Biomerieux Bact-Alert 3D-60 User Manual

User Manual
95269
(01/2010)
|
|
|
bioMérieux, Inc. |
|
|
|
Box 15969 |
|
|
|
Durham, North Carolina 27704-0969 / USA |
|
|
|
Tel. (1) 800-682-2666 |
|
|
|
bioMérieux® SA |
|
EC |
REP |
|
|
au capital de 12 029 370 € |
||
|
|
|
673 620 399 RCS LYON |
|
|
|
|
|
|
|
69280 Marcy l’Etoile / France |
|
|
|
tél. 33 (0)4 78 87 20 00 / fax 33 (0)4 78 87 20 90 |
[03] |
|
|
http://www.biomerieux.com |

Algeria |
China |
Hungary |
|
bioMérieux Algérie EURL |
bioMérieux China Limited |
bioMérieux Hungária Kft. |
|
Algéria Business Center |
Room 1601-02B & 10 |
Fóto út. 56 (5. emelet) |
|
Les Pins Maritimes - Mohammadia |
Est Ocean Centre |
H-1047 Budapest |
|
Alger |
nº 24A Jiang Guo Men Nei Street |
tel. (36) 1 231 3050 |
|
tel. (213) 21 89 14 81 |
100004 Beijing |
fax (36) 1 231 3059 |
|
fax (213) 21 89 14 82 |
tel. (86) 10 6515 6963 |
India |
|
Argentina |
fax (86) 10 6515 6993 |
||
bioMérieux India Pvt. Ltd |
|||
|
|||
bioMérieux Argentina |
bioMérieux China Limited |
A-32, Mohan Co-Operative Ind. Estate |
|
Av. Congreso 1745 |
Room 2605, South Tower, |
New Delhi 110 024 |
|
C1428BUE |
World Trade Center |
tel. (91) 11 42 09 88 00 |
|
Capital Federal Buenos Aires |
371-375 Huan Shi Dong East Road |
fax (91) 11 24 64 88 30 |
|
tel. (54) 11 5555 6800 |
510095 Guangzhou |
Indonesia |
|
fax (54) 11 5555 6888 |
tel. (86) 20 8762 7010 |
||
Representation Office |
|||
Australia |
fax (86) 20 8762 7015 |
||
bioMérieux Indonesia |
|||
Colombia |
|||
bioMérieux Australia P/L |
Enseval Building |
||
Unit 25 - Parkview Business Centre |
bioMérieux Colombia Ltda |
Kawasan Industri Pulo Gadung - |
|
1, Maitland Place |
Avenida 15 No. 100-43 |
JI. Pulo - Lentut No. 10 |
|
Baulkham Hills NSW 2153 |
Piso 2 |
Jakarta Timur 13920 |
|
tel. (61) 2 8852 4700 |
Bogotá, D.C. |
tel. (62) 21 461 51 11 |
|
fax (61) 2 8852 4777 |
tel. (57) 1 520 0080 |
fax (62) 21 460 41 07 |
|
Austria |
fax (57) 1 520 0088 |
Italy |
|
(57) 1 520 0831 |
|||
bioMérieux Austria GmbH |
bioMérieux Italia S.p.A. |
||
Czech Republic |
|||
Eduard-Kittenberger-Gasse 97 |
Via Fiume Bianco, 56 |
||
Top 3 |
bioMérieux CZ s.r.o. |
00144 Roma |
|
A-1230 Wien |
Business Park Kosice |
tel. (39) 06 523 081 |
|
tel. (43) 186 50 650 |
Jinonická 80 |
fax (39) 06 523 08240 |
|
fax (43) 186 50 661 |
158 00 Praha 5 |
Ivory Coast |
|
Belgium |
tel. (420) 2 57 290 623 |
||
bioMérieux Afrique Occidentale |
|||
(420) 2 57 290 232 |
|||
bioMérieux Benelux s.a./n.v. |
08 BP 2634 |
||
fax (420) 2 57 290 964 |
|||
Media Square |
Avenue Joseph Blohorn |
||
Denmark |
|||
18–19 Place des Carabiniers |
Abidjan 08 |
||
Bruxelles 1030 |
bioMérieux Danmark Aps |
tel. (225) 22 40 93 93/22 40 41 40 |
|
tel. (32) 2 743 01 70 |
Smedeholm 13C |
fax (225) 22 40 93 94 |
|
fax (32) 2 733 55 97 |
2730 Herlev |
Japan |
|
Brazil |
tel. (45) 70 10 84 00 |
||
Sysmex bioMérieux, Ltd. |
|||
fax (45) 70 10 84 01 |
|||
bioMérieux Brasil SA |
Osaki Central Tower 8F |
||
Finland |
|||
Estrada Do Mapuá |
1-2-2 Osaki Shinagawa-ku |
||
491 Taquara - Jacarepaguá |
bioMérieux Suomi Oy |
Tokyo 141-0032 |
|
CEP 22710 261 |
Konalantie 47 C |
tel. (81) 3 6834 2666 |
|
Rio de Janeiro RJ |
FI-00390 Helsinki |
fax (81) 3 6834 2667 |
|
tel. (55) 21 2444 1400 |
tel. (358) 9 8545 6000 |
Korea |
|
fax (55) 21 2445 6025 |
fax (358) 9 8545 6045 |
||
bioMérieux Korea Co., Ltd. |
|||
Canada |
France |
||
1st & 2nd Floor, Yoosung Building |
|||
bioMérieux Canada, Inc. |
bioMérieux SA |
# 830-67 Yeoksam-dong, |
|
7815, Henri-Bourassa West |
69280 Marcy l’Etoile |
Kangnam-gu |
|
Saint Laurent, QC |
tel. (33) (0)4 78 87 20 00 |
Séoul 135-080 |
|
H4S 1P7 |
fax (33) (0)4 78 87 20 90 |
tel. (82) 2 2188 4700 |
|
tel. (1) 514 336 7321 |
http://www.biomerieux.com |
fax (82) 2 547 6263 |
|
fax (1) 514 807 0015 |
Germany |
Mexico |
|
Chile |
|||
bioMérieux Deutschland GmbH |
bioMérieux México SA de CV |
||
bioMérieux Chile S.A. |
Weberstrasse 8 |
Chihuahua 88, col. Progreso |
|
Seminario 131 |
D 72622 Nürtingen |
México 01080, D.F. |
|
Providencia |
tel. (49) 7022 30070 |
tel. (52) 55 5481 9550 |
|
Santiago |
fax (49) 7022 36110 |
fax (52) 55 5616 2245 |
|
tel. (56) 2634 20 92 |
Greece |
Netherlands (The) |
|
fax (56) 2634 20 93 |
|||
bioMérieux Hellas S.A. |
bioMérieux Benelux BV |
||
|
|||
|
Papanikoli 70 |
Boseind 15 |
|
|
15232 Halandri |
P.O. Box 23 |
|
|
Athens |
5280 AA Boxtel |
|
|
tel. (30) 2 10 81 72 400 |
tel. (31) 411 65 48 88 |
|
|
fax (30) 2 10 68 00 880 |
fax (31) 411 65 48 73 |
|
|
|
|

New Zealand |
Singapore |
Thailand |
|
bioMérieux New Zealand Ltd. |
bioMérieux Singaporete. Ltd. |
bioMérieux Thailand Ltd |
|
C/- Logical Freight Solutions |
11 Biopolis Way, Helios, Block 11 |
3195/9 Vibulthani Tower, 4th Floor |
|
12C Rennie Drive, Airport Oaks |
#10-03 Singapore 138667 |
Rama IV Road, Klongton, Klongtoey |
|
Auckland |
tel. (65) 6513 9554 |
Bangkok 10110 |
|
tel. (64) 9 918 6354 |
fax (65) 6478 9501 |
tel. (66) 2 661 56 44 |
|
fax (64) 9 918 6355 |
South Africa |
fax (66) 2 661 56 45 |
|
Norway |
Turkey |
||
bioMérieux South Africa Pty |
|||
bioMérieux Norge AS |
7 Malibongwe Drive |
bioMérieux Diagnostik A.S. |
|
Økernveien 145 |
Randburg 2125 |
Değirmen Sok. Nida Plaza Kat:6 |
|
N-0513, Oslo |
tel. (27) 11 801 91 10 |
34742 Kozyataği-Istanbul |
|
tel. (47) 23 37 55 50 |
fax (27) 11 791 24 19 |
tel. (90) 216 444 00 83 |
|
fax (47) 23 37 55 51 |
Spain |
fax (90) 216 373 16 63 |
|
Philippines (The) |
United Kingdom |
||
bioMérieux España S.A. |
|||
Representation Office |
Manual Tovar, 45–47 |
bioMérieux UK Ltd |
|
bioMérieux Philippines |
28034 Madrid |
Grafton Way, Basingstoke |
|
11th Floor, Pearlbank Centre |
tel. (34) 91 358 11 42 |
Hampshire RG22 6HY |
|
146 Valero Street, Salcedo Village |
fax (34) 91 358 06 29 |
tel. (44) 1256 461881 |
|
1227 Makati City |
Sweden |
fax (44) 1256 816863 |
|
tel. (632) 817 7741 |
USA |
||
bioMérieux Sverige AB |
|||
fax (632) 812 0896 |
|||
Hantverksvägen 15 |
bioMérieux, Inc. |
||
Poland |
|||
436 33 Askim |
100 Rodolphe Street |
||
bioMérieux Polska Sp. Z.o.o. |
tel. (46) 31 68 84 90 |
Durham NC 27712 |
|
Ul. Zeromskiego 17 |
fax (46) 31 68 48 48 |
tel. (1) 919 620 2000 |
|
01-882 Warsaw |
Switzerland |
Vietnam |
|
tel. (48) 22 569 85 00 |
|||
bioMérieux Suisse s.a. |
Representation Office |
||
fax (48) 22 569 85 54 |
|||
51, avenue Blanc |
bioMérieux Vietnam |
||
Portugal |
|||
Case postale 2150 |
Room 4A, 4th Floor |
||
bioMérieux Portugal, Lda. |
1211 Genève 2 |
Green House Building |
|
Av. 25 de Abril de 1974, nº 23-3º |
tel. (41) 22 906 57 60 |
62A Pham Ngoc Thach Street, Ward 6 |
|
2795-197 LINDA-A-VELHA |
fax (41) 22 906 57 42 |
District 3 |
|
tel. (351) 21 415 23 50 |
Taiwan |
Ho Chi Minh City |
|
fax (351) 21 418 32 67 |
tel. (84) 88 209 906 |
||
Representation Office |
|||
Russia |
fax (84) 88 209 905 |
||
bioMérieux China Limited |
|||
|
|||
o.o.o. bioMérieux |
Taiwan Branch |
|
|
Derbenevskaya ul. 20, str. 11 |
RM 608, No. 6-3 Ching Cheng Street |
|
|
115 114 Moscow |
Taipei 105 |
|
|
tel. (7) 495 221 10 79 |
tel. (886) 2 2545 2250 |
|
|
fax (7) 495 221 10 79 |
fax (886) 2 2545 0959 |
|

This product and its documentation complies with the In Vitro Diagnostic
Medical Device Directive 98/79/EC.
Liability Disclaimer
bioMérieux, Inc. makes no express or implied warranty regarding this manual, its quality, performance, or appropriate use regarding any type of specific procedure.
Furthermore, this manual may be modified by bioMérieux without notice and without implying any obligation or liability on the part of the company.
Intellectual Property
bioMérieux, the blue logo, BacT/ALERT, and MB/BacT are used, pending, and/or registered trademarks of bioMérieux in the USA and other countries.
CLSI is a registered trademark of Clinical and Laboratory Standards Institute, Inc.
PSC and Quickscan are a registered trademarks of PSC, Inc.
Zip is a registered trademark of Iomega Corporation.
No part of this publication may be reproduced, transmitted, transcribed, stored in a retrieval system, or translated into any language (human or computer) in any form, or by any means whatsoever, without the prior express written permission of bioMérieux, Inc.
© 2010 bioMérieux, Inc. All rights reserved.

Warranty
Seller, bioMérieux, Inc., warrants the BacT/ALERT® 3D 60 instrument (the “instrument”) to the original purchaser for a period of one (1) year after date of installation against defects in material and workmanship and defects arising from failure to conform to specifications applicable on the date of installation. Seller further agrees to correct, either by repair, or, at its election, by replacement, any such defect found on examination to have occurred, under normal use and service, during such one (1) year period, provided Seller is promptly notified in writing upon discovery of such defect.
Seller shall not be liable under this Warranty for any defect arising from abuse of the system, failure to operate and maintain the system in accordance with the documentation included with the Instrument, including repair service, alteration or modification of the system by any person other than service personnel of bioMérieux, Inc., or Seller; or use of modified, changed, or previously used disposables.
The Warranty of Seller set forth above and the obligations and liabilities of Seller thereunder are exclusive and in lieu of all other remedies or warranties, express or implied, arising by law or otherwise, with respect to the system delivered hereunder (including without limitation any obligation of Seller with respect to merchantability, fitness for particular purpose, and consequential damages, and whether or not occasioned by Seller’s negligence).
This Warranty shall not be extended or altered except by written instrument signed by Seller.
All of the product elements in the Seller’s Instrument and the total instrument are warranted to be new or equivalent to new for the full product warranty period of one year. Disposables and replacement items with a normal life expectancy of less than one (1) year, such as batteries and bulbs, are excluded from this warranty.

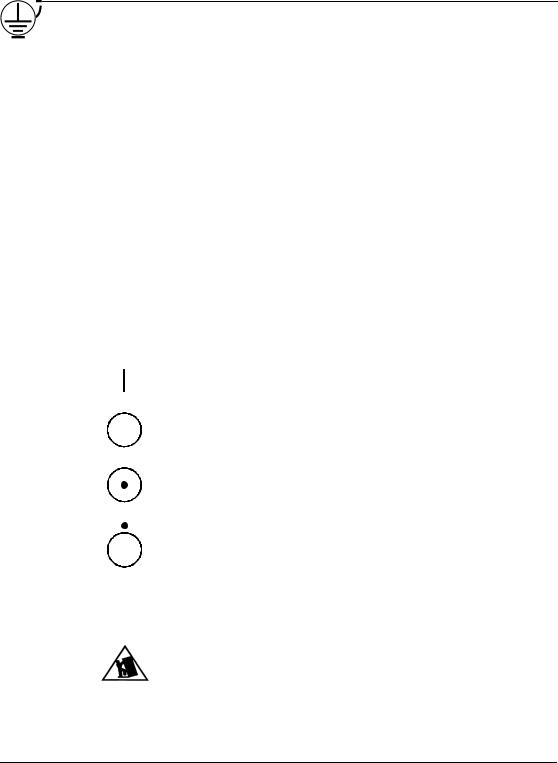
STANDARD SYMBOLS
The following table presents symbols that may appear in the instructions for use or on the instrument, package inserts, or packaging.
CE-Marking of Conformity
Consult Instructions for Use
Use by
Manufacturer
Date of manufacture
Contains sufficient for <n> tests
Keep dry
Fragile, handle with care
Caution, consult accompanying documents
Biological risks
Electric shock warning

Standard Symbols
Radiation warning
Potential pinch-point warning
Laser
Temperature limitation
Upper limit of temperature
Lower limit of temperature
In Vitro Diagnostic Medical Device
Batch code
Authorized Representative in the European Community
Catalog number
Serial Number
Do not reuse
Recyclable
Separate collection for waste electrical and electronic equipment
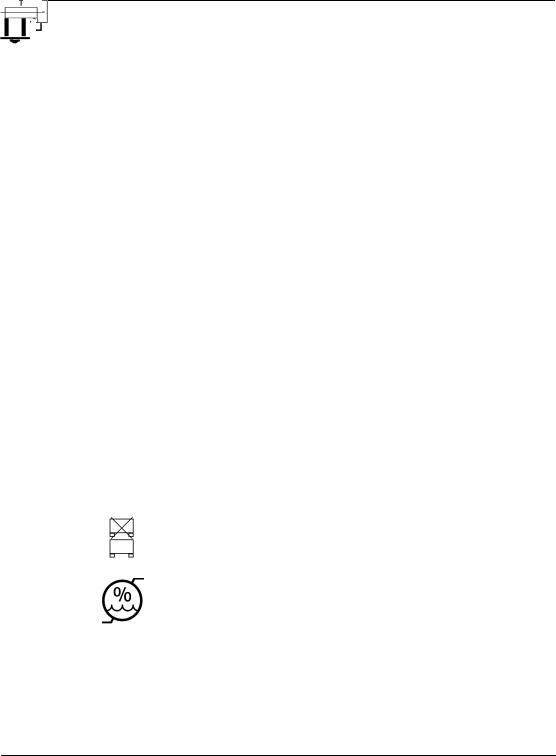
Standard Symbols
Very toxic
Corrosive
Sodium azide
Irritant
Positive control
Negative control
Keep away from sunlight
Protect from light
This way up
Do not stack
Humidity limitation
Fuse
Direct current
Standard Symbols
Alternating current
Both direct and alternating current
Three-phase alternating current
Earth (ground) terminal
Protective conductor terminal
Frame or chassis terminal
Equipotentiality
ON (supply)
OFF (supply)
ON (only for a component of the system equipment)
OFF (only for a component of the system equipment)
Equipment protected throughout by double insulation or reinforced insulation (Equivalent to Class II of IEC 536)
Potential tip over/crush hazard

TABLE OF CONTENTS
Standard Symbols...................................................................................................... |
vi |
List of Figures ............................................................................................................ |
ix |
List of Tables ............................................................................................................ |
xiii |
HOW TO USE THIS MANUAL........................................................................................ |
1-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-1
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-1
Intended Audience . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-2
Purpose of the BacT/ALERT® 3D 60 System . . . . . . . . . . . . . . . . . . . . . . . . . . 1-2
Additional Supplies . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-3
Purpose of This Manual . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-3
Manual Organization. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-4
Chapter Organization . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-5
Finding Topics. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-6
Typographic and Usage Conventions. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-6
Name and Titles. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-6
Click . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-6
Press . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-7
Procedural Steps . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-7
References . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-7
Select. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-7
User Input . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-8
Warnings, Cautions, and Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-8
SYSTEM OVERVIEW ..................................................................................................... |
2-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-1
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-1
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-2
Hardware Configuration. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-2
Software Configuration Options. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-2
21 CFR Part 11 and HIPAA . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-3
Theory Of Operation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-3
Principle of Detection . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-3
Electrical Warnings . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-4
Electrical Grounding . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-6
Electrical and Electronic Recycling . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-7
Fuse Replacement. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-7
BacT/ALERT® 3D 60 Hardware . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-8
BacT/ALERT® 3D 60 User Manual |
i |
95269 |
|

Table of Contents
Instrument . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-8
Monitor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-9 Barcode Reader . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-9 Mouse. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-9 Keyboard . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-9 UPS . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-9 Backup Drive . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-9 UPS Port. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-10 UPS Serial Port. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-10 Monitor Port . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-10 Mouse Ports . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 Keyboard Port. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 Printer Port . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 External Speaker Port. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 Power Connector . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 Power Entry Module . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 Modem Port . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 LIS Port. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 Comm Port . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11
Instrument Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-11
Electrical Power Services Requirements . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-11 Power Consumed in Watts . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Heat Dissipated . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Sound Emission . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Instrument Dimensions. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12
Instrument Environmental Requirements . . . . . . . . . . . . . . . . . . . . . . . . . 2-12
Operating Temperature Range. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Storage Temperature Range . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Operating Humidity Range . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Storage Humidity Range. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Maximum Operating and Storage Altitude . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12 Pollution Degree 2 in accordance with IEC 664 . . . . . . . . . . . . . . . . . . . . . . . .2-12 Overvoltage Category II per IEC 664 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-12
Instrument Installation and Setup . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-13
BacT/ALERT® 3D 60 Software . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-13
Monitor Screens . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-13 Common Screen Elements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-14
Icon. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-14 Button . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-14 Common System Buttons. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-14 Scroll Button . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-15 Slidebar Switch. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-15 Anchor Display/Scroll Buttons . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-15 Text Entry Field. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-16
SYSTEM INSTALLATION................................................................................................ |
3-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-1
ii |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

Table of Contents
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-1 Preparation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-2
Verifying Site Requirements Are Met. . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-2 Verifying Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-2
Installation. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-3
Setting the AC Power . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-3 Choosing Instrument Location . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-6 Making Instrument Connections . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-7 Powering Up the Instrument . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-9 Configuring the Instrument . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-10 Setting the Temperature . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-10 Checking for Errors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-10
Functional Testing. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-10
Modem Functional Test . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-10 Barcode Reader Functional Test . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-11 UPS Functional Test (BacT/ALERT® 3D 60 Only, APC UPS 650) . . . . . 3-12
Installing the Optional Restraint . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-12
BASIC FUNCTIONS....................................................................................................... |
4-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-1 Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-1 Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-2 Monitoring the System . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-2
Main Screen Introduction. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-2
Background Color. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-3 Instrument Icon. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-4 Bottle Count Table . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-4
Viewing Faults . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-5 Viewing the Cell Status Screen . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-6
Understanding the View Cell Status Screen Display . . . . . . . . . . . . . . . . . . . . .4-6
Text/Data Entry . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-8
Common Text Fields and Field Limits . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-8
Using the Barcode Scanner to Enter Data. . . . . . . . . . . . . . . . . . . . . . . . . 4-8 Manually Entering Text into a Data Entry Field (Keyboard). . . . . . . . . . . . 4-9
Loading Bottles . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-10
Loading Bottles . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-10 Changing the Maximum Test Time (Individual Bottles) . . . . . . . . . . . . . . 4-13 Handling Anonymous Bottles. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-14
Unloading Bottles . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-14
Unloading Bottles. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-15 Handling Unconfirmed Positive Bottles (False Positives) . . . . . . . . . . . . 4-17
BacT/ALERT® 3D 60 User Manual |
iii |
95269 |
|

Table of Contents
Accessing the Setup Screen Function Buttons . . . . . . . . . . . . . . . . . . . . . . . . 4-18
Accessing the Setup Screen. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-18
Inactivity Timeout for all Setup Screens . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-19
Setup Screen Function Buttons . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-19
Viewing and Printing . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-21
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-21
Viewing Bottle Data . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-21
Viewing/Printing Reports . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-21
Viewing/Printing Graphs . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-21
Using the Print Screen Function. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-21
Viewing and Printing Test Data. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-22
Viewing and Printing Bottle Graphs . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-29
Display Bottle Readings . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-31
Sending/Requesting LIS Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-36
Sending Results to the LIS . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-36
Requesting Information from the LIS . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-36
EDITING TEST DATA .................................................................................................... |
5-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-1 Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-1 Viewing/Editing Bottle Data . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-2
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-2 Selecting Bottles Using the Edit Cell Contents Button . . . . . . . . . . . . . . . . 5-3 Selecting Bottles Using the Select Bottle to Edit/Graph Button . . . . . . . . . 5-4 Editing Bottle Details Using the Edit Bottle Detail Screen. . . . . . . . . . . . . . 5-5
Edit Bottle ID Field . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-6 View Accession Number Field . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-6 View Hospital ID Field. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-6 View Patient First Name Field . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-6 View Patient Last Name Field. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-6 Edit Load Status Slidebar Switch . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-6 Edit Maximum Test Time Scroll Buttons. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-7 Edit Bottle Type Scroll Button. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-7 View Cell Location Icon . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-7 View First Loaded Time Icon . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-8 View Last Unloaded Time Icon. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-8 View Time of Last Bottle Reading Icon. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-8 View Test Time Icon . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-8 View Test Result Icon . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-8 Edit Test Result Button. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-9 Graph Bottle Readings Button . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-10 View Algorithm/Polynomial Icon . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-10 View How Determined/Positivity Index Icon . . . . . . . . . . . . . . . . . . . . . . . . . . .5-10
Editing Data Relationships . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-11
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-11
iv |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

Table of Contents
Initiating the Edit Data Relationships Function . . . . . . . . . . . . . . . . . . . . 5-12
Editing Bottle ID to Accession Number Relationships . . . . . . . . . . . . . . . 5-13
Attaching Bottle IDs Without an Accession Number to an Accession Number 5-14
Moving a Bottle ID Association from one Accession Number to Another. . . . .5-14
Editing Accession Number to Hospital ID Relationships . . . . . . . . . . . . . 5-15
Attaching Accession Numbers Without a Hospital ID to a Hospital ID . . . . . . .5-16
Moving an Accession Number Association from one Hospital ID to Another . .5-16
Editing Hospital ID to Patient Name Relationships . . . . . . . . . . . . . . . . . 5-17
SOFTWARE CONFIGURATION........................................................................................ |
6-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-1
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-1
Setting the Maximum Test Time . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-2
Setting the Audible Alarms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-3
Priority of Alarms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-5
Terminating an Instrument Alarm . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-5
Changing the System Password. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-5
Initiating Manual Backup. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-7
Configuring Report Screens . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-9
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-9
Entering Report Labels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-9
Configuring Report Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-13
Load Report Configuration Screen . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-16
Status Report Configuration Screen. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-17
Unload Report Configuration Screen . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-18
Viewing and Printing Calibration Data . . . . . . . . . . . . . . . . . . . . . . . . . . . 6-19
Viewing and Printing Calibration History . . . . . . . . . . . . . . . . . . . . . . . . . 6-22
SYSTEM MAINTENANCE ............................................................................................... |
7-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-1 Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-1 Hardware Maintenance. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-2
Preventative Maintenance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-2 Safety Precautions and Procedures . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-2
Spill Cleanup . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-3 Disinfection Procedure for Spills Onto the Instrument . . . . . . . . . . . . . . . . . . . .7-3 Disinfection Procedure for Spills Within the Instrument . . . . . . . . . . . . . . . . . . .7-4
Using the Keyboard in Place of the Mouse . . . . . . . . . . . . . . . . . . . . . . . . 7-6 UPS On/Off Button Location . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-7 Instrument Reboot/Shutdown . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-7
Shutdown Method 1 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-8 Shutdown Method 2 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-8 Shutdown Method 3 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-9
BacT/ALERT® 3D 60 User Manual |
v |
95269 |
|

Table of Contents
Shutdown Method 4 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-9
Instrument Startup . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-9
Software Maintenance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-9
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-9 Restarting the Incubation Chamber . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-10 Setting and Formatting the System Date and Time . . . . . . . . . . . . . . . . . 7-11 Enabling and Disabling Racks and Cells . . . . . . . . . . . . . . . . . . . . . . . . . 7-13
Relocating Bottles. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-14
Adjusting an Instrument’s Temperature . . . . . . . . . . . . . . . . . . . . . . . . . . 7-15
Checking an Instrument's Temperature . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-15 Setting the Optimal Temperature for an Instrument . . . . . . . . . . . . . . . . . . . . .7-17 Calibrating an Instrument's Temperature . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-17
Calibrating an Instrument Cell. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-18
Locating a Cell Which Failed Calibration . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-18 Viewing a Cell's Readings and/or Calibrating a Cell. . . . . . . . . . . . . . . . . . . . .7-19
Viewing Incubation Chamber Information . . . . . . . . . . . . . . . . . . . . . . . . . 7-22
SYSTEM TROUBLESHOOTING ....................................................................................... |
8-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-1
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-1
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-2
Fault Codes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-3
Instrument Fault Codes. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-3
Instrument Status Codes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-16
Operator Error Codes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-18
Bottle Problems. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-29
User Output Device Problems . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-29
21 CFR PART 11 MODE.............................................................................................. |
9-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-1
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-1
Log In/Out of System — 21 CFR Part 11 Mode. . . . . . . . . . . . . . . . . . . . . . . . . 9-2
Logging In for the First Time . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-2
Logging In . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-6
Inactivity Timeout . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-7
Login Errors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-7
Change Password. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-8
Change Password Errors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-9
Logging Out . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-12
Configuring Users — 21 CFR Part 11 Mode . . . . . . . . . . . . . . . . . . . . . . . . . . 9-12
Adding a User . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-13
Deleting a User . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-15
vi |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

Table of Contents
Deleting More Than One User . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .9-16
Clearing a User Password . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-16
Clearing More Than One User Password. . . . . . . . . . . . . . . . . . . . . . . . .9-17
Audit Trail . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-18
Accessing the Audit Trail . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9-20
MYCOBACTERIAL TESTING......................................................................................... |
10-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-1 Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-1 System Description . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-2
Intended Use . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-2
Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-2 BacT/ALERT® 3D 60 Instrument . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-3
Circulation Fan . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-3 Installation Procedures and Special Requirements . . . . . . . . . . . . . . . . . . . . .10-3
BacT/ALERT® 3D 60 Software . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-4
Barcodes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-4 Set Maximum Test Time. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-4 Positive Detection Algorithm. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-4 Bottle Status . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-4
System Startup . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-5
General Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-5
MB Configuration . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-5 Limitations of the Test . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-5 Service and Maintenance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-6
Theory of Operation. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-6
Principle of Detection . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-6
Safety Features . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10-7
General Precautions. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-8 Spill Cleanup . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .10-8 Disinfection Procedure for Spills Within/Onto the Instrument. . . . . . . . . . . . . .10-8
UNPACKING INSTRUCTIONS.......................................................................................... |
A-1 |
BacT/ALERT® 3D 60 Unpacking Instructions . . . . . . . . . . . . . . . . . . . . . . . . . . |
A-1 |
PART CHECKLIST ........................................................................................................ |
B-1 |
BacT/ALERT® 3D 60 Part Checklist . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . B-1
Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . B-1
Optional Equipment . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . B-1
Recommended Tools. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . B-1
INSTALLATION CHECKLIST ........................................................................................... |
C-1 |
BacT/ALERT® 3D 60 Installation Checklist. . . . . . . . . . . . . . . . . . . . . . . . . . . . C-1
BacT/ALERT® 3D 60 User Manual |
vii |
95269 |
|

Table of Contents
INTERNATIONAL CHARACTER ENTRY............................................................................ |
D-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .D-1
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .D-1
International Character Entry . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .D-2
Entering International Characters . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .D-3
BOTTLE QUALITY CONTROL......................................................................................... |
E-1 |
About This Chapter . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .E-1
Chapter Contents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .E-1
BacT/ALERT® Culture Bottles . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .E-2
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .E-2
Flip Cap . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . E-3
Stopper/Seal. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . E-3
Bottle. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . E-3
Volume Designations . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . E-3
Barcode . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . E-3
Sensor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . E-3
Limitations of the Test. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . E-4
Quality Control of Growth Performance . . . . . . . . . . . . . . . . . . . . . . . . . . .E-4
Blood Culture Bottles (SA, SN, FA, FN, and PF) . . . . . . . . . . . . . . . . . . . . . . . E-4
Mycobacteria Culture Bottles (MB and MP) . . . . . . . . . . . . . . . . . . . . . . . . . . . E-5
GLOSSARY................................................................................................... |
GLOSSARY-1 |
INDEX .................................................................................................................. |
INDEX-1 |
viii |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

LIST OF FIGURES
Figure 1-1: |
Procedure Icon ................................................................................................... |
1-7 |
Figure 2-1: |
Electrical Grounding Requirements ................................................................... |
2-7 |
Figure 2-2: |
Instrument Front View ........................................................................................ |
2-8 |
Figure 2-3: |
Instrument Rear View ...................................................................................... |
2-10 |
Figure 2-4: |
Button Examples .............................................................................................. |
2-14 |
Figure 2-5: |
Scroll Button ..................................................................................................... |
2-15 |
Figure 2-6: |
Slidebar Switch ................................................................................................ |
2-15 |
Figure 2-7: |
Anchor Display/Scroll Buttons .......................................................................... |
2-16 |
Figure 3-1: |
Power Entry Module with Fuse Holder Removed .............................................. |
3-3 |
Figure 3-2: |
PEM with 115 VAC Version ............................................................................... |
3-4 |
Figure 3-3: |
PEM with 230 VAC Version ............................................................................... |
3-5 |
Figure 3-4: |
Additional Fuse Holder Views ............................................................................ |
3-6 |
Figure 3-5: |
Installation and Setup Diagram — Port and Power Connections ....................... |
3-9 |
Figure 3-6: |
Modem Configuration and Dip Switch Settings ................................................ |
3-11 |
Figure 3-7: |
Countertop/Surface Mounting Diagram ........................................................... |
3-13 |
Figure 3-8: |
Mounting Surface Diagram .............................................................................. |
3-13 |
Figure 3-9: |
Mounting Surface (Drilling) Template ............................................................... |
3-14 |
Figure 4-1: |
Main Screen ....................................................................................................... |
4-3 |
Figure 4-2: |
Instrument Icons ................................................................................................ |
4-4 |
Figure 4-3: |
Bottle Count Table/Unload Buttons .................................................................... |
4-5 |
Figure 4-4: |
View Cell Status Screen .................................................................................... |
4-6 |
Figure 4-5: |
Disabled Cell ...................................................................................................... |
4-7 |
Figure 4-6: |
Main Screen — Load Mode ............................................................................. |
4-10 |
Figure 4-7: |
Change Maximum Test Time Screen ............................................................... |
4-13 |
Figure 4-8: |
Main Screen — Unload Mode .......................................................................... |
4-15 |
Figure 4-9: |
Setup Screen ................................................................................................... |
4-18 |
Figure 4-10: Report Selection Screen .................................................................................. |
4-22 |
|
Figure 4-11: Sample Report Screen ..................................................................................... |
4-24 |
|
Figure 4-12: Find Text Screen .............................................................................................. |
4-27 |
|
Figure 4-13: Save to File Screen .......................................................................................... |
4-28 |
|
Figure 4-14: Select Bottle to Edit/Graph Screen .................................................................. |
4-29 |
|
Figure 4-15: Graph Bottle Readings Screen ........................................................................ |
4-30 |
|
BacT/ALERT® 3D 60 User Manual |
ix |
95269 |
|

Figure 4-16: |
Bottle Readings Screen .................................................................................. |
4-32 |
Figure 4-17: |
Find Text Screen ............................................................................................. |
4-34 |
Figure 4-18: |
Save to File Screen ......................................................................................... |
4-35 |
Figure 5-1: |
Edit Cell Contents Screen ................................................................................. |
5-3 |
Figure 5-2: |
Select Bottle to Edit/Graph Screen ................................................................... |
5-4 |
Figure 5-3: |
Edit Bottle Detail Screen ................................................................................... |
5-5 |
Figure 5-4: |
Edit Test Result Screen .................................................................................... |
5-9 |
Figure 5-5: |
Edit Data Relationships Screen ...................................................................... |
5-12 |
Figure 5-6: |
Edit Bottle ID to Accession Number Relationships Screen ............................. |
5-13 |
Figure 5-7: |
Edit Accession Number to Hospital ID Relationships Screen ......................... |
5-15 |
Figure 5-8: |
Edit Hospital ID to Patient Name Relationships Screen .................................. |
5-17 |
Figure 6-1: |
Set Maximum Test Time Screen ....................................................................... |
6-2 |
Figure 6-2: |
Set Audible Alarm Options Screen .................................................................... |
6-4 |
Figure 6-3: |
Change Password Screen ................................................................................ |
6-6 |
Figure 6-4: |
Padlock Icon (Full Open Position) ..................................................................... |
6-6 |
Figure 6-5: |
Padlock Icon (Half Open Position) .................................................................... |
6-7 |
Figure 6-6: |
Padlock Icon (Closed Position) ......................................................................... |
6-7 |
Figure 6-7: |
Backup Management Screen ............................................................................ |
6-8 |
Figure 6-8: |
Backup in Progress Icon ................................................................................... |
6-8 |
Figure 6-9: |
Report Selection Screen ................................................................................. |
6-10 |
Figure 6-10: |
Report Label Entry Screen .............................................................................. |
6-11 |
Figure 6-11: |
Report Selection Screen ................................................................................. |
6-14 |
Figure 6-12: |
Load Report Configuration Screen .................................................................. |
6-16 |
Figure 6-13: |
Status Report Configuration Screen ............................................................... |
6-17 |
Figure 6-14: |
Unload Report Configuration Screen .............................................................. |
6-18 |
Figure 6-15: |
Report Selection Screen ................................................................................. |
6-19 |
Figure 6-16: |
Cell Calibration Report Screen ........................................................................ |
6-20 |
Figure 6-17: |
View Window ................................................................................................... |
6-21 |
Figure 6-18: |
Report Selection Screen ................................................................................. |
6-22 |
Figure 6-19: |
Calibration History Screen .............................................................................. |
6-23 |
Figure 7-1: |
View Cell Status Screen .................................................................................. |
7-11 |
Figure 7-2: |
Set Date/Time Screen ..................................................................................... |
7-12 |
Figure 7-3: |
Enable/Disable Rack or Cell Screen ............................................................... |
7-13 |
Figure 7-4: |
Reference Thermometer ................................................................................. |
7-15 |
|
|
|
x |
BacT/ALERT® 3D 60 User Manual |
|
|
|
95269 |

Figure 7-5: |
Calibrate Instrument Temperature Screen ....................................................... |
7-16 |
Figure 7-6: |
Calibrate Cell Screen ....................................................................................... |
7-19 |
Figure 7-7: |
View Incubation Chamber Information Screen ................................................ |
7-23 |
Figure 8-1: |
Instrument Icon with Fault Codes ...................................................................... |
8-3 |
Figure 8-2: |
Instrument Status Code 710 ............................................................................ |
8-16 |
Figure 8-3: |
Operator Error Code 911 ................................................................................. |
8-18 |
Figure 8-4: |
QuickScan® 6000 Reset Barcode .................................................................... |
8-30 |
Figure 8-5: |
Honeywell Reset Barcode ................................................................................ |
8-30 |
Figure 8-6: |
Informational Warning Screen ......................................................................... |
8-31 |
Figure 9-1: |
Main Screen While Logged Out —21 CFR Part 11 Mode .................................. |
9-2 |
Figure 9-2: |
User Login Screen ............................................................................................. |
9-3 |
Figure 9-3: |
User Login Screen with Change Password Fields ............................................. |
9-4 |
Figure 9-4: |
Main Screen While Logged In — 21 CFR Part 11 Mode ................................... |
9-5 |
Figure 9-5: |
User Login Screen ............................................................................................. |
9-6 |
Figure 9-6: |
User Login Screen with Wrong Password Alert ................................................. |
9-8 |
Figure 9-7: |
User Login Screen with Change Password Fields ............................................. |
9-9 |
Figure 9-8: |
User Login Screen with Change Password Error ............................................. |
9-10 |
Figure 9-9: |
User Login Screen with Wrong Password Alert ............................................... |
9-11 |
Figure 9-10: User Configuration Screen ............................................................................... |
9-13 |
|
Figure 9-11: Add User Screen .............................................................................................. |
9-14 |
|
Figure 9-12: Delete User Screen .......................................................................................... |
9-15 |
|
Figure 9-13: Clear Password Screen ................................................................................... |
9-17 |
|
Figure 10-1: Instrument Icon for MB-Configured System ..................................................... |
10-3 |
|
Figure 10-2: Bottle Count Table and Unload Buttons ........................................................... |
10-4 |
|
Figure 10-3: Metabolic Pathway for Mycobacterial CO2 ...................................................... |
10-7 |
|
Figure E-1: |
Typical BacT/ALERT® Culture Bottle ................................................................ |
E-2 |
BacT/ALERT® 3D 60 User Manual |
xi |
95269 |
|

xii |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

LIST OF TABLES
Table 2-1: |
Common System Buttons ................................................................................ |
2-14 |
Table 3-1: Facility Power Rating and Conversion Chart ..................................................... |
3-4 |
|
Table 3-2: |
Restraint Hardware .......................................................................................... |
3-12 |
Table 3-3: |
Table of Equivalent Dimensions (Inches to Centimeters) ................................ |
3-14 |
Table 5-1: |
Bottle Specific Algorithms ................................................................................ |
5-10 |
Table 5-2: |
Status Determination Codes ............................................................................ |
5-10 |
Table 6-1: Report Field Descriptions ................................................................................. |
6-12 |
|
Table 9-1: |
Audit Trail Events ............................................................................................. |
9-18 |
Table D-1: |
International Character Table ........................................................................... |
D-2 |
BacT/ALERT® 3D 60 User Manual |
xiii |
95269 |
|

xiv |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

|
|
HOW TO USE THIS MANUAL |
1 |
|
|
|
|
About This Chapter
This chapter gives you important information about the
BacT/ALERT® 3D 60 system and how to use this manual. We recommend that you read this chapter first.
IMPORTANT: Read this manual carefully before you attempt to operate the BacT/ALERT® 3D 60 system.
Chapter Contents
Intended Audience • 1-2
Purpose of the BacT/ALERT® 3D 60 System • 1-2
Additional Supplies • 1-3
Purpose of This Manual • 1-3
Manual Organization • 1-4
Chapter Organization • 1-5
Finding Topics • 1-6
Typographic and Usage Conventions • 1-6
Name and Titles • 1-6
Click • 1-6
Press • 1-7
Procedural Steps • 1-7
References • 1-7
Select • 1-7
User Input • 1-8
Warnings, Cautions, and Information • 1-8
BacT/ALERT® 3D 60 User Manual |
1-1 |
95269 |
|

How To Use This Manual |
Intended Audience |
Intended Audience
The BacT/ALERT® 3D 60 system and this manual are intended for laboratory use by trained, professional users.
Purpose of the BacT/ALERT® 3D 60 System
The BacT/ALERT® 3D 60 Microbial Detection System is a totally automated test system capable of incubating, agitating, and continuously monitoring aerobic and anaerobic media inoculated with patient specimens suspected of having bacteremia, fungemia, and/or mycobacteremia.
CAUTION: bioMérieux shall not be liable as to any defect arising from abuse of the instrument, failure to operate and maintain the instrument in accordance with the User Manual, operation of the instrument by a person who has not been trained in its operation by bioMérieux, repair, service, alteration or modification of the instrument by any person other than service personnel of bioMérieux, or modification, change or reuse of the disposables supplied by bioMérieux for use in the instrument.
CAUTION: This BacT/ALERT® 3D User Manual is only intended for use with B.30 Software or higher. The software version B.30 (or higher) is displayed at the bottom of the instrument icon on the Main screen.
CAUTION: All figures depicting monitor screens are examples only. Actual screens may differ to the extent they are affected by the actual data entered by the user, or actual data transmitted to the instrument over the LIS interface, or actual data generated by the instrument.
1-2 |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

Additional Supplies |
How To Use This Manual |
CAUTION: Regarding section "Entering Report Labels," the user is solely responsible for the choice of customized report label text and for validating that the intended label text appears in all associated reports. bioMérieux shall not be liable for any consequences resulting from misinterpretation of customized report labels.
Additional Supplies
Contact bioMérieux or your local vendor for laboratory supplies and accessories.
Purpose of This Manual
This manual focuses on the BacT/ALERT® 3D 60 software application and how you use it in your workflow. It contains step-by-step procedures for using your BacT/ALERT® 3D 60 system.
By using these procedures, you can perform all the functions required to operate your system, including:
•accessing the BacT/ALERT® 3D 60 software
•system monitoring
•entering order information (where applicable)
•loading and unloading bottles
•viewing and printing data
•LIS interaction with the BacT/ALERT® 3D 60 SelectLink
•accessing the Setup screen
Note: Screens and figures are intended for illustrative purposes only and are not to be construed as representations of actual test data, results, or components. Screens and components are not shown to scale.
BacT/ALERT® 3D 60 User Manual |
1-3 |
95269 |
|

How To Use This Manual |
Manual Organization |
Manual Organization
This manual is organized by chapters. Chapters are organized according to the order of menu commands in the software application.
Chapter 1, How To Use This Manual — Provides an introduction to the BacT/ALERT® 3D 60 system and how to use this manual. It is recommended that you read this chapter first.
Chapter 2, System Overview — This chapter gives you a complete description of the different BacT/ALERT® 3D 60 hardware and software configurations available. Also, the monitor screens are listed along with a description of common screen elements.
Chapter 3, System Installation — Provides detailed instructions on how to install the instrument and perform functional tests.
Chapter 4, Basic Functions — Introduces you to the Main screen and shows you how to perform basic functions (ex. enter data, view faults, view the Cell Status screen, load/unload bottles, etc.). This chapter also introduces you to the Setup screen and the associated function buttons.
Chapter 5, Editing Test Data — Explains how to access bottle data for loaded and unloaded bottles using the Edit Bottle Detail screen. This chapter shows you how to view and edit bottle data.
Chapter 6, Software Configuration — Explains how to configure the software for your specific needs. This chapter shows you how to set the maximum test time, set the audible alarms, change the system password, initiate a manual backup, configure report screens, and view and print calibration data.
Chapter 7, System Maintenance — This chapter provides you with procedures on how to perform maintenance on the BacT/ALERT® 3D 60 hardware and software.
Chapter 8, System Troubleshooting — Describes the different types of instrument fault, instrument status and operator error codes, as well as bottle and user output device problems, you may encounter when using the instrument. Cause(s) and solutions(s) for each type of fault/error/problem are also listed.
Chapter 9, 21 CFR Part 11 Mode — This chapter explains how to log in and out of the instrument while in 21 CFR Part 11 mode, as well as instructions for configuring users (ex. adding or deleting a user, or clearing a user password).
1-4 |
BacT/ALERT® 3D 60 User Manual |
|
95269 |

Chapter Organization |
How To Use This Manual |
This chapter also describes the events recorded in the audit trail and how to retrieve the audit trail.
Chapter 10, Mycobacterial Testing — This chapter provides you with a complete description of the of the BacT/ALERT® 3D 60 instrument when it is configured for Mycobacterial (MB) functions.
Appendix A, BacT/ALERT® 3D 60 Unpacking Instructions — Contains a diagram illustrating how to unpack the instrument.
Appendix B, BacT/ALERT® 3D 60 Part Checklist — To use when verifying and inspecting kit contents.
Appendix C, BacT/ALERT® 3D 60 Installation Checklist — To use when installing the instrument.
Appendix D, International Character Entry — Explains how to enter international characters on the BacT/ALERT® 3D 60 instrument.
Appendix E, Bottle Quality Control — This chapter provides you with a description of the BacT/ALERT® 3D 60 culture bottle along with a quality control procedure.
Glossary — An alphabetized list of frequently used terms along with a definition for each term.
Chapter Organization
All chapters include the following:
•About This Chapter — Brief description of the chapter’s content and purpose.
•Chapter Contents — A table of contents for the chapter.
•Descriptions and/or Procedures — Chapters contain descriptions and procedures appropriate to their subject matter. See the Manual Organization section in this chapter for more information.
•Background Information, where applicable and useful.
BacT/ALERT® 3D 60 User Manual |
1-5 |
95269 |
|

How To Use This Manual |
Finding Topics |
Finding Topics
This manual uses several methods to help you find information and keep your bearings.
Table of Contents — Located at the front of the manual. It contains the titles of all chapters/appendices and their sections, and the page number of each title and section.
List of Figures — Located at the front of the manual. It contains a list of all figures in the manual and the page number of each figure.
List of Tables — Located at the front of the manual. It contains a list of all tables in the manual and the page number of each table.
Chapter Contents — Located at the front of each chapter. It lists all sections in the chapter and their page numbers.
Page Headers — Located at the top of each page. There are two parts to a header: the chapter title and the primary section title.
Page Footers — Located at the bottom of each page. There are three parts to a footer: the manual’s title, the chapter’s part number, and the page number.
Index — Located at the back of the manual. It contains topical entries and their page numbers.
Typographic and Usage Conventions
Name and Titles
Button, icon and field names are in Proper Case, bold.
Example: Click the Select Maximum Test Time button.
The names of windows and screens are in Proper Case, but are not bolded.
Example: The Set Maximum Test Time screen...
Click
This manual uses the word “click” to refer to using a mouse to choose or select a text entry field, button, or option.
Example: Click OK.
1-6 |
BacT/ALERT® 3D 60 User Manual |
|
95269 |
 Loading...
Loading...