights under the license without the express written permission of ZOLL Medical
Corporation.
4. Use Restrictions: As the Purchaser, you may physically transfer the products from one
location to another provided that the software/firmware is not copied. You may not disclose,
publish, translate, release, or distribute copies of the software/firmware to others. You may
not modify, adapt, translate, reverse engineer, decompile, crosscompile, disassemble, or
create derivative works based on the software/firmware.
General Information
NO IMPLIED LICENSE
Possession or purchase of this device does not convey any express or implied license to use the
device with replacement parts which would, alone, or in combination with this device, fall
within the scope of one or more of the patents relating to this device.
Limited Warranty
ZOLL warrants the device to be free from all defects in material and workmanship for a period
of one (1) year from the date of delivery to the original purchaser.
During the warranty period, ZOLL will repair or replace the device or any part which upon
examination is shown to be defective. At its sole discretion, ZOLL may choose to supply a new
or equivalent replacement product or refund the amount of the purchase price (on the date sold
by ZOLL). To qualify for such repair, replacement, or refund, the defective device must be
returned to the ZOLL Service Center within thirty (30) days from the date that the defect is
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-15
Page 24

General Information
discovered. This warranty does not apply if the device has been repaired or modified without
the authorization of ZOLL or if the damage was caused by incorrect (off-label) use, negligence,
or an accident.
Batteries, which by their nature are consumable and subjected to environmental extremes, will
be warranted only for a period of ninety (90) days. Accessories, also consumable in usage, such
as connecting hose and breathing circuits, are not warranted.
DISCLAIMER OF IMPLIED & OTHER WARRANTIES:
THE PRECEDING WARRANTY IS THE EXCLUSIVE WARRANTY AND ZOLL MAKES
NO OTHER WARRANTY OR REPRESENTATION OF ANY KIND WHATSOEVER,
EXPRESS OR IMPLIED, WITH RESPECT TO MERCHANTABILITY, FITNESS FOR A
PARTICULAR PURPOSE, OR ANY OTHER MATTER. THE REMEDIES STATED IN THIS
DOCUMENT WILL BE THE EXCLUSIVE REMEDIES AVAILABLE TO THE
CUSTOMER FOR ANY DEFECTS OR FOR DAMAGES RESULTING FROM ANY
CAUSE WHATSOEVER AND WITHOUT LIMITATION.
ZOLL WILL NOT IN ANY EVENT BE LIABLE TO THE CUSTOMER FOR
CONSEQUENTIAL OR INCIDENTAL DAMAGES OF ANY KIND, WHETHER FOR
DEFECTIVE OR NONCONFORMING PRODUCTS, BREACH OR REPUDIATION OF
ANY TERM OR CONDITION OF THIS DOCUMENT, NEGLIGENCE, OR ANY OTHER
REASON.
Technical Support
If the ventilator requires service, contact the ZOLL Technical Support Department.
For customers In the U.S.A. For customers outside the U.S.A.
Telephone
Email
When requesting support, please provide the following information to the support
representative:
• Ventilator serial number
• Description of the problem, and service code if available
• Department using the equipment and name of the person to contact
• Purchase order to allow tracking of loan equipment
• Purchase order for a device with an expired warranty
1-800-348-9011
1-978-421-9655
techsupport@zoll.com
Call the nearest authorized ZOLL Medical Corporation
representative.
To locate an authorized service center, contact:
ZOLL Medical Corporation
269 Mill Road
Chelmsford, MA 01824
Telephone: 1-978-421-9655
techsupport@zoll.com
1-16 www.zoll.com 9650-002360-01 Rev. A
Page 25

General Information
Returning a Ventilator for ZOLL Service
Before sending a ventilator to the ZOLL Technical Support Department for repair, obtain a
service request (SR) number from the service representative.
The Li-ion battery should remain inside the ventilator or aspirator. Follow directions provided
on the return authorization form.
Pack the ventilator with its power supply in the original shipping containers
(if available) or equivalent packaging. Be sure the assigned service request (SR) number
appears on each package and follow the Shipping Regulations as described in Chapter 7 of this
manual.
Return the device to:
.
For customers Return the device to
In the U.S.A. ZOLL Medical Corporation
269 Mill Road
Chelmsford, MA 01824
Attention: Technical Support Department (SR number
Telephone: 1-978-421-9655
)
In Canada ZOLL Medical Canada Inc.
1750 Sismet Road, Unit #1
Mississauga, ON L4W 1R6
Attention: Technical Support Department (SR number)
Telephone: 1-866-442-1011
In other locations The nearest authorized ZOLL Medical Corporation
representative.
To locate an authorized service center, contact the
International Sales Department at:
ZOLL Medical Corporation
269 Mill Road
Chelmsford, MA 01824-4105
Telephone: 1-978-421-9655
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 1-17
Page 26

Page 27

Chapter 2
Product Overview
This chapter describes the Z Vent ventilator and provides more detailed descriptions of the
following:
• Main features
• Controls and indicators
• Display screen
• Fresh Gas/Emergency Air Intake and attachments
• Top Panel
• Pulse Oximeter compatibility
• Power sources
• Pneumatic design
• Oxygen Input
• Patient circuits
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-1
Page 28
Product Overview
Z Vent Ventilator Description
The sections that follow provide a detailed description of the Z Vent ventilator.
Main Features
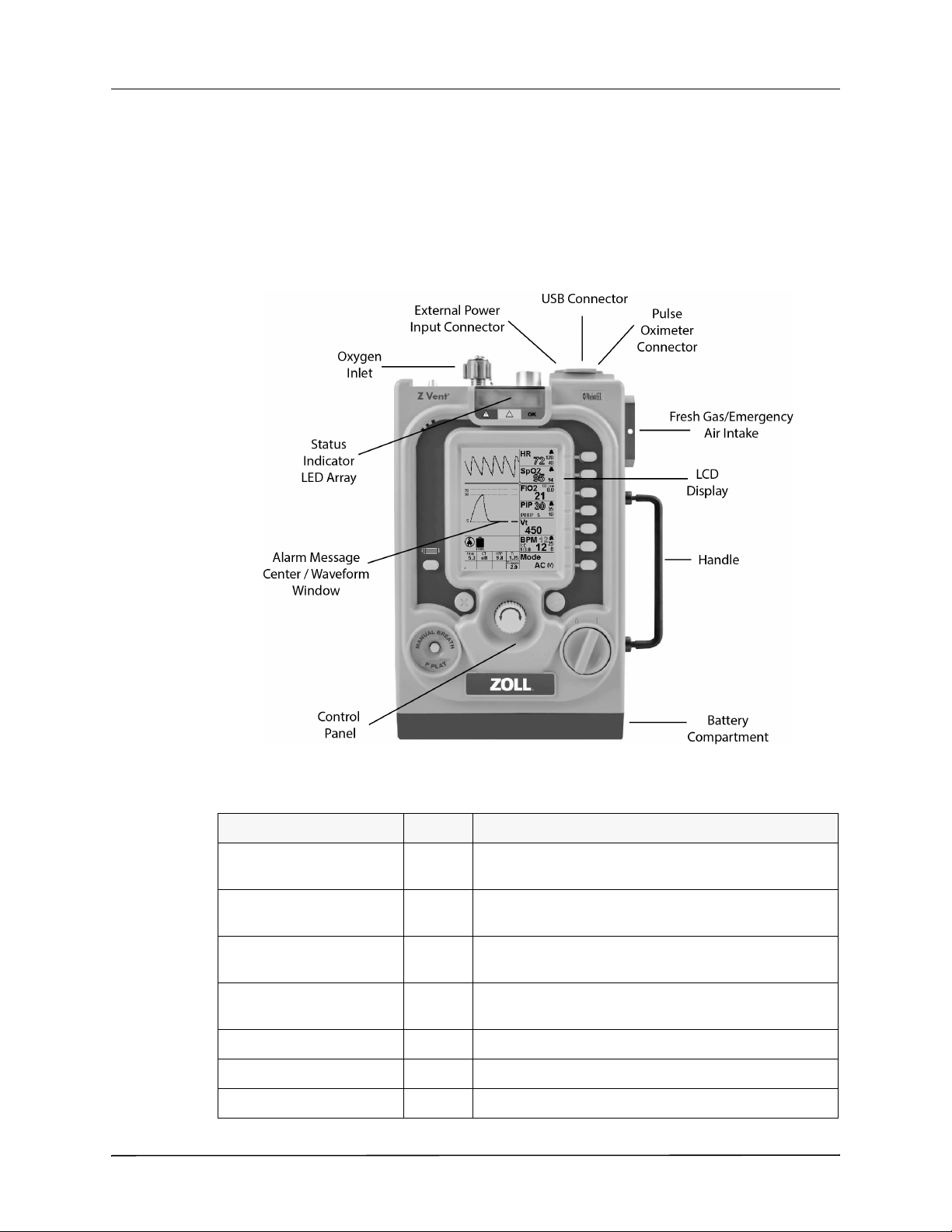
Figure 2-1 shows the ventilator’s main features.
Figure 2-1 Main Features
Item Location Description
Oxygen Inlet Top Enables connection to an external high pressure oxygen
source.
Status Indicator LED Array Top Lights to indicate ventilator status and a visible alarm
indicator.
External Power Input
Connector
USB Connector Top Enables connection to a USB compatible device for
Pulse Oximeter Connector Top Enables connection to a pulse oximeter sensor
LCD Display Front Displays settings, ventilation data, and alarm information.
Alarm Message Center Front Displays active alarms and alarm mitigation information.
2-2 www.zoll.com 9650-002360-01 Rev. A
Top Enables connection to an external power source.
servicing the ventilator.
Page 29

Product Overview
Item Location Description
Control Panel Front Provides user access to the ventilator settings.
Battery Compartment Bottom Holds the ventilator’s rechargeable Li-ion battery.
Fresh Gas/Emergency Air
Intake
Handle Side
Controls and Indicators
The ventilator controls and indicators (shown in Figure 2-2) facilitate ease of use and visibility
in all operating environments.
Side Enables the ventilator internal compressor to use ambient
air and acts as an anti-asphyxia valve.
Figure 2-2 Controls and Indicators
Controls
The ventilator’s controls consist of the following:
Control Function
Power Switch Enables the user to turn the ventilator ON and OFF.
Parameter Buttons Enables the user to access primary parameters, secondary parameters
and context menus associated with a primary parameter (if applicable),
and then modify settings using the Selection Dial).
Menu Button Enables the user to access the Menu.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-3
Page 30

Product Overview
Control Function
Selection Dial Enables the user to set values for a chosen (highlighted) Primary
Parameter, Secondary Parameter, Context Menu item, and Menu item.
Values accelerate with speed of turning.
Mute/Cancel button The Mute/Cancel button mutes the audible alarm allowing the user time
to change parameters. It can also be used to cancel parameter entries.
Accept/Select button The Accept/Select button allows the user to accept parameter value
settings, acknowledge popup messages, and accept a menu choices
Manual Breath Button/
Plateau Pressure
Indicators
The ventilator’s indicators consist of the following:
Indicator Description
LCD Display Displays settings, patient data, and alarm information.
LED Array Indicates operational status (Red, Yellow, or Green).
Display Screen
The ventilator’s display screen has four functional areas as shown in Figure 2-3:
• Alarm Message Center/Waveform Window
• Parameter Windows
• Shared Icon Area
• Auxiliary Parameter Boxes.
These functional areas are discussed in the following sections.
Enables the user to deliver a manual breath and measure Plateau
Pressure
Note: Plateau Pressure is an optional ventilator control.
Figure 2-3 Display Screen Functional Areas
2-4 www.zoll.com 9650-002360-01 Rev. A
Page 31

Product Overview
Message Area
The display screen’s message area can display the following:
• Airway Pressure and Pleth Waveform Plots -- Under normal operation (as in the example
above), the message area displays plots for airway pressure and, when the pulse oximeter is
connected, the Pleth waveform. When a plot is necessary to facilitate a parameter
adjustment, the message area displays both the plot and the parameter’s context menu.
• Menus -- Displays the Menu after you press the Menu button on the ventilator’s control
panel, or displays a parameter’s context menu (which appears after you press and hold the
associated parameter button on the control panel).
• Alarms -- When an alarms occur, the message area displays Smart Help™ messages that
identify the alarms and describe possible causes and actions that you can take in response.
• Popup Windows -- Display information that assists you when adjusting parameter values.
Parameter Windows
Each parameter window displays its primary parameter and associated secondary parameters,
that can include, associated parameters and alarm limits.
Two types of values appear in a parameter window.
• Solid text is used for primary and secondary parameter values you can adjust.
• Outlined text is used for patient-dependant measured values.
Chapter 4, “Using the Z Vent Ventilator” contains more information and instructions for
adjusting parameter values.
Shared Icon Area
Directly below the message area, the device displays icons that indicate
• The ventilator’s power source (operating on external power or its battery)
• The battery charging status
• An oxygen supply is attached
• Alarms are muted or audible
Auxiliary Parameter Boxes
Some parameters have values that the ventilator displays in the parameter boxes at the bottom
of the display screen. You can adjust these values using the parameter’s context menu.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-5
Page 32

Product Overview
Fresh Gas/Emergency Air Intake and Attachments
The Fresh Gas/Emergency Air Intake is located on the side of the ventilator as shown in
Figure 2-4.
Figure 2-4 Fresh Gas/Emergency Air Intake
The Fresh Gas/Emergency Air Intake allows ambient air into the device’s internal compressor.
The intake also acts as an anti-asphyxia valve that enables the patient to breathe ambient air
should the ventilator fail. The Fresh Gas/Emergency Air Intake contains a particulate filter and
permits the user to connect either a bacteria/viral or a chemical/biological filter depending on
ambient conditions.
ZOLL offers an Oxygen Reservoir Bag Assembly Kit to allow for low flow oxygen use with
the ventilator to provide supplemental oxygen to patients. Low flow oxygen sources can be
from a flow meter or an oxygen concentrator. Oxygen is delivered through the
Fresh Gas/Emergency Air Intake when the device's internal compressor cycles to deliver a
breath.
Oxygen Reservoir Bag Assembly
The Oxygen Reservoir Bag Assembly serves the following purposes:
• Acts as a reservoir, collecting oxygen during the expiratory phase of ventilation.
• Provides an interface to the ventilator and the attachment of the low-flow oxygen supply
hose.
• Provides an inlet in the event the low-flow oxygen supply fails or the tidal volume is greater
than the supplied oxygen.
See Chapter 3 for more information about using low flow oxygen sources.
2-6 www.zoll.com 9650-002360-01 Rev. A
Page 33

Figure 2-5 Z Vent with O2 Reservoir Kit
Product Overview
Top Panel
The oxygen hose, patient circuit, external power, and pulse oximeter attach to the top panel of
the ventilator. The USB port is only used when servicing the device.The ventilator top panel
appears as shown in Figure 2-6.
Figure 2-6 Top Panel
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-7
Page 34

Product Overview
Pulse Oximeter Compatibility
The ventilator can accommodate an optional connection of external Masimo Pulse Oximeter.
When the appropriate sensor is connected, the pulse oximeter provides continuous noninvasive
monitoring of the oxyhemoglobin saturation (SpO
sensor) for adult, pediatric and infant patients.
The Masimo LNCS series of probes are approved for use with the ventilator. The Accessory
table in Appendix A lists the sensors which are available for use with the ventilator.
Power Sources
The ventilator can operate using external power or it can operate powered by its internal Li-ion
battery.
The external AC/DC Power cable is a universal supply that can operate with an input of
100 to 240 VAC 50/60 Hz. The external supply can also power the device when provided with
a 400 Hz input.
The external AC/DC Power cable that ZOLL provides with the ventilator delivers a DC input to
the device of 24 V at 4.2 A. When this external power source is present, the ventilator
automatically charges its internal battery while operating.
) and pulse rate (measured by the SpO2
2
Only use the external power supply provided with the ventilator when connecting to AC power.
This power supply provides both Class I and Class II protection.
Operating Using External DC Power
The ventilator can also operate using external DC power. When connected to a standard vehicle
DC outlet using either the 12 or 28 VDC Power Cable that ZOLL offers, the ventilator
automatically charges its internal battery while operating.
Note: The input connector of the ventilator accepts DC voltages between 11.8 to 30.0 VDC.
Caution When using the standard vehicle DC outlet, do not jump start the vehicle during operation of the
ventilator.
Operating Using Battery Power
When an external power failure occurs, the ventilator automatically switches to its internal
battery for operating power and activates the EXTERNAL POWER FAILURE alarm; there is
no interruption in operation. When external power returns, operating power automatically
switches to the external power source and the following symbol displays on the ventilator
screen as shown in Figure 2-7.
2-8 www.zoll.com 9650-002360-01 Rev. A
Page 35

Product Overview
In the event that the ventilator needs to be shutdown, turn the POWER switch to the OFF (“O”)
position. If this fails to work or puts the patient or user at possible risk, disconnect the device
from the external power source.
Pneumatic Design
The ventilator includes an oxygen valve and a compressor to provide the appropriate gas
mixture for the patient. The system includes transducers for pressure measurements including
input supply and barometric pressure.
O
2
The Wye circuit is part of the ventilator’s pneumatic system. The inspiratory side of the wye
circuit provides gas to the patient. The expiratory side exhausts directly to atmosphere without
returning to the ventilator. The ventilator pneumatically controls the exhalation valve and a
transducer within the ventilator measures the airway pressure.
Figure 2-8 depicts a diagram of the ventilator’s pneumatic design.
Figure 2-7 External Power GUI Symbol
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-9
Page 36

Product Overview
Figure 2-8 Pneumatic Design
Oxygen Input: High Pressure Gas Supply
An external high pressure gas source connects to the ventilator using the high pressure oxygen
input port. The device attaches to a regulated medical grade (USP) O
supply of 40 to 87 psig (280 to 600 kPa). Maximum flow rate of the oxygen supply is 100 liters
per minute. The Oxygen Input fitting (See Figure 2-9) has a male oxygen Diameter Index
Safety System (DISS) thread.
Note: If external oxygen is connected, the oxygen pressure must be at least 41 psig (± 2 psig)
at the time the ventilator performs its Self-Check after turning on the ventilator.
High Pressure Oxygen Supply Hose
A standard 6 foot oxygen hose is available for connecting the ventilator to a high pressure
oxygen source. (Also see Chapter 6 “Operating Environments”). Hoses are available from
ZOLL, or a suitable alternative as described below can be used as indicated.
High Pressure Oxygen Hose for compliance with ISO standard (ISO STANDARD 5359)
Ventilator Side
Connections
DISS 6 ft (maximum 20 ft) Green or White (as
Hose Attributes Supply Side Connections
determined by local regulations)
non-conductive
system or O2 cylinder
2
Quick Disconnect, DISS, etc.
2-10 www.zoll.com 9650-002360-01 Rev. A
Page 37

Product Overview
Figure 2-9 Ventilator Gas Sources
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-11
Page 38

Product Overview
Patient Circuits
The ventilator can use 6 ft or 12 ft patient circuits (see Figure 2-10) to support adult, pediatric,
and infant patients.
Note: Troubleshooting information regarding patient circuits is found in Appendix D.
Figure 2-10 Patient Circuits
ZOLL provides the following circuit types:
• Pediatric/Adult, 6 ft and 12 ft
• Infant/Pediatric, 6 ft and 12 ft
Caution Always dispose of the circuit after single patient use following the institutional guidelines for
biologically contaminated material. Reusing the circuit can result in cross contamination
between patients.
2-12 www.zoll.com 9650-002360-01 Rev. A
Page 39

Product Overview
Intended Use
The pediatric/adult patient circuits are intended for use when delivering tidal volume from
200 ml to Adult.
The infant/pediatric patient circuit is intended for use when delivering tidal volume from
50 ml to 300 ml.
Warning! Patient circuits are non-sterile and intended for Single Patient Use Only
Caution During use the circuit may come into contact with biohazard material. Handle carefully to avoid
cross-contamination.
Not intended for use with heated humidifier.
Note: ZOLL Medical Corporation recommends that you examine the patient circuit on a
daily basis for damage or wear, such as cracking, discoloration, or disfigurement. If
there is any sign of physical degradation or if the ventilator has patient circuit alarm
conditions replace the patient circuit.
Use of Heat and Moisture Exchangers
Heat and Moisture Exchangers (HMEs) can be used with the device. The HME provides heat
and moisture to the inspired gas by recycling the heat and moisture contained in the patient's
exhaled gas. While HMEs may not be suitable for all applications, they facilitate portability in
a way that conventional humidifiers cannot. The device can be used with an optional HME or
an optional HME/bacterial viral filter (HMEF). Be sure to follow all instructions provided by
the manufacturer.
Note: Use of the HME will cause a slight increase in the inspiratory and expiratory
resistance. Always monitor the patient and adjust the ventilator as needed.
ZOLL does not offer a heated humidification option for the device.
Warning! Users should use the appropriate HME for the patient's size. Failure to do so can result
in excessive dead space and lead to hypercapnia and hypoxia.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-13
Page 40

Product Overview
Attaching a Patient Circuit to the Ventilator
Figure 2-11 shows how to attach a patient circuit to the ventilator.
Figure 2-11 Patient Circuit Attachment
The list that follows identifies the circuit connections.
Connection Symbol on the Ventilator Description
Inspiratory Line Gas Output
Pressure Line (Green) Transducer
2-14 www.zoll.com 9650-002360-01 Rev. A
Page 41

Connection Symbol on the Ventilator Description
Expiratory Line (Clear) Exhalation Valve
Oxygen In High Pressure Oxygen
Exhaust Do Not Occlude
Specifications
Product Overview
Outlet
Pediatric/Adult, 6 ft Patient Circuit
The Pediatric/Adult, 6 ft patient circuit has the following specifications:
• Internal Diameter: 22 mm
• Inspiratory Resistance: R
• Expiratory Resistance: R
• Tubing Compliance: C
• Dead Space: 22 ml
• Maximum Working Pressure: 100 cm H
@ 30 Lpm: 0.01 hPa/l/min
INSP
@ 30 Lpm: 0.10 hPa/l/min
EXP
@ 60 hPa: 1.6 ml/cm H20 ml/(hPa)
T
0 (hPa)
2
Pediatric/Adult, 12 ft Patient Circuit
The Pediatric/Adult, 12 ft patient circuit has the following specifications:
• Internal Diameter: 22 mm
• Inspiratory Resistance: R
• Expiratory Resistance: R
• Tubing Compliance: C
• Dead Space: 22 ml
• Maximum Working Pressure: 100 hPa (cm H
@ 30 Lpm: 0.02 hPa/l/min
INSP
@ 30 Lpm: 0.10 hPa/l/min
EXP
@ 60 hPa: 2.8 ml/hPa
T
O)
2
Infant/Pediatric, 6 ft Patient Circuit
The Infant/Pediatric, 6 ft patient circuit has the following specifications:
• Internal Diameter: 10 mm
• Inspiratory Resistance: R
• Expiratory Resistance: R
• Tubing Compliance: C
• Dead Space: 4.2 ml
• Maximum Working Pressure: 100 hPa (cm H
@ 15 Lpm: 0.11 hPa/l/min
INSP
@ 15 Lpm: 0.17 hPa/l/min
EXP
@ 60 hPa: 0.5 ml/hPa
T
O)
2
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 2-15
Page 42

Product Overview
Infant/Pediatric, 12 ft Patient Circuit
The Infant/Pediatric, 12 ft patient circuit has the following specifications:
• Internal Diameter: 10 mm
• Inspiratory Resistance: R
• Expiratory Resistance: R
• Tubing Compliance: C
• Dead Space: 4.2 ml
• Maximum Working Pressure: 100 hPa (cm H
@ 15 Lpm: 0.17 hPa/l/min*
INSP
@ 15 Lpm: 0.17 hPa/l/min
EXP
@ 60 hPa: 0.8 ml/hPa
T
O)
2
Note: The extended length of the tubing in the 12 ft circuit results in a higher RINSP
compared to the 6 ft circuit.
Warning! Compressible volume can significantly decrease the delivered tidal volume. When
managing patients at risk, always correct for compressible volume Use the Vt Context
Menu to adjust Tubing Compliance and Compressible Volume Measurements.
Warning! Do not use the 12 ft circuit with PEEP settings below 5 cm H2O.
Warning! Given the additional length of the 12ft circuit, the system may not be able to trap PEEP
in patients with short expiatory time. Always ensure that the device is performing as
required.
2-16 www.zoll.com 9650-002360-01 Rev. A
Page 43

Chapter 3
Setting Up the Z Vent
Ventilator
This chapter describes how to set up the Z Vent ventilator. It lists the tasks required to set up the
ventilator for safe, effective use, and describes each task in detail.
Warning! You must properly set up the ventilator before use. Failure to do so can result in
inadequate care or death of the patient.
To set up the ventilator, you must perform the following tasks:
1. Attach the patient circuit
2. Attach the high pressure oxygen supply (optional)
3. Inspect Fresh Gas/Emergency Air Intake filters
4. Connect Fresh Gas/Emergency Air Intake attachments (optional)
5. Select the ventilator’s power source
6. Power on the ventilator
7. Select Start Up default configurations
8. Change the operating mode (optional)
9. Change parameter values
10. Perform an operational test
11. Attach the pulse oximeter (optional)
12. Attach patient
We describe how to perform these tasks in the following sections of this chapter.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 3-1
Page 44

Setting Up the Z Vent Ventilator
Warning! Always follow physicians orders and local protocols that includes preparations to
manually ventilate (bag) the patient. Ensure there is a functioning Bag Valve Mask
available to support the patient in the event of a ventilator failure. DO NOT start up the
ventilator with the patient attached.
1. Attach the Patient Circuit
Select the correct patient circuit for the patient and environment (as we describe in the previous
chapter). Always follow the instructions included with the circuit. Attach the patient circuit to
the ventilator’s top panel as follows. See Figure 3-1.
• The 22 mm corrugated hose to the ventilator’s gas output
• The green 3/16 inch ID airway pressure line to the pressure transducer
• The clear 1/4 inch ID exhalation valve control line to the exhalation valve fitting.
• The oxygen hose to the Oxygen Input connector.
Note: The circuit recommended temperature range for use is -40 °C to 70 °C
(-40 °F to 158 °F).
Figure 3-1 Patient Circuit Device Connections
Warning! Adult patients should only be ventilated with Pediatric/Adult circuits. Infant patients
should only be ventilated with Infant/Pediatric circuits.
Warning! ZOLL recommends the use of the patient circuits that ZOLL offers for the ventilator. If
circuits with different resistance/compliance are used or additional accessories are
placed in line with the circuit, you must use the appropriate compliance factors for the
new circuit, and make sure the dead space volume of the added accessories are
considered so that the device delivers an effective tidal volume to the patient.
3-2 www.zoll.com 9650-002360-01 Rev. A
Page 45

Setting Up the Z Vent Ventilator
Warning! Dead space increases with mask ventilation; always follow the mask manufacturer's
directions.
2. Attach the High Pressure Oxygen Supply (Optional)
Since the ventilator includes an internal compressor, the attachment of a high pressure oxygen
supply is optional. Review the high pressure supply requirements that we describe in Chapter 2,
and use the oxygen hose to attach the ventilator’s oxygen inlet to the high pressure O
The ventilator oxygen inlet is shown in Figure 3-2.
Warning! Use only with medical-grade (USP) oxygen. When using with an oxygen cylinder, the
cylinder must be secured. The O
country specifications.
hose is either colored green or white, depending on
2
source.
2
Figure 3-2 Oxygen Inlet
3. Inspect Fresh Gas/Emergency Air Intake Filters
The Fresh Gas/Emergency Air Intake provides the gas path for the ventilator’s internal
compressor. Two built-in filters protect the compressor and patient from particulate matter (a
removable foam filter and a Fresh Gas/Emergency Air Intake disk filter).
The ventilator’s Fresh Gas/Emergency Air Intake is shown in Figure 3-3. Inspect the filters and,
if dirty, replace them (See the section, “Replacing Ventilator Filters” in Chapter 7.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 3-3
Page 46

Setting Up the Z Vent Ventilator
Figure 3-3 Fresh Gas/Emergency Air Intake
Warning! Never block the Fresh Gas/ Emergency Air Intake, free flow of air is required during
compressor operation or in the event of device failure to allow spontaneous breathing.
The Fresh Gas/Emergency Air Intake also acts as an anti-asphyxia port in the event of a
ventilator failure.
4. Connect Fresh Gas/Emergency Air Intake Attachments (Optional)
The operating environment of the ventilator may require you to connect the following
attachments to the Fresh Gas/Emergency Air Intake:
3-Liter Reservoir Bag Assembly
If the ventilator will use oxygen from low-flow sources, you may choose to attach an Oxygen
Reservoir Bag Assembly. Follow these steps:
1. Press the Menu button and use the Dial to choose O
ventilator that the reservoir is attached and prevents the FRESH GAS INTAKE
RESTRICTED alarm from sounding.
2. Attach the Oxygen Reservoir Bag Assembly to the Fresh Gas/Emergency Air Intake. This
port is located in the side of the ventilator. It will be necessary to use the supplied 22 mm
male-to-male adapter with 731 Series Ventilators.
3. Connect the O
4. Adjust the O
Note: The assembly will function when the reservoir bag is hanging down or lying
horizontally provided the bag does not fall in such a way that occludes the neck of the
bag. The ventilator will sound a Low Priority FRESH GAS INTAKE RESTRICTED
Alarm if the menu has not been changed (see "1"above). Operating with the alarm
active does not affect the ability of the ventilator to deliver breaths at the current
settings. It is to alert the user that a restriction has been detected at the inlet.
supply tubing between the O2 source and the hose barb on the Reservoir Kit.
2
flow to achieve an acceptable O2 saturation.
2
Reservoir "On". This tells the
2
Always allow 5 to 10 minutes between adjustments to assure the patient oxygenation
has stabilized. This is very important when decreasing the O
several minutes for a patient to stabilize at the new O
3-4 www.zoll.com 9650-002360-01 Rev. A
2
supply where it may take
2
flow.
Page 47

Setting Up the Z Vent Ventilator
Never use O2 flows > 10-12 liters/min. Flows greater than this can cause the baseline
pressure to drift, waste oxygen, and may cause an INCOMPLETE EXHALATION
alarm.
Warning! Always monitor the patient's oxygenation using a pulse oximeter. The O2 flow from a
concentrator or other O
target. Failure to follow the Instructions and WARNINGS provided with the O
source may not be adequate to achieve the desired SPO2
2
2
Reservoir could result in an adverse effect on the patient.
Note: Due to the slight difference between the densities of air and O2, the tidal volume will
decrease slightly as O
volume when the entrained O
is entrained. The worst case is a < 10% decrease in tidal
2
results in FIO2 of 100%.
2
The table below shows both the affect on tidal volume and the resultant FIO
rate.
Z Vent AC 12, Vt 700, PEEP 5, I:E 1:2.5
Flow 0 1 2 3 4 5 6 7 8
O
2
FIO
2
Vt (set) 740 732 725 718 711 703 691 689 682
Vt (actual) 700 692 685 678 671 663 651 649 642
% Chg 0 -1.1 -2.1 -3.1 -4.1 -5.3 -7.0 -7.3 -8.3
O2 Flow 0 1 2 3 4 5 6
FIO
2
Vt (set) 527 523 514 506 502 493 486
Vt (actual) 500 496 487 479 475 466 459
% Chg 0 -0.8 -2.6 -4.2 -5.0 -6.8 -8.2
Flow 0 1 2 3 4 5 6
O
2
FIO
2
Vt (set) 312 307 303 299 298 291 287
Vt (actual) 300 295 291 287 286 279 275
% Chg 0 -1.7 -3.0 -4.3 -4.7 -7.0 -8.3
21 30 38 48 57 70 80 89 100
AC 12, Vt 500, PEEP 5, I:E 1:2.5
21 30 43 56 69 89 100
AC 18, Vt300, PEEP 5, I:E 1:2.5
21 32 47 62 76 96 100
supply
2
Bacterial/Viral (BV) Filter
If the ventilator will operate in an environment where the patient is at risk from cross
contamination or airborne pathogens, you may choose to attach a BV filter (See Chapter 6,
“Operating Environments” for more information on this filter).
Chemical/Biological C2A1 Filter
If the ventilator will operate in a contaminated environment, you may choose to attach a
chemical/biological C2A1 filter obtained from a Chemical/Biological Filter supplier.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 3-5
Page 48

Setting Up the Z Vent Ventilator
Note: ZOLL does not offer this filter. (See Chapter 6, “Operating Environments” for more
information on this filter).
Warning! Always monitor the patient and ventilator when using external filters or the external O2
reservoir. Changing modes can trigger false compressor failure alarms when the
device’s parameter configurations requires very high air flow.
5. Select the Ventilator’s Power Source
The ventilator can run using one of the following power sources:
1. Internal 14.4 VDC Li-ion rechargeable battery with 6.75 Ah capacity (fully charged, the
battery provides 10 hours of operation at factory default settings with pulse oximeter
operating at 25 °C (77 °F).
2. External AC/DC Power Supply that ZOLL provides (100 to 240 VAC 50/60 and 400 Hz with
an IEC 320 style AC input connector. The AC/DC Power Supply provides a DC output of
24 V at 4.2 A.
3. External DC power from a standard vehicle DC outlet using either the 12 or 28 VDC Power
Cable that ZOLL provides to connect the ventilator to the DC outlet. The ventilator’s input
connector accepts DC voltages between 11.8 to 30.0 VDC.
The ventilator uses external power when available rather than its internal battery pack. When an
acceptable external power source is present, the ventilator automatically charges the internal
battery while the device operates. When an external power failure occurs, the device
automatically switches to its internal battery for operating power and activates the EXTERNAL
POWER FAILURE alarm; there is no interruption in operation or loss of any alarms. When
external power returns, operating power automatically switches from internal power to the
external source.
In the event that the device needs to be shutdown, turn the Power switch to the OFF (“O”)
position. If this fails to work or puts the patient or user at possible risk, disconnect the device
from the mains power.
To connect the ventilator to an external power source, connect an AC/DC Power Supply plug to
the device’s External Power Input and an acceptable electrical outlet.
3-6 www.zoll.com 9650-002360-01 Rev. A
Page 49

Connecting the Power Supply
Connect the external power cable to the ventilator as described in Figure 3-4 and Figure 3-5.
Setting Up the Z Vent Ventilator
Figure 3-4 Connecting and Disconnecting the Power Supply
Warning! If the power supply, power cable, or power connection plugs are damaged or become
damaged during use, immediately disconnect the power cable from external power and
the device.
Caution Do not twist the power cable connection plug. Pinch the plug and slide up to release the safety
latches. Failure to do so may damage the power connection plug and prevent it from functioning.
Figure 3-5 Power Supply Latching
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 3-7
Page 50

Setting Up the Z Vent Ventilator
6. Power On the Ventilator
To power on the ventilator, turn the Power switch to “I”. Figure 3-6 shows the location of the
ventilator’s Power switch.
Figure 3-6 Power Switch
After powering on, the device performs its Self-Check test, which checks for alarm conditions
and the operation of the pneumatic system, internal communications, and power system. After
completing the Self-Check test, the ventilator waits for the user to select a starting
configuration before it begins to operate. Once operation begins, the ventilator continuously
monitors for alarm conditions.
During normal start-up, the ventilator’s alarms are muted for 2 minutes (120 seconds) to allow
you to connect the patient circuit, pulse oximeter, adjust ventilator settings, and perform an
operational test without distraction. The start-up mute self clears when there are not any active
medium priority alarms and no un-muted low priority alarms for a period of 15 seconds.
Warning! Always start the ventilator, select the patient settings, ensure operation, and then
connect the patient. Always manually ventilate the patient when they are not connected
to the ventilator.
7. Select Start Menu Option
When you power on the ventilator, the Start Menu appears, from which you choose an
appropriate starting configuration for the patient. You can select from these patient defaults:
• Adult
• Pediatric
• Mask CPAP -- Continuous Positive Airway Pressure (CPAP)
• Custom -- Values saved in a previous session
• Last Settings -- Values set for the last patient treated before turning OFF the ventilator
Note: Gas flow at start up is used to detect the patient in event proper procedures are not
followed.
3-8 www.zoll.com 9650-002360-01 Rev. A
Page 51

Setting Up the Z Vent Ventilator
Warning! Default settings are intended to speed the configuration of the ventilator. Particular
care should be taken to adjust the ventilator appropriately before ventilating infants
and children. The ventilator should always be adjusted before placing the patient on the
ventilator.
The predefined configurations (Adult, Pediatric, Mask CPAP) are default settings defined
within the specified use. The Custom default can be used to define a configuration that supports
your use and/or patient population. See Chapter 4 for more information.
To select the device’s default parameter values, highlight one of the above settings in the Start
Menu and press the Accept button. To operate with parameter values that differ from the
default values, use the device’s parameter buttons (see the “Changing Parameter Values”
section later in this chapter).
Note: You can configure the ventilator to automatically select Adult parameter defaults at
start up from the start config submenu from the menu.
Warning! Never use the CPAP and BL mode on a patient that is NOT spontaneously breathing
and/or may stop spontaneous breathing. CPAP and BL are intended for ventilatory
support, NOT ventilation.
When noninvasive CPAP and BL with LC is used, the head with mask icon appears in the
location used by the speaker/mute icons. Low and Medium priority alarms cause this head with
mask icon to disappear. It reappears when low priority alarms are muted.
When Medium priority alarms are muted, the muted speaker icon appears.
8. Change Operating Mode (Optional)
The ventilator offers four operating modes that you can use to manage the patient (active
modes, AC and SIMV can provide either pressure or targeted ventilation):
1. AC (Assist/Control) -- The patient receives either controlled or assisted breaths. When the
patient triggers an assisted breath, the patient receives a breath based on either the volume or
pressure target.
2. SIMV (Synchronized Intermittent Mandatory Ventilation)-- The patient receives controlled
breaths based on the set breathing rate. Spontaneous breaths are either unsupported demand
flow or supported using Pressure Support.
3. CPAP (Continuous Positive Airway Pressure) -- The patient receives constant positive
airway pressure while breathing spontaneously. Spontaneous breaths are either demand flow
or supported using Pressure Support.
4. BL (Bilevel) -- the ventilator provides two pressure settings to assist patients breathing
spontaneously: a higher inspired pressure (IPAP) and a lower expiratory pressure (EPAP).
Note: SIMV mode is an optional mode on the Z Vent ventilator that might not be available on
your Z Vent ventilator.
To select the operating mode, press the Mode parameter button, turn the Dial to highlight the
mode you want to use, and press the Accept button.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 3-9
Page 52

Setting Up the Z Vent Ventilator
When transitioning from active ventilation to CPAP/BL modes, or from CPAP/BL with LC
mode to active ventilation, the following parameter/alarm limit may be adjusted:
• Low BPM Alarm
• High BPM Alarm
• Low Airway Pressure Alarm
• PEEP
• VT High Limit
• VT Low Limit
• Rise Time
• Pressure Support
Warning! The transition into CPAP/BL automatically sets the rise time to 3, which may be too fast
for some patients as well as infants and small children. Before using the ventilator with
an infant or small child, you should always configure the ventilator appropriately
before attaching the patient and monitor the patient to ensure optimal support.
Note: The Patient Detect alarm triggers when you connect the patient to the ventilator while
the Start Menu is still active. To resolve the alarm, you must select a mode of
ventilation and configure the device appropriately for the patient. In addition, you
should perform the Operational Test procedure before reconnecting the patient to the
device.
9. Change Parameter Values
If the patient requires parameter values that differ from the default values, you can use the
parameter buttons to change these values. To change the parameter values, press the parameter
buttons to highlight the primary parameter and secondary parameter values, or press and hold
the parameter button to display the parameter’s context menu. Use the Dial to adjust the value
of the highlighted parameter. Press the Accept button to implement the change.
Warning! The alarm limits must be appropriate for the patient being ventilated. If a parameter is
changed, adjust the high and low alarm limit to bracket the new value.
3-10 www.zoll.com 9650-002360-01 Rev. A
Page 53

10. Perform Operational Test
Before attaching the patient to the ventilator, you should perform an Operational Test to ensure
that the breathing circuit is properly attached and that the primary patient safety alarms, such as
PATIENT DISCONNECT and AIRWAY PRESSURE HIGH are functioning properly.
To perform the operational test procedure, do the following:
a. Press the Manual Breath button; gas should flow out of the patient connection each time
the button is pressed.
b. Close the patient port with a clean gloved hand. The HIGH AIRWAY PRESSURE LIMIT
alarm should activate after 2 breaths that reach the PIP High Limit.
If the AIRWAY PRESSURE HIGH alarm fails to activate, check to determine that all of the
circuit connections are secure, the exhalation valve is closing during inhalation, and that the
High Airway Pressure Limit is set to 35 cm H2O or less.
c. After a breath or two, release the patient port while allowing the ventilator to operate. The
PATIENT DISCONNECT alarm should activate.
d. Partially close the patient port to reset the PATIENT DISCONNECT alarm.
e. With no other alarms occurring, remove external power from the ventilator. The
EXTERNAL POWER
LOW/DISCONNECT alarms should activate. Reconnect external power to reset alarms.
If either the HIGH AIRWAY PRESSURE, PATIENT DISCONNECT, or EXTERNAL
POWER LOW/DISCONNECT alarms fail to activate, continue to manually ventilate the
patient, check the patient circuit for leaks or a faulty exhalation valve and repeat the
Operational Test.
Setting Up the Z Vent Ventilator
If operating using the internal battery, verify that the Battery icon indicates sufficient available
battery capacity remains to support the anticipated duration of operation. If not, begin
ventilation and find an alternate source of power.
Warning! Until you have determined that the ventilator is functioning properly and that the
ventilator parameters are set correctly for the patient, do not connect the patient to the
ventilator.
11. Attach the Pulse Oximeter (Optional)
The pulse oximeter operates in all ventilator modes when its cable and sensor are properly
attached to the SpO
and HR parameter windows display stby).
To operate the pulse oximeter, connect the sensor to the patient and the cable to the SpO
connector on the top of the ventilator as shown in the Figure 3-7.
connector (during start up, the pulse oximeter is on standby -- the SpO2
2
2
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 3-11
Page 54

Setting Up the Z Vent Ventilator
Monitoring begins automatically when a valid patient signal is detected for greater than
10 seconds. For more information about the Masimo pulse oximetry technology that the
ventilator uses, see Appendix C, Pulse Oximeter Principles.
Figure 3-7 Connecting the Pulse Oximeter Sensor
12. Attach Patient
After you confirm that the ventilator is operating correctly, detach the test lung (if used in the
Operational Test) from the patient circuit. Attach the patient airway (endotracheal tube,
supraglottic airway or tracheotomy tubes) or mask to the patient circuit connection port.
Note: If there are circuit-related alarms during set up or initial use, such as Disconnect, PEEP
Leak, Low Airway Pressure, or Auto PEEP, check all circuit connections and the
exhalation valve.
Warning! Never leave the patient unattended.
Warning! Always assure that there is an alternate means of providing mechanical ventilation.
A bag-valve resuscitator and an appropriate mask for the patient being ventilated
should be immediately available.
Warning! Do not connect the patient to the ventilator until you determine that the ventilator is
functioning properly and that the ventilator parameters are set correctly for the
patient.
Warning! Do not connect anything to the USB connection. The USB connection does not provide
any signal output or input to the user. The USB connection is a tooled access used
during service of the device.
3-12 www.zoll.com 9650-002360-01 Rev. A
Page 55

Chapter 4
Using the Z Vent Ventilator
This chapter describes how to use the ZOLL Z Vent ventilator. Effective operation of the
ventilator requires understanding of the following topics:
• Initial operation with default (predefined) parameter settings
• Changing parameter settings
• Saving custom configurations for future use
• Using the last settings enabled on the ventilator
• Mode Parameter Window Options
• BPM Parameter Window Options
• Vt Parameter Window Options
• PIP Parameter Window Options
• FIO
• SpO
• HR Parameter Window Options
• Popup Messages
• Using the Menu
Parameter Window Options
2
Parameter Window Options
2
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-1
Page 56

Using the Z Vent Ventilator
Initial Operation with Default Parameter Settings
Following power on, the ventilator goes through a Self-Check (a set of system tests and
checks). If the Self-Check passes, the LED array turns green and the Start Menu displays,
indicating that the ventilator is operational.
The Start Menu enables the user to choose between predefined ventilator parameter settings
(for adult patients, pediatric patients, patients requiring Mask CPAP), a previously saved set of
Custom parameter settings, or the parameter settings last used during ventilator operation. The
Start Menu choices include:
Choice Description
Adult Preset ventilation parameter settings for adult patients.
Pediatric Preset ventilation parameter settings for pediatric patients.
Mask CPAP Preset ventilation parameter settings for Mask CPAP (Continuous
Positive Airway Pressure) ventilation.
Custom Ventilation parameter settings previously saved by a user.
Last Settings The settings enabled on the ventilator during its last use (but not
saved as Custom Settings by the user).
Warning! Do not connect patient to the ventilator while the Start Menu is active.
Default Parameter Settings for Adult, Pediatric and Mask CPAP
The default settings for Adult, Pediatric, and Mask CPAP are as follows:
Adult Default Parameter Setting Values
Parameter Default Setting Value
Mode AC (V)
BPM 12
I:E 1:3
VT 450
PEEP 5
PIP Limit 35
FIO
2
21
4-2 www.zoll.com 9650-002360-01 Rev. A
Page 57

Pediatric Default Parameter Setting Values
Parameter Default Setting Values
Mode SIMV (P)* AC(P)
BPM 20 20
Ti 0.6 0.6
PIP 20 20
PEEP 4 4
PIP Limit 30 30
Using the Z Vent Ventilator
FIO
2
* Note: SIMV (V) and SIMV (P) are optional modes on the Z Vent.
21 21
Mask CPAP Default Parameter Setting Values
Parameter Default Setting Value
Mode CPAP
Backup BPM 12
Backup I:E 1:3
Backup PIP 20
PEEP 5
PIP Limit 30
FIO
2
21
Making a Choice From the Start Menu
Choose the option that is most is appropriate for the patient. With the Start Menu displayed,
follow these steps:
1. Turn the Dial to highlight your choice. For example, to choose the Mask CPAP default, turn
the Dial until Mask CPAP highlights.
2. Press the Accept button to enable your choice. The ventilator begins operation with the
choice you made. For initial set-up, see Chapter 3.
Note: The ventilator provides a 120-second alarm mute it automatically clears as described
above to allow the provider time to adjust parameter settings for the patient.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-3
Page 58

Using the Z Vent Ventilator
Changing Parameter Settings
The Z Vent ventilator helps you to manage the patient by organizing ventilator parameters in
groups that are accessed through parameter windows on the right side of the display screen.
A button corresponding to each window, enables you to select and set parameters.
Parameter Group Item Description
Primary Parameter The primary parameter setting accessed and controlled through the
parameter window. These are labeled with large font as: HR,
SP
O
, FIO2, PIP, Vt, BPM, and Mode.
2
Secondary Parameters and
Alarm Thresholds/Limits
Context Menu Additional settings that further adjust the performance of the device
Parameters controlled by the user appear as solid text (or a solid symbol) in either the parameter
window or auxiliary boxes. Parameters dependent on the patient appear as outlined text in the
parameter window. Figure 4-1 shows parameter windows.
Secondary parameters associated with the primary parameter and
alarm thresholds for alarms associated with the primary parameter,
(small font).
related to the primary parameter.
Figure 4-1 Parameter Windows and Auxiliary Boxes
Figure 4-2 shows the buttons associated with parameter windows.
4-4 www.zoll.com 9650-002360-01 Rev. A
Page 59

Using the Z Vent Ventilator
Figure 4-2 Parameter Buttons
Navigating the Parameter Windows Using Parameter Buttons
Parameter values are accessed with a parameter button as follows:
• Single Press: highlights the primary parameter for the chosen parameter window
• Multiple Presses: highlights secondary parameters and alarm limits. (Multiple presses
highlight secondary parameters moving in a clockwise direction)
• Press and Hold: opens the context menu (for those primary parameters that have a context
menu). Turning the Dial highlights context menu items.
Note: If you attempt to set parameter values that are outside the typical clinical range of
settings, the ventilator displays popup messages that ask if you are sure you would like
to set the parameter to that value. We describe popup messages in more detail later in
this chapter. To set the parameter above the limit, you must press the Accept button,
and then adjust the values and press the Accept button again.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-5
Page 60

Using the Z Vent Ventilator
Changing a Parameter Setting
To change a parameter, follow these steps.
1. Access the parameter you intend to change using the parameter buttons as described in the
previous sections.
2. Press the Accept button to highlight your choice. Multiple presses are required for secondary
parameters.
3. Turn the Dial to adjust the parameter value
4. Press the Accept button to enable your choice.
The examples that follow describe how to change a primary parameter, secondary parameter,
and a context menu parameter.
Example 1 — Changing a Primary Parameter
In Example 1, the ventilator is operating with the default Adult parameters. The user wants to
change the Mode from AC (Adult Default) to CPAP.
To change the Mode parameter from AC to CPAP, follow these steps:
1. Press the Mode parameter button once.
2. Turn the Dial until the Mode parameter window displays CPAP.
3. Press the Accept button.
Example 2 — Changing a Secondary Parameter
In Example 2, the ventilator is operating with the default Adult parameters and user wants to
adjust the PEEP secondary parameter setting from 5 (the adult default setting) to 7 cm H
O.
2
To change the secondary parameter PEEP setting from 5 to 7, follow these steps:
1. Press the PIP parameter button until PEEP is highlighted.
2. Turn the Dial until the PEEP setting is 7 as displayed in the PIP parameter window.
3. Press the Accept button.
Example 3— Changing a Context Menu Parameter
In Example 3, the ventilator is operating with the default Adult parameters and user wants to
change the Masimo Pulse Oximeter Sensitivity from Norm (normal) which is the adult default
setting – to Max (maximum).
To change the Sensitivity setting from Norm to Max, follow these steps:
1. Press and hold the HR or SpO
2. Release the parameter button, then turn the Dial until the Sensitivity choice highlights, then
parameter button until the Masimo context menu displays.
2
press the Accept button. The Sensitivity setting highlights.
3. Turn the Dial until the setting changes to Max (maximum).
4. Press the Accept button.
4-6 www.zoll.com 9650-002360-01 Rev. A
Page 61

Saving Changed Parameters for Future Use
When you change parameters, you can save them for future use through the Custom Settings
Menu option.
To save changed parameters for future use, follow these steps.
1. With the ventilator turned on, make the desired parameter changes using the parameter
buttons, Dial and Accept button.
2. Press Menu button.
3. Using the Dial, highlight Powerup Settings, and then press the Accept button.
4. Use Dial to highlight Custom Settings, then press the Accept button to highlight Save, and
then press the Accept button again to save the changed parameters.
To use or confirm that the changed parameters are saved as Custom settings, follow these steps:
1. Turn on the ventilator.
2. When the Start up Menu displays, turn the Dial to highlight Custom Settings, and then press
the Accept button.
3. The ventilator displays the custom parameter settings in the parameter windows.
Using the Z Vent Ventilator
Warning! The Custom and Last Setting options save the current configuration of the ventilator.
Always ensure that the both the ventilation settings and alarm limits are appropriate
for the patient.
Using the Last Settings Enabled on the Ventilator
The ventilator preserves the last setting used on the ventilator (even if they were not saved as
Custom Setting). To use the last settings, do the following:
1. Turn ON the ventilator, wait for the Self-Check to complete, and the Start Menu to display.
2. Turn the Dial to highlight the Last Settings choice.
3. Press the Accept button to enable your choice.
The ventilator begins operation with the last settings used.
Warning! Last Settings uses all of the parameters including alarms. You must ensure that both the
setting and alarms are appropriate for the patient.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-7
Page 62

Using the Z Vent Ventilator
Mode Parameter Window Options
The ventilator allows you to select different ventilation modes to optimally manage the patient.
Primary Parameter
The Mode parameter window has four primary parameter choices.
• AC (Assist/Control) -- The patient receives either controlled or assisted breaths. When the
patient triggers an assisted breath, they receive a breath based on either the volume or
pressure target.
• SIMV (Synchronized Intermittent Mandatory Ventilation) -- The patient receives
controlled breaths based on the set breathing rate. Spontaneous breaths are either
unsupported demand flow or supported using Pressure Support.
Note: SIMV is an optional mode on the Z Vent ventilator.
• CPAP (Continuous Positive Airway Pressure) -- The patient receives constant positive
airway pressure while breathing spontaneously. Spontaneous breaths are either demand flow
or supported using Pressure Support.
• BL (Bilevel) -- the ventilator provides two pressure settings to assist patients breathing
spontaneously: a higher inspiratory pressure (IPAP) and a lower expiratory pressure
(EPAP).
Secondary Parameters and Alarm Thresholds/Limits
The Mode parameter window has two secondary parameter choices.
• Breath target
• Leak Compensation
Breath Target
• Volume targeting (V) -- assures a constant volume is delivered to the patient in the
inspiratory time using a constant flow. During volume targeting, the PIP parameter appears
as outlined text.
• Pressure targeting (P) -- provides a constant airway pressure for the duration of the
inspiratory time using a decelerating flow pattern. During pressure targeting, the Vt
parameter appears as outlined text.
To set (or change) the Breath Target, follow these steps:
1. Press the Mode parameter button twice to highlight the Breath Target parameter
2. Use the Dial to switch between V (Volume Targeting) and P (Pressure Targeting)
3. Press the Accept. button.
Note: Please note that tidal volume cannot be adjusted in pressure-targeted ventilation. You
may have to adjust PIP to maintain desired tidal volume.
To adjust the PIP target in Pressure Targeted ventilation, follow these steps:
1. Press the PIP parameter button.
2. Use the Dial to set the desired PIP target value.
3. Press the Accept button.
In pressure target ventilation, setting the high and low tidal volume limits protect against leaks
and ensures delivery of a minimum tidal volume.
4-8 www.zoll.com 9650-002360-01 Rev. A
Page 63

Using the Z Vent Ventilator
Warning! During pressure-targeted ventilation, always set the high tidal volume just above the
patient's maximum tidal volume. In the event of disconnection or decannulation, the
increase in volume will trigger the alarm.
Leak Compensation
Leak Compensation (LC) is available in all pressure-targeted ventilation modes and provides
flow during the expiratory phase to maintain the baseline pressure caused by a leaking airway
or facemask. Leak compensation can accommodate leaks in the range of 0 to 30 liters per
minute.
To activate leak compensation, follow these steps:
1. Press the Mode parameter button twice. The “No Leak Compensation” icon highlights.
2. Turn the Dial to turn leak compensation ON. A pop-up message appears asking for
confirmation.
3. Press the Accept button to activate Leak Compensation.
To avoid nuisance alarms in patients with active leaks, Leak Compensation suppresses the
following alarms:
• Low Airway Pressure (# 2071)
• High Tidal Volume (# 2072)
• Low Tidal Volume (#2073)
Caution Once the patient is stable on CPAP or BL, the suppressed alarms should be set to ensure safe
ventilation of the patient. When in BL or CPAP modes, the Insufficient Flow alarm (Service
Code: 2095) triggers when the patient's inspiratory flow is greater than 100 lpm for two
concurrent breaths. The user may choose to decrease the magnitude of the trigger pressure (from
default setting of -2 cm H2O to -1 or -.5 cm H2O) and decrease the Rise Time to 1 so that the
flow reaches the maximum in the shortest amount of time.
Context Menu
The Apnea Back Up context menu is accessible when the primary parameter is set to CPAP or
BL modes.
Pressing and holding the Mode parameter button in CPAP or BL Modes displays the menu. The
menu displays:
• PIP Target
• BPM
• I:E
Apnea Back Up default settings are PIP target of 20, BPM 12, I:E (1:3), these settings should be
adjusted for the patient you are supporting in CPAP or BL modes.
When Apnea ventilation is enabled, the ventilator provides pressure targeted breaths at the
default Apnea settings or the provider can chose to change the settings. Apnea Backup is
triggered using the Low Breath rate alarm limit.
If the low breath limit is set to 4 and a breath is not detected every 15 seconds the ventilator will
begin ventilating at the Apnea back up settings and trigger an alarm. In CPAP or BL modes,
Apnea Backup is automatic.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-9
Page 64

Using the Z Vent Ventilator
To adjust or change any Apnea Backup settings,
1. From the menu, use the Dial to highlight the parameter to change, and then press the Accept
button (The current highlighted parameter value highlights).
2. Use the Dial to set the desired value
3. Press Accept to enable the change.
Mode Parameter Window Reference
The following table provides a reference to the primary parameters, secondary parameters, and
context menu parameters and other options for the Mode parameter window.
Mode Parameter Window Options/Range Availability/Notes
Primary Parameter Mode
Breath Target (V) or (P)
Secondary Parameters
LC
(Leak Compensation)
AC
SIMV Z Vent Optional Mode
CPAP
BL
ON or OFF
(Default OFF)
OFF BL Mode
Default ON CPAP Mode
AC (P) and SIMV(P)
modes
Alarms None
Measured Value None
Mode Context Menu Context menu is available
in CPAP and BL Modes
BPM 1 to 80
Apnea Backup
PIP 10 to 80
I:E, Ti 1:99, 0.1 to 3 Control selected in context
BPM context menu
Warning! Apnea Backup settings are appropriate for most adult patients. For small adult and
pediatric patients, adjust the Apnea Backup settings so that they are appropriate for the
patient.
4-10 www.zoll.com 9650-002360-01 Rev. A
Page 65

BPM Parameter Window Options
The BPM (Breathes Per Minute) parameter window defines the number of breaths per minute
delivered by the ventilator, as well as other parameters related to breath timing.
For AC and SIMV ventilation modes, the BPM parameter displays the setting as solid text. The
BPM parameter range is 0 to 80.
The BPM measured range is from 0 to 99.9
When CPAP and BL mode are used the primary parameter is the patient's breathing rate, shown
in the outlined text.
Assisted and controlled breaths are time-triggered and time-cycled. Spontaneous breathes are
patient triggered and flow cycled.
Secondary Parameters and Alarm Thresholds/Limits
The secondary parameters for the BPM parameter window are the following:
• High BPM alarm limit (number immediately below the Alarm Bell symbol)
• Low BPM alarm limit (lower number below the Alarm Bell symbol)
• Control Parameter (I:E ratio or Ti)
Note: Display of I:E or Ti in the BPM parameter window is determined by the Control
Parameter setting in the BPM context menu
The high breath rate alarm can be set off, or it can be set from 20 to 99.
Using the Z Vent Ventilator
The low breath rate alarm can be set from 2 to 40.
To change an alarm threshold setting, follow these steps:
1. Press the BPM parameter button until the desired alarm threshold highlights.
2. Turn the Dial to the desired threshold setting.
3. Press the Accept button.
To change the I:E or Ti setting, follow these steps:
1. Press the BPM parameter button until the I:E or Ti secondary parameter highlights.
2. Turn the Dial to change the setting.
3. Press the Accept button.
Context Menu
Pressing and holding the BPM parameter button brings up the BPM context menu. Different
parameters associated with the BPM can be adjusted in this context menu. The menu offers the
option of changing the following:
• Control Parameter (I:E or Ti)
• Rise Time (when pressure targeted breathes are available)
• Cycle Off %
• Spont Ti Limit
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-11
Page 66

Using the Z Vent Ventilator
Control Parameter (I:E or Ti)
The control parameter allows you to select between I:E ratio and Ti. The control parameter you
select is displayed in the parameter window and the parameter you are measuring is always
calculated and shown in the auxiliary boxes.
For AC and SIMV ventilation modes, control breaths are time-triggered and
time-cycled. The ventilator uses the inspiratory time (Ti) parameter setting to terminate the
breath being delivered.
For volume targeted breaths, the Ti parameter is used to determine the constant flow rate of the
delivered breath. Popup messages are triggered to alert the user if inappropriate settings are
selected.
The default inspiratory/expiratory control parameter is I:E ratio unless the pediatric default is
selected. The ventilator automatically calculates the inspiratory time, and displays it when the
I:E ratio is controlled, and it calculates the I:E ratio when the inspiratory time is used. Both are
always displayed on the screen.
As an advanced feature, I:E Ratio may be adjusted so that inspiration time is longer than
expiration time (referred to as inverse I:E with a range from 4.0:1 to 1.0:1, whereas normal I:E
is from 1:1.0 to 1:99). Going inverse requires a popup confirmation due to the potential for
harm when this setting is inappropriate for the patient’s condition.
Inverse I:E ratio is not available on all devices.
Rise Time
Rise Time allows the user to adjust the time it takes to reach the full inspiratory flow and Peak
Inspiratory Pressure (PIP) during pressure targeted breathing. The Rise Time displays in the
auxiliary boxes at the bottom of the display (See Figure 4-1). Use the PIP waveform as a
reference when adjusting the Rise Time for the patient. Rise time is available in these
ventilation modes:
• AC (P)
• SIMV (V or P)
• CPAP
• BL
When Pressure Support is enabled, you can adjust the time it takes to reach the PIP, where 1 is
the shortest and 10 is the longest. You should reassess and readjust the Rise Time settings after
the patient is placed on the ventilator and initially stabilized. To minimize patient's work of
breathing and potential for pressure overshoots, the following should be considered when
setting the Rise Time:
• Patient tidal volume
• Patient repiratory pattern
• Patient's comfort
• Patient's flow demand
• Patient's lung mechanics - resistance and compliance
Rise Time for a passive lung is driven primarily by airway resistance, and is fairly independent
of compliance.
Increasing Rise Time also decreases the maximum flow from the ventilator to allow for the
management of infant and pediatric patients.
4-12 www.zoll.com 9650-002360-01 Rev. A
Page 67

Using the Z Vent Ventilator
Note: An adult patient with high resistance may benefit from a Rise Time setting of 3 to 4 for
optimal breath delivery. Rise Times of 8 to 10 are optimized for infants and are flow
limited. (The infant circuit is not intended for flows > 60 LPM.)
Cycle Off % Parameter
For flow cycled breaths, the ventilator transitions from inspiratory to expiratory phase when the
flow drops below a set percentage of the peak flow. The default is 25% of peak flow with a
range of 10 to 70% Cycle Off % is typically used during noninvasive ventilation to ensure the
breath cycles in synchrony with the patient. Some patients may have difficulty cycling due to
their lung mechanics or a bag that prevents the flow from reaching the cycle flow rate. When
this happens, spontaneous breathe may time cycle causing asynchrony.
You can adjust the Cycle Off% value to account for patient leaks and weak respiratory effort.
Users must carefully assess the patient's response when adjusting the Cycle Off % to avoid
cycle asynchrony and patient discomfort
Note: The longest duration of a spontaneous breath is 5 seconds. At the end of this time, the
ventilator ends flow and opens the exhalation valve.
If there is no leak, increasing the Cycle Off % parameter causes breaths to cycle sooner, and
deliver less volume. If you set the Cycle Off % parameter too high, the breath ends early
relative to patient effort, which may lead to the triggering of a second breath.
Spont Ti Limit Parameter
The Spontaneous Inspiration Time (Spont Ti) Limit parameter provides an additional method to
limit the duration of spontaneous breaths in the presence of a leak or weak respiratory effort.
The parameter provides an inspiratory time limit for spontaneous breaths.
Adult default is 3.00 sec, Pediatric default is 2.00 sec, and Mask CPAP default is 3.00 sec.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-13
Page 68

Using the Z Vent Ventilator
BPM Parameter Window Reference
The following table provides the options and ranges for the BPM parameters:
BPM Parameter Window Options/Range Availability/Notes
Primary Parameter BPM
Breaths per minute
Secondary Parameters Ti (sec)
or
I:E
Alarm Limits High breath rate 20 to 99, OFF
Low breath rate 2 to 99
Measured Value Minute Volume (I) 0 to 99
BPM Context Menu
Control
Parameter
Rise Time - Pediatric Default 5
Cycle Off %
(% Cycle )
Spont Ti Limit Default
Default I:E I:E or Ti (sec) The control value is shown
Mask CPAP Default 3
Default 25% 10 to 70% Auxiliary Box
Adult = 3.00
Infant = 2.00
Mask CPAP = 3.00
1 to 80
Ti 0.3 to 3.0
or
I:E 1:1 to 1:99
Ti 0.1 to 5.0
or
I:E 4:1 to 1:99
1 to 10 Auxiliary Box
0.30 to 4.00
For the Basic Model, see
the Control Parameter in
the BPM context menu:
Inverse I:E is available as
an advanced feature of the
Z Vent
in the parameter window,
the dependent value is
shown in the Auxiliary Box.
4-14 www.zoll.com 9650-002360-01 Rev. A
Page 69

Using the Z Vent Ventilator
Vt Parameter Window Options
The Vt parameter defines the volume (ml) delivered to the lung with each breath. Tidal volume
is calculated by integrating the flow over the inspiratory time. During volume targeted
breathing, pressing the Vt parameter button highlights the currently set tidal volume and
enables it to be changed. When breathing is pressure targeted, the delivered tidal volume is
shown as outlined text and is based on the PIP and patient’s pulmonary mechanics.
Warning! In CPAP and BL, a Vt that is lower than anticipated may be an indication that the
patient is not able to adequately ventilate.
Note: In CPAP, the Vt delivered and V
the patient when leaks are present. The O
though the amount used is more than if no leak was present.
may be overestimates of the true volume going to
min
Use values accurately display the O2 use,
2
Warning! If significant leaks are present during CPAP and BL modes, the Vt delivered and V
shown may be overestimates of what is actually being delivered to the patient. The
adequacy of ventilation should be assessed using an alternate method.
Secondary Parameters and Alarm Thresholds/Limits
The secondary parameters for the Vt parameter window are the following:
• Vt High Limit (lower number below the alarm bell)
• Vt Low Limit (number below the Vt High Limit)
Note: The high/low Vt alarm limits are not presented during volume targeted ventilation
because the ventilator automatically alarms if 2 consecutive breathes are out of range.
The high Vt alarm can be set off, or it can be set from 50 to 2000 (ml). The low Vt alarm can be
set off, or it can be set from 5 to 500 (ml).
The Vt High Limit setting cannot be adjusted below the Vt setting or the Vt Low Limit setting
and attempting to do so generates a conflict popup message.
The Vt High Limit setting is used to limit the delivered tidal volume Pressure Targeted breaths.
If two consecutive breaths are limited by this setting, the High Tidal Volume alarm (2072) is
triggered.
min
The Vt Low Limit setting is used to trigger the Low Tidal Volume alarm (2073).
Warning! During pressure targeted breathing with infant and pediatric patients, always set the
high tidal volume limit just above the desired tidal volume. Doing this ensures an alarm
in the event of a leak or decanulation.
To change an alarm threshold setting, follow these steps:
1. Press the Vt parameter button more than once until desired alarm threshold highlights.
2. Turn the Dial until the desired value is reached.
3. Press the Accept button to enable the change.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-15
Page 70

Using the Z Vent Ventilator
Context Menu
Pressing and holding the Vt parameter button brings up the Vt context menu that includes:
• Compliance
• Adult
• Infant
• Compliance Volume
The tubing compliance can be set to off, adult, or infant.
Users can chose to use the adult or infant circuit defaults for the standard ZOLL adult/pediatric
and infant/pediatric patient circuits.
The patient circuit is part of the breathing system of the ventilator. Tubing compliance of the
circuit is a physical property that affects the tidal volume delivered to the patient. The ventilator
allows you to adjust the compliance value of the circuit (see Chapter 6 for more information).
Tubing compliance is adjustable and a different compliance value can be entered for each
circuit type by selecting either of the menu items. The range is limited for the adult patient
circuits from 0 to 3.5 ml/ cm H
a selection beyond the range of the standard circuits.
In order to prevent inappropriate or unsafe use of this feature, the tubing compliance
compensation is turned OFF at startup.
O and from 0 to 2 ml/ cm H2O for the infant circuits to prevent
2
Adult and Infant settings correspond to the standard WYE patient circuit used. Adult CT range
is between 0 and 3.50 ml/cm H2O (default of 1.60 ml/cm H2O), and the Infant CT range is
between 0 and 2.00 ml/cm H
O (default of.5 ml/cm H2O).
2
During volume targeted breathes the ventilator corrects for the loss due to CT by adding the
correction to the set Vt ensuring that the set volume is delivered to the patient. During pressure
targeted breaths the compensation volume is subtracted from the displayed Vt to indicate the
volume delivered to the patient.
To change a Vt context menu setting, follow these steps:
1. Press and hold the Vt parameter button until the Vt context menu displays.
2. Release the Vt parameter button, then turn the Dial until the context menu item you want to
change highlights, then press the Accept button. The current setting for the context menu
parameter highlights.
3. Turn the Dial to change the value of the parameter.
4. Press the Accept button.
4-16 www.zoll.com 9650-002360-01 Rev. A
Page 71

Vt Parameter Window Reference
The following table provides the options and ranges for the Vt window parameters:
Vt Parameter Window Options/Range Availability/Notes
Primary Parameter Vt
ml
Secondary Parameters None
High Vt 50 to 2000, Off Limits are not present during
Alarm Limits
Low Vt 5 to 500, Off
Vt Context Menu
Tubing Compliance (CT) Default: Off OFF, Adult,
Adult Default: 1.60 0 to 3.50 User entered values are not
Infant Default: 0.50 0 to 2.00
Compliance Volume (ml) (Measured value) 0 to 349
Using the Z Vent Ventilator
50 to 2000 Volume Target: Control Setting
Pressure Target: Measured
volume targeted ventilation
Auxiliary Box
Infant
retained when the ventilator is
turned OFF
PIP Parameter Window Options
The PIP (Peak Inspiratory Pressure) parameter window displays and/or controls airway
pressure. The breath target determines if the PIP value is a measurement or a control setting
(solid text). When PIP is a control setting, the user can adjust PIP from 10 to 80 cm H
Note: When the user attempts to set a PIP value greater than 60 cm H
displays prompting for a confirmation before values above 60 cm H
During pressure targeted ventilation, the PIP parameter is displayed as solid text.
During volume targeted ventilation, the PIP primary parameter is a measurement and displays
as outlined text.
Positive End Expiratory Pressure (PEEP) is the baseline pressure maintained above
atmospheric pressure at the end of exhalation to prevent alveolar collapse and improve gas
exchange.
For BL mode, the ventilator provides noninvasive ventilation with the ability to manage the
patient by adjusting the IPAP and EPAP parameters. IPAP is adjustable from 6 to 60 cm H
EPAP is adjustable from 3 to 30 cm H
Secondary Parameters and Alarm Thresholds/Limits
In pressure targeted ventilation mode, the following secondary parameters are available.
O.
2
0.
2
O, a popup message
2
0 are available.
2
2
O,
• High PIP Alarm Limit
• Low PIP Alarm Limit
• PEEP
• PS (Available for SIMV(P), CPAP, and BL)
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-17
Page 72

Using the Z Vent Ventilator
For PIP High and Low Alarm limits, the user can set the High PIP alarm from 20 to 100 cm
0 and the low PIP alarm from 3 to 35 cm H20, or turn OFF the Low PIP alarm.
H
2
Note: PEEP cannot be set within 5 cm H
0 of the High PIP setting.
2
Warning! Set the PIP Low Limit to be at least PEEP +5. This reduces the likelihood of not
detecting a kinked hose. Setting the PIP Low Limit just below the PIP can also serve to
detect leaks and patient circuit as a PIP compensated disconnected threshold.
To change an a PIP secondary parameter, follow these steps:
1. Press the PIP parameter button more than once until desired alarm threshold highlights in
the PIP parameter window.
2. Turn the Dial to set a parameter value.
3. Press the Accept button.
Context Menu
The PIP context menu provides the user the ability to adjust the pressure needed to trigger a
breath minimizing the work of breathing and prevent auto-triggering in the presence of leaks
and very high motion environment.
When ventilating infants and other patients with weak inspiratory effort, the trigger threshold
may need to be adjusted. To reduce the work required for the patient to trigger a breath,
decrease the magnitude.
To prevent auto-triggering, the magnitude of the trigger pressure may need to be increased.
(This should be done with caution as it will increase the work of breathing and may lead to
asynchrony.)
The Spontaneous/Assisted Breath Trigger is preset to -2.0 cm H
-6.0 to -0.5 cm H
breath, the patient must generate -2.0 cm H
O below the baseline (PEEP) pressure. To initiate a spontaneous or assisted
2
O. When the pressure drop is detected, an assisted
2
O and can be adjusted from
2
breath is delivered. The trigger automatically adjusts when the PEEP is changed. The set
Trigger Level threshold displays on the bottom of the screen.
Warning! Set the trigger level so as to minimize the patient's inspiratory effort and prevent
autotriggering. When ventilating infants and other patients with weak inspiratory
effort, lower the trigger threshold magnitude (> -2cm H2O) to reduce the work required
for the patient to trigger a breath. If a large leak is present while ventilating in CPAP or
BL with LC turned on, the trigger threshold magnitude may need to be increased to
prevent auto-triggering with the variable baseline pressure.
Airway pressure measurements are also displayed. The Airway Pressure waveform
(0 to 100 cm H2O) is plotted over time with indications of the PEEP and PIP high limit. See
Figure 4-1. The Mean Airway Pressure (MAP) measurement (0 to 99.9 cm H2O) is provided in
the auxiliary box area of the ventilator display.
4-18 www.zoll.com 9650-002360-01 Rev. A
Page 73

To change a the Trigger Level in the PIP context menu, follow these steps:
1. Press and hold the PIP parameter button until the PIP context menu displays.
2. Release the PIP parameter button, then turn the Dial until the Trigger Level highlights, then
press the Accept button. The current setting for the context menu parameter highlights.
3. Turn the Dial to adjust the value of the parameter.
4. Press the Accept button.
Note: The scale of the Airway Pressure waveform varies based on the PIP High Limit. If the
waveform is too small the PIP High limit maybe set too high.
PIP Parameter Window Reference
The following table provides the options and ranges for the PIP parameter window.
PIP Parameter Window Options/Range Availability/Notes
Primary Parameter PIP 10 to 80 Volume Target:
Secondary Parameters PEEP 0 to 30
Using the Z Vent Ventilator
Measurement
Pressure Target: Control
Setting
PIP values greater than 60
O require the user to
cm H
2
perform a separate
confirmation.
AC and SIMV
Note: SIMV is an optional
mode for the Z Vent.
3 to 30 CPAP
PS 0 to 60 cm H20 Spontaneous Breaths
(SIMV and CPAP)
Note: SIMV is an optional
mode for the Z Vent.
EPAP 3 to 30
IPAP 6 to 60
Alarm Limits High PIP 20 to 100 PEEP cannot be within 5
Low PIP 3 to 35, Off
Measured Value Mean Airway
Pressure
MAP
Paw Waveform 0 to 100
PIP Context Menu
Breath Trigger
(Assisted, Spontaneous)
Default: -2 -6 to -0.5 Adjustment Increments:0.5
0 to 99.9
BL
O of the PIP High
cm H
2
Limit setting.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-19
Page 74

Using the Z Vent Ventilator
FIO2 Parameter Window Options
The FIO2 (Fraction of Inspired Oxygen) parameter window manages oxygen delivery. Pressing
the FIO
Dial. The default value at start up is 21% whether oxygen is present or not.
parameter button highlights the current FIO2 value so that you can adjust it using the
2
If an FIO
begins with that saved FIO
not present, the ventilator begins operation with FIO
value greater than 21% is saved as a custom default or the last setting, the ventilator
2
value if high-pressure oxygen is present. If high-pressure oxygen is
2
= 21% and the O2 SUPPLY PRESSURE
2
LOW alarm is not activated to prevent a nuisance alarm.
The secondary display in the parameter window is O
Use1. This value indicates the flow of
2
high pressure oxygen (l/min) consumed with the current FIO
(from 0 to 99.9 liters/minute). This value can be used to calculate the time of useful O
from an O
cylinder: Flow time (min) = cylinder volume (l) /O2 flow (l/min). O2 Reservoir use
2
is indicated on the display with a plus “+” sign next to the FIO
information.
Note: The “O
Use” parameter does not calculate low flow oxygen only high pressure
2
oxygen use is calculated.
Secondary Parameters and Alarm Thresholds/Limits
There are no secondary parameters for the FIO2 parameter window.
Context Menu
Access the FIO2 context menu to turn the O2 Reservoir ON or OFF.
setting to support the patient
2
flow
2
value. See Chapter 3 for more
2
Note: The O
• When using the O
• In a high vibration environment
• When using biological or chemical filters.
When the O
O
is coming from a low-pressure external source. In addition, it also disables the Gas Intake
2
reservoir feature ON can be used for following:
2
Reservoir for low flow oxygen
2
reservoir feature is on 21+ displays in the FIO2 parameter window indicating that
2
Restricted alarm (3031) that can trigger due to the small resistance caused by the reservoir. This
feature is used to eliminate nuisance alarms caused by the restriction of the external filter or
extremely high vibration. To turn the O
Reservoir feature ON or OFF from the FIO2 context
2
menu, follow these steps:
1. Press and hold the FIO
2. Release the FIO
2
press the Accept button. The current setting for the O
3. Turn the Dial to change the setting to ON or OFF.
4. Press the Accept button.
1. O2 Use = ((FIO2-0.21)/0.79)*Minute Volume where FIO2 is represented as a fraction and minute
volume is the actual minute volume (controlled and spontaneous breaths * tidal volume).
parameter button until the FIO2 context menu displays.
2
parameter button, then turn the Dial until O2 Reservoir highlights, then
Reservoir parameter highlights.
2
4-20 www.zoll.com 9650-002360-01 Rev. A
Page 75

FIO2 Parameter Window Reference
The following table lists the options and ranges for the FIO2 parameter window:
Using the Z Vent Ventilator
FIO2 Parameter Window
Primary Parameter FIO
Primary Parameter FiO2 + 21 Demonstrates O2 reservoir
Secondary Parameters None
Alarm Limits None
Measured Value O2 Use (L/min) 0 to 99.9 Shows when High Pressure
FIO2 Context Menu
O2 Reservoir Default: OFF OFF/ON “+” icon indicates when “ON” for
2
%
SpO2 Parameter Window Options
The SpO2 parameter window controls the Masimo Pulse Oximeter used in the Z Vent
ventilator. The SpO
waveform is plotted when the sensor is used. The parameter window displays a “--”.
measurement (0 to 100%) is shown in the parameter window and the Pleth
2
Options/Range Availability/Notes
21 to 100
mode is in use.
Oxygen Supply is present and
the consumption of flow.
low pressure O
2
Note: The pulse oximeter operates only when the ventilator is providing ventilation.
The following conditions can affect the pulse oximeter reading:
• The sensor is too tight.
• There is excessive illumination from light sources such as a surgical lamp, a bilirubin lamp,
or sunlight.
• A blood pressure cuff is inflated on the same extremity as the one with a SpO
sensor
2
attached.
• Poor perfusion
• The patient has hypotension, severe vasoconstriction, severe anemia, or hypothermia.
• There is an arterial occlusion proximal to the sensor.
• The patient is in cardiac arrest or is in shock.
• Patient movement
• Nail polish or finger pigment
• Carbon monoxide poisoning
The SpO
display is active only when the pulse oximeter is connected. The pulse oximeter is in
2
standby (and displays STBY in the parameter window) when:
• No SpO
• The sensor is off the patient during start up
• You place the pulse oximeter in standby
sensor is connected
2
Note: You can place the pulse oximeter in standby only when the probe is disconnected from
the patient. A valid signal automatically brings the pulse oximeter out of standby.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-21
Page 76

Using the Z Vent Ventilator
Secondary Parameters and Alarm Thresholds/Limits
Pressing the SpO2 parameter button highlights the Low SpO2 Alarm Limit alarm and enables
its value to be changed.
The Low SpO
window. The default Low SpO
adjustable from 86% to 99% (or OFF).
To change an alarm threshold setting, follow these steps:
1. Press the SpO
2. Turn the Dial to adjust the value of the parameter (%).
3. Press the Accept button.
Context Menu
The Masimo context menu is the same for both the SpO2 and HR parameter windows.The
menu provides for the following pulse oximeter controls:
• Standby
• Pulse Ox
• Fast SAT
• Sensitivity
• APOD
• Averaging
• Signal Strength
• Signal IQ
The menu allows for the Pulse Oximeter monitoring to be placed in standby. It also provides
options to turn on the fast SAT, adjust Sensitivity, and other Masimo specific parameters, see
the table below for additional details.
Alarm Limit alarm is the only secondary parameter for the SpO2 parameter
2
value at start up is 94%. The Low SpO2 Alarm Limit is
2
parameter button until the Low SpO2 Alarm Limit highlights.
2
To change a menu setting, follow these steps:
1. Press and hold the SpO
2. Release the SpO
2
parameter button until the menu displays.
2
parameter button, turn the Dial until the Context menu item you want to
change highlights, then press the Accept button. The current setting for the context menu
parameter highlights.
3. Turn the Dial to adjust the value of the parameter.
4. Press the Accept button.
4-22 www.zoll.com 9650-002360-01 Rev. A
Page 77

SpO2 Parameter Window Reference
The following table provides the options and ranges for the SpO2 Masimo context menu.
Using the Z Vent Ventilator
SpO2 Parameter Window
Primary Parameter SpO
Secondary Parameters None
Alarm Limits Low SpO2 Limit
Measured Value Pleth Waveform
Masimo Context Menu
Pulse Ox Default: Standby Standby, Off,
Fast SAT Default: Off Off, On Fast SAT enables rapid tracking
Sensitivity Norm Max, Norm Norm adjusts the pleth signal
2
%
Alarm
Options/Range Availability/Notes
0 to 100 Measurement only
86 to 99, Off
Default: 94%
On
of arterial oxygen saturation
changes by minimizing the
averaging. This mode is
clinically applicable during
procedures when detecting
rapid changes in SpO
paramount such as induction,
intubation, and sleep studies.
sensitivity. Max interprets and
displays data for even the
weakest of signals. Max is
recommended during
procedures and when clinician
and patient contact is
continuous.
is
2
APOD Off Off, On When on, this mode improves
detection of the “probe off
patient” condition, but reduces
the ability to acquire a reading
on patients of low perfusion.
Averaging 8 Seconds 2 to 4, 4 to 6, 8,
10, 12, 16
Seconds
Signal Strength Measured Value 0 to 20 Current signal strength value,
Signal IQ Measured Value Bar Graph
Adjusts the SpO
averaging durations.
not adjustable. A value of zero
indicates that no measurement
is available. This value helps
clinicians place sensors on the
optimal site.
Bar graph displays the relative
reliability of the pulse oximeter
signal.
and HR
2
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-23
Page 78

Using the Z Vent Ventilator
HR (Heart Rate)
The HR parameter window displays the patient’s heart rate when the pulse oximeter is working
and the sensor is attached. The HR measurement (0 to 240 beats per minute) is shown in the
parameter window and the Pleth waveform is plotted when the pulse oximeter is used. During
monitoring “--” displays when the sensor does not detect a measurement. The heart icon blinks
at the pulse rate.
Secondary Parameters and Alarm Thresholds/Limits
The secondary parameters for the HR parameter window are the following:
• HR High Limit (adjustable from 80 to 240 beats per minute (or OFF)
• HR Low Limit (adjustable from 30 to 79 beats per minute (or OFF)
To change an alarm threshold setting, follow these steps:
1. Press the HR parameter button more than once until desired limit setting highlights.
2. Turn the Dial to adjust the value of the parameter.
3. Press the Accept button.
Context Menu
The menu supports both HR and SpO2 parameter windows. The menu provides for the
following pulse oximeter controls:
• Standby
• Pulse Ox
• Fast SAT
• Sensitivity
• APOD
• Averaging
• Signal Strength
• Signal IQ
The menu allows for the Pulse Oximeter monitoring to be placed in standby. It also provides
options to turn on the fast SAT, adjust Sensitivity, and other Masimo specific parameters, see
table below for additional details.
To change a menu setting, follow these steps:
1. Press and hold the HR parameter button until the menu displays.
2. Release the HR parameter button, turn the Dial until the Context menu item you want to
3. Turn the Dial to change the setting to the desired value.
4. Press the Accept button.
change highlights, then press the Accept button. The current setting for the context menu
parameter highlights.
4-24 www.zoll.com 9650-002360-01 Rev. A
Page 79

HR Parameter Window Reference
The following table gives the options and ranges for the HR parameter:
HR Parameter Window Options/Range Availability/Notes
Primary Parameter HR
b/min
Secondary Parameters None
Using the Z Vent Ventilator
0 to 255 Measurement - Heart icon
blinks at the pulse rate
Alarm Limits
Masimo Context Menu
Pulse Ox Default: Standby Standby, Off,
Fast SAT Default: Off Off, On Fast SAT enables rapid tracking
Sensitivity Norm Max, Norm Norm adjusts the pleth signal
APOD Off Off, On When on, this mode improves
Averaging 8 Seconds 2 to 4, 4 to 6, 8,
Signal Strength Measured Value 0 to 20 Current signal strength value,
Signal IQ Measured Value Bar Graph
HR High Limit 80 to 240, OFF
HR Low Limit 30 to 79, OFF
On
10, 12, 16
Seconds
of arterial oxygen saturation
changes by minimizing the
averaging. This mode is
clinically applicable during
procedures when detecting
rapid changes in SpO
paramount such as induction,
intubation, and sleep studies.
sensitivity. Max interprets and
displays data for even the
weakest of signals. Max is
recommended during
procedures and when clinician
and patient contact is
continuous.
detection of the “probe off
patient” condition, but reduces
the ability to acquire a reading
on patients of low perfusion.
Adjusts the SpO
averaging durations.
not adjustable. A value of zero
indicates that no measurement
is available. This value helps
clinicians place sensors on
optimal sites
Bar graph displays the relative
reliability of the pulse oximeter
signal.
2
and HR
2
is
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-25
Page 80

Using the Z Vent Ventilator
Manual Breath
The Manual Breath button enables the user to deliver a manual breath to a patient when pressed
during the expiratory phase and then only after the airway pressure drops to the PEEP target.
The effect of the manual breath button depends on the ventilation mode set on the ventilator.
• In AC and SIMV, a button press will deliver a mandatory breath defined by the settings.
• In CPAP and BL, it delivers a mandatory breath based on the Apnea Backup settings.
Figure 4-3 shows the Manual Breath Button on the Z Vent ventilator.
Note: If the ventilator supports the Plateau Pressure option, the button is Labeled “Manual
Breath/ P Plat”.
Plateau Pressure
Plateau pressure (P Plat) is the pressure applied to small airways and alveoli. It is measured
during an inspiratory pause on the ventilator. Current clinical practice seeks to maintain the
plateau pressure < 28-30 cm H
Note: Plateau Pressure is optional on the Z Vent ventilator. If the ventilator support the
Plateau Pressure option, the Manual Breath button is Labeled “Manual Breath/ P Plat”
(see Figure 4-3).When Plateau Pressure is available on the Z Vent ventilator, the
Manual Breath button performs two functions:
• Allow the user to deliver a manual breath
• Enables the user to perform a plateau pressure maneuver.
O to prevent excessive pressure in the lung.
2
Plateau Pressure Maneuver
To perform a P Plat maneuver, press and hold the Manual Breath button during the inspiratory
period. The maneuver is used during volume targeted breath. During pressure targeted
breathing the PIP typically represents to alveolar pressure as flow decelerates to 0 l/min at the
end of inspiration.
Holding the button past the end of inspiration during a mandatory breath causes the ventilator
to delay opening the exhalation valve for as long as the button is held (up to 3 seconds).
During this period the ventilator measures the pressure while there is no gas flow. At the end of
the period the exhalation valve opens and the device continues normal operation while the P
plat is displayed for ~20s
The activation of a new alarm or pressing the Mute/Cancel button also clears the Plateau
Pressure measurement.
4-26 www.zoll.com 9650-002360-01 Rev. A
Page 81

Using the Z Vent Ventilator
Popup Messages
To prevent the setting of parameter values that are outside the typical clinical range of settings,
the ventilator presents popup messages that ask if you are sure you would like to set the
parameter beyond the typical range. A sample popup message is shown in Figure 4-4.
When a message occurs, you are asked to press the Accept button before you can adjust a
parameter beyond the typical range. Popup messages are also used to alert you that certain
settings are not permitted or possible based on the current parameter settings. In addition,
popup messages can call for you to press the Accept button to acknowledge that you are
entering configurations where certain alarms are being suppressed, turned “off”, and/or
canceled.
Figure 4-3 Manual Breath/Pressure Plateau Button
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-27
Page 82

Using the Z Vent Ventilator
Figure 4-4 Popup Message Example
In addition to addressing setting conflicts, popup messages are used for confirmation (for
example, alarms). These messages also appear whenever you attempt to adjust the ventilator in
a way that is outside clinical norms or is outside its performance range.
Popup messages also appear when you are required to confirm their action before you proceed.
For example, setting the low breath rate alarm below 4 would allow for 15 second apnea
periods and would, in essence, disable the alarm for some patients. If the desired value is
outside the performance range, the popup message alerts you to why you cannot make the
change. (Example: trying to set the PEEP level greater than the PIP setting).
A list of all popup messages follows.
4-28 www.zoll.com 9650-002360-01 Rev. A
Page 83

Popup Message List
Popup/Information/Message
Requested Compressor Flow Too Low
Popup message triggers when the rate/tidal volume/FIO
than the flow capability of the compressor. Resolution involves changing a setting to increase the
flow required from the compressor if possible. Note: this condition is only possible with infant setting
and for FIO
Message: Reduce FiO
Requested Compressor Flow Too High
Popup message triggers when the user attempts to adjust the ventilator so that flow from the
compressor
is > 100 l/min.
Message: Cannot exceed 100 LPM total flow
2
combination requires a flow that is less
2
< 25% at very low tidal volume with long inspiratory time.
, increase BPM, reduce I Time, or increase Vt
2
Using the Z Vent Ventilator
Requested O
Flow Too Low
2
Popup message triggers when the rate/tidal volume/FIO2 combination requires a flow that is less
than the flow capability of the O
required from the O
valve if possible.
2
Note: This condition is only possible with infant setting and for FIO
valve. Resolution involves changing a setting to increase the flow
2
< 25%.
2
Message: Increase FiO2 , increase BPM, reduce I Time, or increase Vt
Requested O2 Flow Too High
Popup message triggers when the user attempts to adjust the ventilator so that flow from the O2
valve
is > 100 l/min.
Message: Cannot exceed 100 LPM total flow
Total Requested Flow Too High
Popup message triggers when the user attempts to adjust the ventilator so that the combined flow
from the compressor and O
valve is > 100 l/min.
2
Message: Cannot exceed 100 LPM total flow
Total Requested Flow Too Low
Popup message triggers when the user attempts to adjust the ventilator so that combined flow from
the compressor and O
valve is < 2 l/min.
2
Message: Cannot flow less than 2 LPM total flow
Alarm Disable
Popup message triggers when the user attempts to adjust to disable an alarm by setting the value
to 0 or the maximum value which would render the alarm essentially off.
Message: Confirmation required -- press accept key to disable alarm
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-29
Page 84

Using the Z Vent Ventilator
Popup/Information/Message
BPM Setting Conflict
Popup message triggers when the user attempts to set the BPM to a value that would result in an
inspiratory time (I Time) > 3 seconds.
Message: I Time cannot exceed 3 seconds
BPM Setting Conflict
Popup message triggers when the user attempts to set the BPM to a value that would result in an
inspiratory time (I Time) > 5 seconds during inverse I:E ratio ventilation.
Message: I Time cannot exceed 5 seconds with inverse I:E
E Time Range Exception
Popup message triggers when the user attempts to set the BPM to a value that would result in an
expiratory time (E Time) < 0.3 seconds.
Message: E Time must be greater than 0.3 seconds
I:E Setting Conflict
Popup message triggers when the user attempts to transition from AC mode using an inverse I:E
ratio to another mode where inverse I:E is not allowed.
Message: Inverse I:E only allowed in AC - Mode change will reset I:E to 1:3
I:E Setting Conflict
Popup message triggers when the user attempts to set an inverse I:E ratio in an mode other than
Assist/Control (AC).
Message: Inverse I:E Not Allowed
BPM Setting Conflict
Popup message triggers when the user attempts to set a BPM rate that will result in an I:E ratio >
1:99.
Message: I:E > 1:99 not allowed
I Time Range Exception
Popup message triggers when the user attempts to adjust the ventilator so that flow from the
compressor
is > 100 l/min.
Message: Cannot exceed 100 LPM total flow
I Time Range Exception
Popup message triggers when the user attempts to SET inspiratory time (I Time) > 5 seconds
during inverse I:E ratio ventilation.
Message: I Time cannot exceed 5 seconds with inverse I:E
I Time Range Exception
Popup message triggers when the user attempts to set an inspiratory time (I Time) < 0.1 seconds.
Message: I Time must be greater than 0.1 seconds
4-30 www.zoll.com 9650-002360-01 Rev. A
Page 85

Using the Z Vent Ventilator
Popup/Information/Message
I:E Range Exception
Popup message triggers when the user attempts to set an inverse I:E ratio < 4:1.
Message: I:E < 4:1 not allowed
I:E Range Exception
Popup message triggers when the user attempts to set an I:E ratio > 1:99.
Message: I:E > 1:99 not allowed
Vt Limit Conflict
Popup message triggers when the user attempts to set the Vt lower than the Vt Low alarm limit.
Message: Cannot adjust Vt Set below Vt Low Alarm
Vt Limit Conflict
Popup message triggers when the user attempts to set the Vt higher than the Vt High alarm limit.
Message: Cannot adjust Vt Set above Vt High Limit
High Vt Setting
Popup message triggers when the user attempts to set Vt > 1000 ml. To do this, the user must
press the Accept button and then continue to set a value > 1000 ml followed by Accept again to
confirm the setting change.
Message: Confirmation required -- press accept key to allow Vt > 1000ml
PEEP Setting Conflict
Popup message triggers when the user attempts to set the PEEP setting ≤ 5 cm H
O below the PIP
2
High pressure limit.
Message: Cannot adjust PEEP target to within 5 of PIP High Limits
PEEP Setting Conflict
Popup message triggers when the user attempts to configure the ventilator so that the PEEP plus
the pressure support (PS) are > the PIP High pressure limit.
Message: PEEP + PS cannot be greater than PIP High Limit
PEEP Backup Setting Conflict
Popup message triggers when the user attempts to set the PEEP setting ≤ 5 cm H
O below the
2
Apnea Backup PIP pressure during CPAP or BL mode ventilation.
Message: Cannot adjust PEEP target to within 5 of backup PIP target
PEEP Setting Conflict
Popup message triggers when the user attempts to set the PEEP ≤ 5 cm H
O below PIP pressure.
2
Message: Cannot adjust PEEP target to within 5 of PIP target
PEEP+PS Setting Conflict
Popup message triggers when the user attempts to set a combination of PEEP and PS that is < 3
cm H
O.
2
Message: Cannot adjust PEEP+PS below 3
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-31
Page 86

Using the Z Vent Ventilator
Popup/Information/Message
High Pressure Target Setting
Popup message triggers when the user attempts to set the PIP pressure > 60 cm H
the user must press the Accept button and then continue to set a value > 60 cm H
Accept again to confirm the setting change.
Message: Confirmation required -- press accept key to exceed 60 cm H
PIP Setting Conflict
Popup message triggers when the user attempts to set the PIP target ≤ 5 of PEEP pressure.
Message: Cannot adjust PIP target to less than 5 more than PEEP
PIP Setting Conflict
Popup message triggers when the user attempts to set the PIP > the PIP High pressure limit.
Message: Cannot adjust PIP target higher than PIP High Limit
BPM Limit Conflict
Popup message triggers when the user attempts to set the BPM High Limit < the BPM Low limit.
Message: Cannot adjust high limit lower than low limit
O. To do this,
2
O followed by
2
O
2
Low Breath Rate Setting
Popup message triggers when the user attempts to set the BPM < 6 bpm. Doing this could, in
effect, disable the alarm for some patients. To do this, the user must press the Accept button and
then continue to set a value < 6 bpm followed by Accept again to confirm the setting change.
Message: Confirmation required -- press accept key for values below 6 BPM
BPM Limit Conflict
Popup message triggers when the user attempts to set the BPM Low limit > the BPM High limit.
Message: Cannot adjust low limit higher than high limit
Vt Limit Conflict
Popup message triggers when the user attempts to set the Vt high limit < the Vt low limit.
Message: Cannot adjust high limit lower than low limit
Vt Limit Backup Setting Conflict
Popup message triggers during CPAP or BL mode when the user attempts to set the Vt high limit <
the Vt setting in the Apnea Backup settings.
Message: Cannot adjust high limit lower than Backup Vt Setting
Vt Limit Conflict
Popup message triggers when the user attempts to set the Vt high limit < the Vt setting.
Message: Cannot adjust high limit lower than Vt Setting
High Vt Limit Setting
Popup message triggers when the user attempts to set the Vt limit > 1500 ml. Doing this could, in
effect, disable the alarm for some patients. To do this, the user must press the Accept button and
then continue to set a value > 1500 ml followed by Accept again to confirm the setting change.
Message: Confirmation required -- press accept key for values above 1500ml
4-32 www.zoll.com 9650-002360-01 Rev. A
Page 87

Using the Z Vent Ventilator
Popup/Information/Message
Vt Limit Conflict
Popup message triggers when the user attempts to set the Vt low limit < Vt high limit.
Message: Cannot adjust low limit higher than high limit
Vt Limit Conflict
Popup message triggers during SIMV (V) when the user attempts to set the Vt low limit > the
current Vt.
Message: Cannot adjust low limit higher than Vt Setting
Vt Limit Backup Setting Conflict
Popup message triggers during CPAP or BL mode when the user attempts to set the Vt low limit >
the Vt setting in the Apnea Backup settings.
Message: Cannot adjust low limit higher than Backup Vt Setting.
High Pressure Limit Setting
Popup message triggers when the user attempts to set the PIP > 60 cm H
must press the Accept button and then continue to set a PIP > 60 cm H
again to confirm the setting change.
Message: Confirmation required -- press accept key to exceed 60 cm H
O. To do this, the user
2
O followed by Accept
2
O
2
PIP Limit Conflict
Popup message triggers when the user attempts to set the PIP High limit > the PIP Low limit.
Message: Cannot adjust high limit lower than low limit
PIP Limit Backup Setting Conflict
Popup message triggers during CPAP or BL mode when the user attempts to set the PIP High limit
< Apnea Backup PIP limit.
Message: Cannot adjust high limit lower than backup PIP target
PIP Limit Conflict
Popup message triggers when the user attempts to set the PIP High limit < the PIP Low limit.
Message: Cannot adjust high limit lower than PIP target
PIP Limit Conflict
Popup message triggers when the user attempts to set the PIP High limit < the combination of PS
and PEEP pressures.
Message: Cannot adjust high limit lower than PS + PEEP
PIP Limit Conflict
Popup message triggers when the user attempts to set the PIP Low limit > the PIP high limit.
Message: Cannot adjust low limit higher than high limit
Heart Rate Limit Conflict
Popup message triggers when the user attempts to set the HR High limit < the HR Low limit.
Message: Cannot adjust high limit lower than low limit
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-33
Page 88

Using the Z Vent Ventilator
Popup/Information/Message
Heart Rate Limit Conflict
Popup message triggers when the user attempts to set the HR Low limit > the HR High limit.
Message: Cannot adjust low limit higher than high limit
PS Conflict
Popup message triggers when the user attempts to set the PS > the PIP High limit - PEEP
pressure.
Message: Cannot adjust PS higher than PIP High Limit - PEEP
Leak Comp.
Popup message triggers when the user attempts to initiate leak compensation (LC). To do this, the
user must press the Accept button and then select LC followed by Accept again to confirm the
setting change.
Message: Some Alarms Disabled! Configure Alarms for Patient!
Mode Conflict
Popup message triggers when the user attempts to initiate leak compensation (LC) during volume
targeted ventilation. Note: LC is only available during pressure targeted ventilation.
Message: Cannot select Volume targeted control breaths with Leak Compensation on -- first turn
Leak Compensation off
Inverse I:E
Popup message triggers when the user attempts to set an inverse I:E ratio. To do this, the user
must press the Accept button and then adjust the I:E ratio to the desired inverse value and press
Accept again to confirm the setting change.
Message: Confirmation required -- press accept key to allow inverse I:E
Excessive Volume for Infant Circuit
Popup message triggers when the user attempts to set a Vt > 300 ml when the tubing compliance
compensation is set to Pediatric.
Message: Press accept to confirm use of adult circuit
Insufficient Volume for Adult/Ped Circuit
Popup message triggers when the user attempts to set a Vt < 200 ml when the tubing compliance
compensation is set to Adult.
Message: Press accept to confirm use of infant circuit
High PEEP Setting
Popup message triggers during CPAP mode when the user attempts to set the PEEP > 15 cm H
To do this, the user must press the Accept button and then adjust PEEP to the desired value and
press Accept again to confirm the setting change.
Message: Confirmation required -- press accept key to allow PEEP above 15
High EPAP Setting
Popup message triggers during BL mode when the user attempts to set the EPAP > 15 cm H2O. To
do this, the user must press the Accept button and then adjust PEEP to the desired value and
press Accept again to confirm the setting change.
Message: Confirmation required -- press accept key to allow EPAP above 15
2
O.
4-34 www.zoll.com 9650-002360-01 Rev. A
Page 89

Using the Z Vent Ventilator
Popup/Information/Message
EPAP Setting Conflict
Popup message triggers during BL mode when the user attempts to set the EPAP < 3 cm H
below the IPAP target.
Message: Cannot adjust EPAP target to within 3 of IPAP target
EPAP Setting Conflict
Popup message triggers during BL mode when the user attempts to set the EPAP < 5 cm H2O
below the Apnea Backup PIP target.
Message: Cannot adjust EPAP target to within 5 of backup PIP
PIP Limit Conflict
Popup message triggers during BL mode when the user attempts to set the PIP Limit) < the IPAP
target.
Message: Cannot adjust high limit lower than IPAP target
IPAP Setting Conflict
Popup message triggers during BL mode when the user attempts to set the IPAP < 3 cm H
above the EPAP setting.
Message: Cannot adjust IPAP target to less than 3 more than EPAP
O
2
O
2
IPAP Setting Conflict
Popup message triggers during BL mode when the user attempts to set the IPAP < the PIP limit.
Message: Cannot adjust IPAP target higher than PIP High Limit
High IPAP Setting
Popup message triggers during BL mode when the user attempts to set the IPAP > 30 cm H
O. To
2
do this, the user must press the Accept button and then adjust IPAP to the desired value and press
Accept again to confirm the setting change.
Message: Confirmation required -- press accept key to allow IPAP above 30 cm H
O
2
High PEEP+PS Setting
Popup message triggers in CPAP mode when the user attempts to set the combination of PEEP +
PS < 30 cm H2O. To do this, the user must press the Accept button and then adjust PEEP or PS to
the desired value and press Accept again to confirm the setting change.
Message: Confirmation required -- press accept key to allow PEEP+PS above 30 cm H
O
2
High Pressure Limit Setting
Popup message triggers when the Start Menu is active and the user selects either the Custom or
Last Setting options and the PIP high limit is >35 cm H
O. When the user selects one of the options
2
where this is true, the popup message is triggered requiring the user to provide additional
conformation, pressing Accept, to initiate ventilation with the option.
Message: Confirmation required -- press accept key to recall limit setting above 35 cm H
O
2
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-35
Page 90

Using the Z Vent Ventilator
Transitions
Transitioning between modes or between volume and pressure targeting the device may adjust
or set any of the following parameters:
• BPM, BPM High Limit, BPM Low Limit
• VT, VT High Limit, VT Low Limit
• PIP, PIP High Limit, PIP Low Limit
• PEEP
• PS / IPAP
• I:E Ratio / Ti
• Rise Time
• LC
These parameters are adjusted or populated to safely transition the settings that were not
available in the previous mode. After changing the mode or ventilation target, the parameters
and alarm limits should be adjusted to be appropriate for the patient being ventilated. It is
recommended to adjust the parameters and alarm limits by window moving in order up the
device from the Mode parameter window.
Warning! The alarm limits must be appropriate for the patient being ventilated. If a parameter is
changed, adjust the high and low alarm limit to bracket selected value.
Warning! The transition into CPAP or BL from invasive modes automatically sets the rise time
to 3, which may be too fast for infants and small children. Before using with an infant or
small child, the user should always configure the ventilator appropriately before
attaching the patient.
Using the Menu
The Menu button displays the Menu, which allows you to change various ventilator options,
such as the contrast or brightness of the Display Screen (LCD Contrast/LCD Brightness).
When you press the Menu button, the Menu appears:
• Alarm Config
• Powerup Settings
• LCD Contrast
• LCD Brightness
• UTC Offset
• Unit Info
• Alarm History
Alarm Configuration
Selecting Alarm Configuration allows the user to enable or disable the audible and visible alerts
associated with specific alarms to prevent nuisance alarms.
Refer to, “Managing Alarms” in Chapter 5.
4-36 www.zoll.com 9650-002360-01 Rev. A
Page 91

Powerup
Using the Z Vent Ventilator
You can configure how the ventilator starts (powers up) based on your use or patient
population. The Powerup settings include:
Powerup Setting
(Option)
Powerup Adult, Pediatric, or Start Menu
Custom Settings Saves the currently set operating parameters
Language English, Language Option
Setting Choices
In addition to controlling how the ventilator starts up, the Powerup settings sub-menu also
allows the user to save the current ventilator configuration for use as the Custom starting
configuration and which language will be used when the ventilator is turned ON.
Specifying Powerup Settings
To configure new Powerup settings, follow these steps:
1. Press the Menu button to display the menu, then turn the Dial to highlight the Powerup
settings.
2. Press the Accept button. The following list of configurable items with
Powerup highlighted displays.
• Powerup
• Custom Settings
• Language
3. Press the Accept button, then turn the Dial to the desired starting configuration.
• Adult Settings
• Pediatric Settings
• Start Menu
4. Press the Accept button to confirm the new starting configuration.
5. Press the Mute/Cancel button to return to the Menu.
6. Press the Mute/Cancel button again to return to the configured Powerup setting (Start menu,
Adult, or Pediatric).
Specifying Custom Settings
To save the current configuration parameter settings (if different than Adult or Pediatric default
parameter settings) for use at start up, follow these steps:
1. Press the Menu button, then turn the Dial to Powerup Settings, then press the Accept
button.
2. Turn the Dial to highlight the Custom Settings choice, and then press the Accept button
(Save becomes highlighted).
3. Press the Accept button again to save the current parameter settings.
4. Press the Mute/Cancel button to return to the menu.
5. Press the Mute/Cancel button again to return to the configured Powerup setting (Start Menu,
Adult, or Pediatric).
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-37
Page 92

Using the Z Vent Ventilator
Specifying a Language
The ventilator is provided with the English language and a local language. (This feature is
limited to ventilators with multiple languages.).
LCD Contrast
The LCD Contrast submenu allows the user to adjust the contrast of the ventilator display
screen to optimize visibility in the current lighting environment. Given the use environment, the
ventilator automatically compensates for the temperature changes that can effect LCD performance.
However, in temperatures outside its performance specification of- 26 °C to 55 °C (15 °F to 131 °F), or
when the ventilator temperature is rapidly changing (warm storage into a very cold environment) the
contrast can fade making viewing difficult.
To adjust the LCD screen contrast, follow these steps:
1. Press the Menu button to display the Menu.
2. Turn the Dial to highlight LCD Contrast, and then press the Accept button.
3. Turn the Dial clockwise to increase contrast (counterclockwise to decrease it) while watching
the LCD screen to determine the best contrast value based on visibility, then press the Accept
button to set the new contrast value.
4. Press the Mute/Cancel button to return to the Menu.
Note: The contrast value is saved between uses. This may require adjusting the contrast value
when the temperature is within the ventilator’s operating range or with the next use.
LCD Brightness
The LCD Brightness control allows the user to adjust the brightness of the LCD display screen
and LED Array to optimize visibility in the current lighting environment.
The initial numerical value is 25. The brightness ranges from 0 to 32.
To adjust the brightness, follow these steps:
1. Press the Menu button to display the menu.
2. Turn the Dial to highlight LCD Brightness, and then press the Accept button.
3. Turn the Dial clockwise to increase brightness (counterclockwise to decrease it) while
watching the LCD screen and LED Array to determine the best brightness value based on
visibility, then press the Accept button to set the new brightness value.
4. Press the Mute/Cancel button to return to the Menu.
In the event the LCD is not viewable due to extreme temperature conditions there is a
short cut to the contrast control. Press and hold the Menu button for 2 to 3 seconds.
Doing this, jumps the menu highlight directly to the contrast control where the contrast
can be adjust as described above.
4-38 www.zoll.com 9650-002360-01 Rev. A
Page 93

UTC Offset
Using the Z Vent Ventilator
The UTC Offset control allows the user to set the ventilator clock to match the local time zone
relative to Coordinated Universal Time (UTC). Matching the local time zone through the
UTC Offset setting allows an accurate time record of events recorded in the alarm log.
Before setting the UTC Offset, the user must know the UTC Offset value for your timezone.
When determining the UTC Offset value, remember to consider variances for Daylight Saving
Time (DST).
Unit Info
Once set,
• You change the setting (required for daylight savings time)
• The ventilator is serviced.
the setting is retained until:
To adjust the UTC Offset, follow these steps:
1. Press the Menu button to display the Menu.
2. Turn the Dial to highlight UTC Offset, and then press the Accept button.
3. Turn the Dial clockwise or counterclockwise to find the offset value for your timezone, then
press the Accept button to set the new contrast value.
4. Press the Mute/Cancel button to return to the Menu.
Note: The UTC offset changes the time (min/max of 12 hrs). This offset is only applied to the
time, not to the date stored in the device.
The Unit Info choice allows the user to view the following information about the ventilator as
follows:
Item Description
Use Statistics Provides access to ventilator usage in hours and minutes.
Date The current calendar date
Cal Date Last date the calibration was checked
PM Cycle Annual
SPM SW Rev Software revision of the Smart Pneumatic Module
SPM SN Serial number of the Smart Pneumatic Module
SPM Model Smart Pneumatic Module model
EMV Soft Rev Software revision of the ventilator model
Device SN Serial number of the ventilator model
Device Model Ventilator model
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 4-39
Page 94

Using the Z Vent Ventilator
To view unit information for the ventilator, follow these steps:
1. Press the Menu button to display the Menu.
2. Turn the Dial to highlight Unit Info, and then press the Accept button. The list of information
displays with Use Statistics highlighted.
3. Press the Accept button to display ventilator usage in hours and minutes (hh:mm)
4. Press the Mute/Cancel button to return to the Menu.
Alarm History
The Alarm History provides a list of the alarm messages generated by the ventilator during use.
The history holds a maximum of 256 events.
Note: Power cycling does not clear the alarm history.
Each list entry provides the following information
• The Alarm Name
• The Service Code
• The calendar date when the event was entered
• Whether the alarm is set or cleared
• The time (24hr) when the event was entered
• The log entry number <NNN> The higher the number the more recent the alarm message
event was logged
To display the Alarm History, follow these steps:
1. Press the Menu button to display the Menu.
2. Turn the Dial to highlight Alarm History, and then press the Accept button. The most recent
history entry displays.
3. Turn the Dial counterclockwise to view earlier entries. Turn the Dial clockwise to view more
recent entries.
4. Press the Mute/Cancel button to return to the Menu.
5. Press the Mute/Cancel button again to return to the user interface.
The alarm condition is not logged when alarm is active but not displayed (due to other higher
priority alarm indication as explained in the Alarms Chapter of this manual), or if the alarm is
disabled in the alarm configuration the condition is not logged.
4-40 www.zoll.com 9650-002360-01 Rev. A
Page 95

This chapter provides a detailed description and comprehensive reference for the
ZOLL Z Vent ventilator’s alarms. This chapter addresses the following:
• Describes alarm types and priorities
• Provides a comprehensive list of alarms
Alarm Overview
To safeguard the patient, the ventilator continuously monitors the patient, device, and
environment to ensure that all of the systems are functioning as intended. When device detects
a problem, it triggers an alarm and displays a Smart Help message to alert you.
Smart Help presents, a multi-line message in the upper left-hand corner of the display screen.
This screen area is the Alarm Message Center (AMC). The AMC displays the alarm name with
a series of instructions to help you resolve the alarm. The device prioritizes alarms based on the
risk to the patient and always presents the alarm with the greatest risk to the patient first. All
messages are context-based and suggest what is causing the alarm, as well as show how it can
be resolved.
Chapter 5
Alarms
The Alarm Message Center (AMC) contains the information and instructions for all active
alarms, as shown in Figure 5-1.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 5-1
Page 96

Alarms
Figure 5-1 Smart Help Alarm Display
A. Alarm Name: describes the type or cause of the alarm. When more than one alarm occurs at
the same time, the device prioritizes the alarms based on the highest risk to the patient.
B. Mitigation/Resolution Instructions: prioritized instructions that give more information
about the nature of the alarm and describe how to resolve the alarm state.
C. If Not Resolved Instructions: instructions for what to do you cannot resolve the alarm
state. The instruction is always shown in the following format **Message...**.
D. Alarm Icons: for each active alarm, an alarm bell appears. When multiple alarms are active,
the number of bells corresponds to the number of alarms. The alarm in the AMC is
displayed as the solid bell. To view each active alarm, turn the Dial to scroll through all
active alarms. The plot icon is also in this list. It allows you to see the current waveform to
better assess the nature of the failure. A maximum of five alarms can be displayed without
the plot icon.
E. Service Code: each alarm has a 4 digit number associated with it, which helps the user
indicate the specific alarm when communicating with technical support. The service codes
appear in the following format:
1### High Priority Alarm
2### Medium Priority Alarm
3### High Priority Alarm
F. Attention Warning Icon: identifies the severity of the alarm: Low, Medium, or High
priority. See the Symbols table in Chapter 1 for the appearance of the warning triangle for
each of these three alarms.
5-2 www.zoll.com 9650-002360-01 Rev. A
Page 97

Alarms
Muting Alarms
During device operation, the mute behavior depends on the alarm priority (see section below).
There are other mute behaviors used by the alarm system. See “Managing Alarms” for
Preemptive Mute and Startup Mute descriptions.
Warning! Use in High Noise Environments — in high noise environments, you may be inclined not
to mute the alarm while addressing the problem. Not pressing Mute limits the user's
ability to resolve the alarm because with each breath the alarm is re-triggered and any
parameter changes you are attempting are canceled as the alarm re-triggers.
Alarm Priorities
Alarm priorities define the operational status of the device and its ability to provide mechanical
ventilation. The alarm priorities are as follows:
High Priority
Mechanical ventilation under user control is no longer possible. This alarm priority requires
immediate intervention indicated by fast red blinking alarm LED and audio signal. This
includes system failure alarms where the CPU has failed and a backup has taken over to sound
the audible and visual alarms. It can also occur when the device is turned on and there is no
internal or external power source.
Pressing the Mute/Cancel button has no effect on a high priority alarm. The alarm can only be
silenced by turning off the ventilator. Some alarms can be resolved by turning the ventilator off,
then on again.
Medium Priority
Mechanical ventilation is active or is possible (maybe for a finite period of time) but, there is a
failure or fault with the patient, ventilator circuit, a pneumatic subsystem, or pulse oximeter.
This alarm priority requires immediate intervention by the user indicated by a slow red blinking
alarm LED and audio signal.
Pressing the Mute/Cancel button mutes medium priority alarms for 30 seconds. If the alarm
trigger still exists after 30 seconds, the audible alarm recurs until you mute it again for another
30 second period or the alarm is resolved.
Low Priority (Advisory)
Safe mechanical ventilation is active but, there is a fault that you must be aware of to ensure
safe management of the patient or ventilator. Low priority alarms present with both an audible
and yellow alarm LED signal (slow blink) alerting you to the condition.
Pressing the Mute/Cancel button cancels the audible signal. If the alarm is not resolved, the
yellow LED remains illuminated to remind you of the fault or failure.
Note: Some Low Priority alarms are canceled and the Alarm LED turns green when you
push the Mute/Cancel button. For others, the audible alarm is canceled but the Alarm
LED stays yellow to remind you that the device is operating in a state that needs
monitoring.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 5-3
Page 98

Alarms
Popup Messages
As described in Chapter 4, popup messages are used to help the user make alarm adjustments
and prevent unintended adjustment of the device. See Chapter 4 for a list of popup messages.
Alarm Types
Alarm types are identified to provide a framework for the alarm scheme and titles. The alarm
indication may be due to patient conditions, breathing circuit conditions, device conditions, or
environment. For a list of all alarms, with descriptions of ventilator behavior and mitigation
actions see Alarm Summary section later in this chapter.
The alarm types are:
• Patient Safety — address the ventilation of the patient and their respiratory effort. Patient
Safety alarm types also includes: pulse oximetry monitoring and circuit/exhalation valve
issues.
• Environmental and Use — address the device inputs: external power, battery, high pressure
O2 supply, and the fresh gas intake. Environmental and Use alarm types also includes:
ambient and device temperature, barometric pressure, and altitude.
• Self Check — address the performance of the device systems and include:
• Internal Communication (Comm): faults/failures of inter-device communication,
cyclic redundancy checking, or processor-related issues
• Pneumatic Sensor: faults/failures of the pneumotachographs that measure gas flow
or the pressure transducers.
• Pneumatic System: faults/failures of the compressor or O
• Power System: faults/failures of the power system that render the device unable to
operate from external power or charge/operate from the internal rechargeable
battery.
• Pulse Oximeter Module: faults/failures of the pulse oximeter module that are not
related to monitoring of the patient, a fault or failure of the module.
• Preventive Maintenance: alarms that occur when device is due for preventive
maintenance.
supply valve.
2
Patient Safety Alarms
The ventilation mode determines whether parameters are controlled by the user. As a result, the
alarms monitoring the patient safety behave differently based on the ventilation mode. Patient
safety alarms may be muted or turned off if the use determines this is appropriate for the
management of the patient.
Note: Popup messages are used to prevent inadvertent setting of alarm limits at their extreme
which could limit the effectiveness of the alarm.
The table below lists alarms related to patient safety, describes default settings and adjustable
ranges in different ventilation modes and provides the Service Code number for each alarm.
5-4 www.zoll.com 9650-002360-01 Rev. A
Page 99

Alarms
Alarm Title (units) Adjustable Range, Default Service Code
Airway Pressure High
PIP High (cm H
0)
2
Low Airway Pressure
PIP Low (cm H
0)
2
High Tidal Volume
Vt High
(ml)
Low Tidal Volume
Vt Low
(ml)
20 to 100 (>60 requires confirmation)
Default: Adult:35 Pediatric: 30
3 to 35, OFF (with popup confirmation; 3
without confirmation)
Default: 10 in AC, SIMV
Default: OFF in CPAP or BL
See transition and feature notes in
Chapter 4.
0 to 2000, OFF (with popup confirmation;
2000 without confirmation)
Default: Adult: N/A Pediatric: 500
Default: OFF in CPAP or BL
See transition and feature notes in
Chapter 4.
0 to 2000, OFF (with popup confirmation;
0 without confirmation)
Default: Adult: N/A Pediatric: 500
Default: OFF in CPAP or BL
See transition and feature notes in
Chapter 4.
2070
2071
2072
2073
High Breath Rate
(breath per minute)
20 to 99, OFF (with popup confirmation;
99 without confirmation)
2074
Default: Adult: 25 Pediatric: 30
Low Breath Rate/Apnea 2 to 25 (with popup confirmation below 6)
Apnea 2076
Default: Adult: 8 Pediatric: 10
2075
PEEP Leak Not adjustable, OFF (with Alarm Menu) 2090
Insufficient Flow Not adjustable 2095
Spontaneous Breath-PIP High 20 to 100
2170
Default = Adult:35 Pediatric: 30
Spontaneous Breath-PIP Low 3 to 35, OFF (with popup confirmation; 3
2171
without confirmation)
Default: 10 in AC, SIMV
Default: OFF in CPAP or BL
See transition and feature notes in
Chapter 4.
9650-002360-01 Rev. A ZOLL Z Vent Ventilator Operator’s Guide 5-5
Page 100

Alarms
Alarm Title (units) Adjustable Range, Default Service Code
Spontaneous Breath-VT High 0 to 2000, OFF (with popup confirmation;
2000 without confirmation)
Default: Adult: N/A Pediatric: 500
Default: OFF in CPAP or BL
See transition and feature notes in
Chapter 4.
Spontaneous Breath-VT Low 0 to 2000, OFF (with popup Confirmation;
0 without confirmation)
Default: Adult: N/A Pediatric: 500
Default: OFF in CPAP or BL
See transition and feature notes in
Chapter 4.
Patient Inspiratory Demand not
Met
Auto-PEEP Not adjustable, OFF (with Alarm Menu) 3091
Patient Disconnect Not adjustable 2100
Exhalation System Failure Patient Can Not Exhale
Exhalation System Failure Patient Can Not Exhale
Not adjustable, OFF (with Alarm Menu) 3092
Not adjustable 1060
Not adjustable 1061
2172
2173
Exhalation Fault Not adjustable 2062
Calibration of Airway Pressure not
successful
Suspicious Triggers Not adjustable 2053
Tubing Compliance Fault Not adjustable 3073
Not adjustable 1051
Patient Safety Alarms Related to Use of a Pulse Oximeter
The primary use of the ventilator is to provide patient ventilation: however, the ventilator has
an integrated pulse oximeter. The pulse oximeter operates only when the ventilator is
ventilating. When there is an issue with the SpO
or HR signals or the value is outside of the
2
alarm limits the device triggers alarms as identified in the table below.
Note: Pulse oximetry is not available when the Start Menu is active, only after ventilation is
started.
Alarm Title (units) Adjustable Range, Default Service Code
SpO
Sensor Off Patient Not adjustable, OFF (with alarm
2
SpO
Low (%) 86 to 99, (with popup confirmation; 86
2
menu)
without confirmation)
Default: 94
2314
2401
5-6 www.zoll.com 9650-002360-01 Rev. A
 Loading...
Loading...