Page 1
Order no.: DD+DIS084.97E
*11NL351*
1 piece 1NL35 MA 1
1. Edition
Technical
Documentation
DRYSTAR 3000
Type 5361
printed in Germany
Page 2

Overview
0. Order list
1. Pre-installation
2. Controls and Connectors
3. Installation
4. Operating instruction s
5. Theory of operation
6. Service and maintenance
7. Reference and Circuit Diagrams
8. Spare Parts List
9. Options
10. Modifications
11. Technical Modifications
12. Maintenance
13. Fieldservice Bulletins
14. Installation Planning
15. Glossary
Page 3

HEALTHCARE
Imaging Services
Document No: DD+DIS238.06E
► Purpose of this Document
Generic Safety Directions
for HealthCare Imaging Products
This Generic Safety Directions document comprises the general safety relevant information including
relevant environmental and occupational safety instructions for the Service Engineer.
It is valid for all Agfa HealthCare Imaging Products and part of each Service Documentation as well as
Installation Planning document.
The latest version is available via MedNet, GSO Library path:
General Info => Agfa HealthCare => Publications => Service Manual
Generic Safety Directions
► Document History
Edition.
Revision
1.3 07-2009
Release
Date
Changes
compared to previous Revision 1.2:
• Updated table with laser classification to latest changes of the
corresponding standard. See section 3.3.
• Added section Environmental and occupational Safety Instructions.
See section 9.
• Added safety note concerning inroom installations of CR equipment
and corresponding X-ray shielding. See section 17.
• Added laser safety note and safety note concerning electrical checks
after repairs. See section 19.
• Added treatment for Lithium batteries in sections 19 and 21.
• Updated information concerning the recycling pass. See section 24.
► Referenced Documents
Document Title
Not applicable Not applicable
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3
07-2009 printed in Germany
Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Document Node ID: 11849633
eq_generic_safety_directions_e_template_v01
Copyright © 2009 Agfa HealthCare N.V.
Page 4

DD+DIS238.06E
► Manufacturer
Agfa HealthCare N.V.
Publisher
Agfa-Gevaert HealthCare GmbH
Tegernseer Landstraße 161
D - 81539 München
Germany
Copyright © 2009 Agfa HealthCare N.V.
All rights reserved.
Technical modifications reserved.
AGFA and the Agfa-Rhombus are trademarks of Agfa HealthCare N.V.
Generic Safety Directions
WARNING:
Improper operation or service activities may cause damage or injuries.
INSTRUCTION:
(1) Read the "Generic Safety Directions" document
(see MEDNET GSO => General Info => Agfa HealthCare => Publications =>
Service Manual) prior to attempting any operation, repair or maintenance task on the
equipment.
(2) Strictly observe all safety directions within the "Generic Safety Directions" and on
the product.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 2 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 5

DD+DIS238.06E
LIST OF CONTENTS
1
DISCLAIMER......................................................................................................... 5
2 USED ICONS ........................................................................................................ 6
3 LABELS .................................................................................................................7
4 PRODUCT COMPLAINTS .................................................................................. 10
Generic Safety Directions
3.1 CE Mark................................................................................................... 7
3.2 System Labels ......................................................................................... 7
3.3 System Labels concerning Laser Radiation ............................................ 8
5 REFERENCES .................................................................................................... 10
6 INTENDED USE.................................................................................................. 11
7 INTENDED USER ............................................................................................... 11
8 QUALIFICATIONS FOR OPERATION AND SERVICE TASKS.......................... 11
9 ENVIRONMENTAL AND OCCUPATIONAL SAFETY INSTRUCTIONS............. 12
10 CONNECTIONS TO OTHER EQUIPMENT ........................................................ 13
11 ACCESSORIES AND SPARE PARTS................................................................ 14
12 COMPLIANCE..................................................................................................... 14
13 SAFETY DIRECTIONS FOR OPERATION......................................................... 17
14 RADIATION PROTECTION ................................................................................ 18
15 SAFETY DIRECTIONS FOR CLEANING AND DISINFECTION......................... 18
16 GENERAL SAFETY DIRECTIONS FOR SERVICE ACTIVITIES ....................... 19
17 SAFETY DIRECTIONS FOR INSTALLATION PLANNING ACTIVITIES ............ 20
18 SAFETY DIRECTIONS FOR INSTALLATION ACTIVITIES................................ 22
19 SAFETY DIRECTIONS FOR MAINTENANCE AND REPAIR ACTIVITIES ........ 23
20 SAFETY DIRECTIONS FOR REMOTE SERVICE ACTIVITIES ......................... 26
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 3 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 6

DD+DIS238.06E
21 SAFETY DIRECTIONS FOR TRANSPORT AND SHIPMENT OF SPARE
Generic Safety Directions
PARTS, ACCESSORIES AND DEVICES............................................................27
22
SAFETY DIRECTIONS CONCERNING MODIFICATIONS.................................27
23 SAFETY DIRECTIONS CONCERNING HAZARDOUS MATERIALS .................27
24 RECYCLING ........................................................................................................27
25 WASTE DISPOSAL .............................................................................................28
26 ERASING PROTECTED HEALTH INFORMATION (PHI) ...................................28
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 4 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 7

DD+DIS238.06E
1 Disclaimer
The installation and service of equipment described herein is to be performed by qualified
personnel who are employed by Agfa HealthCare or one of its affiliates or who are
otherwise authorized by Agfa HealthCare or one of its affiliates to provide such services.
Fitters, engineers and other persons who are not employed by or otherwise directly
affiliated with or authorized by Agfa HealthCare or one of its affiliates are directed to
contact one of the local offices of Agfa HealthCare or one of its affiliates before attempting
installation or service procedures.
No part of this document may be reproduced, copied, adapted or transmitted in any form
or by any means without the written permission of Agfa HealthCare.
Agfa HealthCare makes no warranties or representation, expressed or implied, with
respect to the accuracy, completeness or usefulness of the information contained in this
document and specifically disclaims warranties of suitability for any particular purpose.
Agfa HealthCare shall under no circumstances be liable for any damage arising from the
use or inability to use any information, apparatus, method or process disclosed in this
document.
Agfa HealthCare is not liable for resulting consequences, damages or injuries if you don’t
operate the product correctly or if you don’t have it serviced correctly.
Agfa HealthCare reserves the right to change the product, the characteristics and its
documentation without further notice to improve reliability, function or design.
NOTE:
In the United States, Federal Law stipulates that medical devices should only be sold to,
distributed and used by or by order of a licensed physician.
Generic Safety Directions
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 5 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 8

DD+DIS238.06E
2 Used Icons
Icon Name and Circumstances
Generic Safety Directions
CAUTION: Possible dangerous situation: Light injuries or
damage to the equipment described in the manual and/or
damage to any other equipment or goods and/or environmental
pollution can be the consequence.
WARNING: Dangerous situation: Potential serious injury to a
user, engineer, patient or any other person and possible
mistreatment of patients can be the consequence.
DANGER: Direct, immediate danger: Death or heavy injuries
can be the consequence.
INSTRUCTION:
If used in combination
with the warning or caution sign: Indicates
a specific instruction, which if followed exactly, avoids the
subject of the warning or caution.
If used without warning or caution sign: Indicates an instruction
where it is important to follow literally as described.
IMPORTANT:
Highlights very important actions which have to be carried out to
prevent malfunction.
NOTE:
Indicates advice to facilitate the following step or action.
Highlights unusual points.
Indicates background information.
Can be used to explain or highlight displays of the
graphical user interface.
Is additional information without influence on the action or step!
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 6 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 9

_
DD+DIS238.06E
3 Labels
Generic Safety Directions
3.1
3.2
CE Mark
CE Mark
This product carries the CE Mark. The CE Declaration (CE Conformity)
becomes invalid if the product is changed without explicit consent of the
manufacturer! This applies to all parts, not only to safety elements.
System Labels
All system labels and software version number locations are referred to within this service
document in the appropriate section.
Enclosed an overview of common labels, according to ISO 3864.
This list is not complete.
Hot Surface
Obstacles
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Laser Beam
Corrosive Liquid
DOCUMENT CONTROL NOTE:
Magnetic Field
High Voltage
Ionizing Radiation
Hand Injuries
C&W
005.cdr
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 7 of 28
07-2009 Agfa Company Confidential
Page 10

DD+DIS238.06E
Generic Safety Directions
3.3
System Labels concerning Laser Radiation
According to its classification, laser radiation can lead to eye and skin injuries.
Each laser source is classified from class 1 to class 4, based on standard
DIN EN 60825-1:2007.
The table below lists the meaning of the different laser classes. Note the detailed
instructions in the user manual and technical documentation.
Class # Meaning Example Label
Class 1:
Class 1 M:
Not dangerous to the human eye, even
when using optical instruments. Can
nevertheless produce irritating effects,
especially with low ambient light
conditions.
Not dangerous to the human eye if no
optical instruments (magnifying glass or
binocular) are used.
Can nevertheless produce irritating
effects, especially with low ambient light
conditions.
CLASS 1 LASER PRODUCT
LASER RADIATION
DO NOT VIEW DIRECTLY WITH
OPTICAL INSTRUMENTS
CLASS 1M LASER PRODUCT
Class 2:
Dangerous to the human eye for
intentional staring into the beam.
Not dangerous for short term exposure
< 0,25 seconds.
Using optical instruments does not
increase the risk of eye injury.
LASER RADIATION
Can even for short term exposure < 0,25
seconds produce dazzling and irritating
effects, especially with low ambient light
DO NOT STARE INTO BEAM
CLASS 2 LASER PRODUCT
conditions.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
DOCUMENT CONTROL NOTE:
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 8 of 28
07-2009 Agfa Company Confidential
Page 11

DD+DIS238.06E
Class # Meaning Example Label
Generic Safety Directions
Class 2M:
Class 3R:
Dangerous to the human eye when
staring into the beam or when using
optical instruments (magnifying glass or
telescope). No hazard for short term
exposure < 0,25 seconds (aversion
response of the eye) without use of
optical instruments. Can produce
dazzling and irritating effects even for
short term exposure < 0,25 seconds,
especially at low ambient light conditions.
Possibly dangerous to the human eye for
direct view into the beam.
Risks of an eye injury is increasing with
duration of exposure.
Can produce dazzling and irritating
effects, especially with low ambient light
conditions.
LASER RADIATION
DO NOT STARE INTO THE BEAM
OR VIEW DIRECTLY WITH
OPTICAL INSTRUMENTS
CLASS 2M LASER PRODUCT
LASER RADIATION
AVOID DIRECT EYE EXPOSURE
CLASS 3R LASER PRODUCT
Class 3B:
Class 4:
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Normally dangerous to the human eye
for direct view into the beam.
Viewing diffuse reflections is normally not
dangerous.
Risk of small skin injuries or ignition of
explosive material if the power of the
laser beam is close to the upper limits of
class 3 B.
Dangerous to the human eye for direct
view into the beam or viewing diffuse
reflections.
Very often class 4 lasers also implicate a
fire hazard.
DOCUMENT CONTROL NOTE:
LASER RADIATION
AVOID EXPOSURE TO BEAM
CLASS 3B LASER PRODUCT
LASER RADIATION
AVOID EYE OR SKIN EXPOSURE
TO DIRECT OR SCATTERED
RADIATION
CLASS 4 LASER PRODUCT
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 9 of 28
07-2009 Agfa Company Confidential
Page 12

DD+DIS238.06E
4 Product Complaints
Any service person who has any complaints or has experienced any dissatisfaction in the
quality, durability, reliability, safety, effectiveness or performance of this product must
notify Agfa HealthCare by the Agfa HealthCare complaint procedure.
If the product malfunctions and may have caused or contributed to a serious injury of a
patient or an accident or if there are any hazards which may cause an accident
Agfa HealthCare must be notified immediately by telephone, fax or written
correspondence to the following address:
Agfa Service Support - local support addresses and phone numbers listed on:
www.agfa.com
Agfa – Gevaert N.V.
Generic Safety Directions
Septestraat 27
2640 Mortsel, Belgium.
Fax +32 3 444 4485
5 References
Technical Documentation is available via MedNet (PDF) and your local Agfa HealthCare
support organisation (Paper).
Access to MedNet:
IntraNet: http://docs.agfanet/bu/mi/mednet/mednetcso.nsf
ExtraNet: http://extranet.agfa.com/bu/mi/mednet/mednetcso.nsf
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 10 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 13

DD+DIS238.06E
6 Intended Use
This Agfa HealthCare product should only be operated in a hospital or clinical radiological
environment by qualified staff.
It must only be operated according to its specifications and its intended use. Any
operation not corresponding to the specifications or intended use may result in hazards,
which in turn may lead to serious injuries or fatal accidents (for example electric shocks).
AGFA will not assume any liability whatsoever in these cases.
Make sure that the product is constantly monitored in order to avoid inappropriate
handling, especially by children.
The product must only be installed and put into operation under the specified conditions.
Generic Safety Directions
7 Intended User
This manual is written for Agfa trained Field Service Engineers and Clinical Application
Specialists, trained users of Agfa HealthCare products and trained diagnostic X–Ray
clinical personnel who have received proper training. Users are considered as the persons
who handle the equipment as well as the persons having authority over the equipment.
8 Qualifications for Operation and Service Tasks
This Technical Documentation describes adjustments and routines which must only to be
performed by qualified technical personnel.
The Agfa (trained) Field Service Engineers and Clinical Application Specialists must have
received adequate Agfa HealthCare training on the safe and effective use of the product
and applicable environmental and occupational safety matters before attempting to work
with it. Training requirements may vary from country to country.
Agfa (trained) Field Service Engineers and Clinical Application Specialists must make
sure that training is received in accordance with local laws or regulations that have the
force of law.
Your local Agfa HealthCare representative can provide further information on training.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 11 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 14

DD+DIS238.06E
Generic Safety Directions
9 Environmental and occupational Safety Instructions
Each Agfa (trained) Field Service Engineer and Clinical Application Specialist:
• Must make his or her personal contribution to improve safety and protect the
environment.
• When working on a customers site, has a duty to take reasonable care to avoid injury
to himself or herself or to others who may be affected by their acts or omissions.
• Is obligated to adhere strictly to regulations and instructions.
• Shall familiarise himself or herself with the provisions of the Agfa Healthcare
Health, Safety and Environment Policy and any specific rules or procedures relating to
occupational safety at work and the protection of the environment.
• Shall promptly report any near misses, accidents, incidents or dangerous occurrences
to their line manager and co-operate fully in any investigation.
• Shall co-operate with company management on matters relating to
health, safety and environment and, where appropriate, discuss with and / or assist
their manager in resolving matters relating to health, safety and environment.
• Shall ensure that any company equipment issued to them, or, for which they are
responsible, is correctly used and properly maintained.
• Shall wear protective equipment whenever instructed or if it is recommended to do so.
• Shall be responsible for good housekeeping in the area in which he or she is working.
• Shall report situations, which could put them at risk, on either company or
customers' premises, to their manager or supervisor; and, if warranted, directly and in
confidence, to the Health and Safety Co-ordinator, Global HSE Manager, or ultimately
to the Managing Director.
• Shall report any injuries, diseases or dangerous occurrences to his or her line
manager.
• Shall report any accidents, incidents or near misses to his or her line manager.
• Shall report any situation of which he or she is aware that is potentially dangerous.
• Shall comply with any health surveillance procedure instituted for his or her benefit or
for compliance with regulations.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 12 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 15

DD+DIS238.06E
10 Connections to other Equipment
Agfa HealthCare equipment must only be used in combination with other Agfa HealthCare
equipment or components if these are expressly recognized by Agfa HealthCare as
compatible. A list of such equipment and components is available from Agfa HealthCare
service on request.
Changes or additions to the equipment must only be carried out by persons authorized to
do so by Agfa HealthCare. Such changes must comply with best engineering practice and
all applicable laws and regulations that have the force of law within the jurisdiction of the
hospital.
The Agfa HealthCare products are designed to communicate with other devices in the
hospital network using DICOM protocols.
Generic Safety Directions
Connections to other equipment:
Warning:
Accessory equipment not complying with the safety requirements of this product
may lead to a safety hazard.
INSTRUCTION:
Consult the Technical Documentation before making any connections to other equipment.
Consideration relating to the choice of accessory equipment shall include:
• Use of the accessory equipment in the patient vicinity.
• Evidence that the safety certification of the accessory equipment has been performed
in accordance with the appropriate IEC 60601-1 and IEC 60601-1-1 harmonized
national standard.
In addition all configurations must comply with the medical electrical systems standard IEC
60601-1-1. The party that makes the connections acts as system Configurer and is
responsible for complying with the systems standard.
If required, contact your local service organization.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 13 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 16

DD+DIS238.06E
11 Accessories and Spare Parts
Parts and accessories replacement:
WARNING:
Hazards may be introduced because of component failure or improper operation.
INSTRUCTION:
• Replace defective parts with Agfa HealthCare original spare parts.
• Use only tools and measuring instruments which are suitable for the procedure.
• Only approved Agfa HealthCare accessories must be used. For a list of compatible
accessories contact your local Agfa HealthCare organization or www.agfa.com.
Generic Safety Directions
12 Compliance
Directive for HealthCare Imaging Products:
Council Directive 93/42/EEC of 14 June 1993 concerning medical devices
(OJ No L 169/1 of 1993-07-12)
• ANNEX I - ESSENTIAL REQUIREMENTS - GENERAL REQUIREMENTS The
products are designed and manufactured in such a way that, when used under the
conditions and for the purposes intended and, where applicable, by virtue of the
technical knowledge, experience, education or training of intended users, they will not
compromise the clinical condition or the safety of patients, or the safety and health of
users.
• ANNEX II - EC DECLARATION OF CONFORMITY: Full quality assurance system
ISO 13485
• ANNEX X - CLINICAL EVALUATION: The clinical evaluation follows a defined and
methodologically sound procedure.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 14 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 17

DD+DIS238.06E
Applied Standards for HealthCare Imaging Products
• IEC 60601-1, Ed. 3: Medical electrical equipment - Part 1: General requirements for
• ISO 14971:2000, Medical devices – Application of risk management to medical devices
• IEC 60601-1-2, It specifies the MANUFACTURER of the ME EQUIPMENT or ME
SYSTEM provides information to the RESPONSIBLE ORGANIZATION that is essential
in determining the suitability of the ME EQUIPMENT or ME SYSTEM for the
electromagnetic environment of use, and in managing the electromagnetic environment
of use to permit the ME EQUIPMENT or ME SYSTEM to maintain BASIC SAFETY and
provide its ESSENTIAL PERFORMANCE without disturbing other equipment.
Additional standards for documentation:
IEC 62079 Ed. 1: Preparation of instructions - Structuring, content and presentation
Harmonization:
Global Harmonization Task Force (GHTF) www.ghtf.org/
This document has been prepared to comply with Study Group 1 guidance document
of the Global Harmonization Task Force (GHTF) www.ghtf.org/
of a consistent, harmonized definition for a medical device that could be used within a
global regulatory model and would offer significant benefits to the manufacturer, user,
patient or consumer, and to Regulatory Authorities and support global convergence of
regulatory systems.
IECEE CB SCHEME
The IECEE CB (C
for acceptance of test reports dealing with the safety of electrical and electronic products.
It is a multilateral agreement among participating countries and certification organizations.
Agfa has produced a CB test report and claims national certification in all other member
countries of the CB Scheme.
Details see www.iecee.org
Generic Safety Directions
basic safety and essential performance
to assist development
ertification Body) Scheme is the world's first truly international system
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 15 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 18

DD+DIS238.06E
Radiation of radio frequency:
CAUTION: For USA only:
This equipment generates, uses and can radiate radio frequency energy and, if not
installed and used in accordance with the service manual, may cause interference
to radio communication.
Note:
This product has been tested and found to comply with the limits for a Class A
computing device pursuant to Subpart B of Part 15 of FCC Rules, which are designed to
provide reasonable protection against such interference when operated in a commercial
environment.
Generic Safety Directions
Operation of this equipment in a residential area is likely to cause interference.
The user will be required to take all necessary measures to correct the interference at
his own expense.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 16 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 19

DD+DIS238.06E
13 Safety Directions for Operation
Accessibility of the mains power switch:
CAUTION:
Do not obstruct the mains power switch.
Position the Agfa HealthCare product so that it is possible to disconnect the mains power
connection.
• Under certain conditions the Agfa HealthCare product will show a display containing a
message. This message will show that either a problem or action has occurred or that
a requested action is required or cannot be performed. The user must read these
messages carefully they will provide information on what to do. This will be either
performing an action to resolve the problem or to contact the Agfa HealthCare
service organization. Details on the contents of messages can be found in this
Technical Documentation.
• All images created using any image technology can show artifacts which could be
confused with diagnostic information. If there is any doubt that the diagnostic
information could be corrupted, additional investigations must be performed to get
clear diagnostic information.
• Ventilation openings must not be covered.
• If you notice conspicuous noise or smoke, disconnect the product immediately from
the mains.
• Do not pour water or any other liquid over the device.
• If a system malfunction causes an emergency situation involving the patient, operating
personnel or any system component, activate the emergency stop for the system
concerned. All motor driven system movements will be stopped.
• Do not store any magnetic media near or on devices, which produce magnetic fields,
since stored data may be lost.
Generic Safety Directions
Explosive environment:
DANGER:
Risk of explosion.
Never operate this device in zones where there are flammable anesthetics or oxygen which
may cause an explosion.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 17 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 20

DD+DIS238.06E
Usage of an un-interruptible power supply:
Warning:
Images can be lost due to power failure.
Connect the equipment to an un-interruptible power supply (UPS) or an institutional
standby generator.
14 Radiation Protection
Only qualified and authorized personnel shall operate any X-Ray system. In this context
qualified means those legally permitted to operate this X-Ray equipment in the jurisdiction
in which the X-Ray equipment is being used, and authorized means those authorized by
the authority controlling the use of the X-Ray equipment. Full use must be made of all
radiation protection features, devices, systems, procedures and accessories.
Ionizing radiation can lead to radiation injuries if handled incorrectly. When radiation is
applied, the required protective measures must be complied with.
Generic Safety Directions
15 Safety Directions for Cleaning and Disinfection
• Details about cleaning and disinfection or sterilization methods that may be used on
SYSTEM parts or ACCESSORIES that can become contaminated through contact
with the PATIENT or with body fluids, are referred to within the individual service
documents.
• Disconnect the power supply from the equipment prior to cleaning the equipment.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
DOCUMENT CONTROL NOTE:
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 18 of 28
07-2009 Agfa Company Confidential
Page 21

DD+DIS238.06E
Generic Safety Directions
16 General Safety Directions for Service Activities
• This system uses high voltage. Please consider the respective safety regulations.
• Electrical repairs and connections must only be performed by a qualified electrician.
• Mechanical repairs and connections must only be performed by a qualified technician.
• The safety directions for operation (see section 13) are also valid for all service
activities.
• During all service activities observe prescribed local and country-specific
requirements (e.g. occupational safety and accident prevention regulations).
• All existing screw connections must be tightened sufficiently firmly, but they may not
be overstressed when tightening. There must always be compliance with stated
torque values!
• Damaged or missing screws may be replaced only with the same screw types that
have the specified hardness rating. Unless a different value is listed in the
instructions, all Allen screws used must be hardness rated 8.8.
• All screws must be secured in accordance with the corresponding data.
If "Loctite" has to be used to secure screws, this is stated in the text.
• Any Agfa service PC or tool which is to be connected via RS232, RJ45, USB or other
interface to an Agfa device must not be connected to the mains but must be operated
on its internal battery or indirect supply (low voltage).
• When handling printed circuit boards (abbr.: PCBs) the following points must be
observed:
o Always switch off the equipment and unplug the power cord, before you
disconnect or connect cables on printed circuit boards.
o When working on PCBs, always wear an anti-static wrist strap. Never touch any
parts or components on PCBs with your bare fingers.
o PCBs have to be kept or transported in their protection bags. Never carry a
PCB without protection bag and walk on carpet or plastic floor covering
(electrostatic charge).
o Once the PCB is taken out of its protection bag, it has to be protected from
electrostatic charge by a grounded mat.
Static discharge at electrical components:
CAUTION:
Static discharge! Electrical components may be destroyed:
For the repair on electrical components, wear a grounding strap
(Order number: CM+9 9999 0830 0) around the wrist and connect the other end of this
strap on a grounded conducting metal piece.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 19 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 22

DD+DIS238.06E
Generic Safety Directions
17 Safety Directions for Installation Planning Activities
Protecting CR (Computed Radiography) Equipment against scattered X-Rays:
Warning:
Image plate is sensitive for X-rays. Poor image quality possible.
The digitizer and the cassette storage shall be protected against X-ray radiation this way,
that the annual dose equivalent at the installation place will not exceed 1 mSv.
Protecting Film-Screen Systems against scattered X-Rays:
Warning:
Film is sensitive for X-rays. Poor image quality possible.
The film-screen system shall be protected against X-ray radiation this way, that the annual
dose equivalent at the installation place will not exceed 1 mSv.
Accessibility of the power disconnection device:
Warning:
Electrical device. Shock possible.
INSTRUCTION:
• Do not position Agfa EQUIPMENT so it is difficult to operate the disconnection device
when an APPLIANCE COUPLER or separable plug is used as isolation.
• Local and International wiring regulations must be observed. Check all supplies and
voltages, currents, trips and fuses with the Hospital facilities department or their
engineers.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 20 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 23

DD+DIS238.06E
• The device complies with the EN 60601-1, 2006 standard for Information Technology.
This means that, although it is absolutely safe, patients may not come in direct contact
with the equipment. Therefore the operator console must be placed outside a radius
of 1.5 m around the patient.
• This device should be installed behind the institution firewall for network security and
anti-virus protection. No ongoing computer virus protection or network security for this
medical device is provided (e.g. a computer firewall). Network security and anti-virus
provisions are the ongoing responsibility of the user or institution.
Generic Safety Directions
R = 1.5m
1.5m
1.5m
Patient environment
Fixing equipment at the wall or floor:
Warning:
Unknown composition of wall or floor structure: Risk of injury or damage:
Hospital management is responsible for the position, location and fixing of all equipment.
Floor load:
CAUTION:
Heavy device may damage the floor covering.
Make sure that the floor covering is solid enough to stand the weight of the device.
Fixing equipment at the ceiling:
CAUTION:
Ceiling construction may be inadequate for fixing of equipment: Risk of injury or
damage:
Hospital management is responsible for the position, location and fixing of all equipment.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 21 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 24
DD+DIS238.06E
18 Safety Directions for Installation Activities
• If not otherwise stated, installation and configuration is performed by Agfa HealthCare
trained personnel.
• If damage of the package is visible from the outside contact your local AGFA
representative.
• Apart from wearing the required protective clothing, e.g. safety boots and gloves, care
must be taken that heavy loads are correctly lifted/carried to avoid injury. The relevant
instructions must be complied with. Heavy or awkward loads must be moved by
mechanical means or by several people.
• When installing the product be sure that there is either a mains plug or an all-cable
disconnecting device in the internal installation fitted near the product and that it is
easily accessible.
• Defective covers, sharp edges or protruding parts of equipment can cause injuries, if
accidentally knocked into. Route cables and position equipment safely.
• This device should be installed behind the institution firewall for network security and
anti-virus protection. No ongoing computer virus protection or network security for this
medical device is provided (e.g., a computer firewall). Network security and anti-virus
provisions are the ongoing responsibility of the user or institution.
Generic Safety Directions
Connection of the device to the power supply:
CAUTION:
Risk of damaging the device by using the wrong power supply:
INSTRUCTION:
Prior to connecting the device to the mains:
• Compare the power requirements indicated on the type label with the available
power supply in the installation room.
• Check the service manual for the type of input voltage selection, manual or
automatic: If manual, select the appropriate voltage and fuses.
• Confirm to use the correct socket and plug for the required power supply.
• Check the equipment will work with the power supply available.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 22 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 25

DD+DIS238.06E
Ground potential differences:
CAUTION:
To comply with ISO 60601-1 (annex I) all computers and peripherals must be
connected to the same power source.
INSTRUCTION:
• Always connect the associated monitor to the same Uninterruptible Power Source as
• When different combinations of equipment are used in various medical environments
Generic Safety Directions
the PC.
a potential difference (V) can exist between the protective earths in different localities.
If the protective earthing fails this potential difference can cause a HAZARD for the
OPERATOR or for the PATIENT.
19 Safety Directions for Maintenance and Repair Activities
• This Technical Documentation identifies the parts on which preventive inspection and
maintenance shall be performed by Agfa HealthCare service personnel, including the
periods to be applied.
• In general the device has to be switched off during service activities. Exception: If the
device is switched on to perform tests pay particular attention to any hazards due to
moving and rotating parts. Avoid lose clothing or finger traps. Switch off the device
immediately after the tests.
• Do not turn motors manually. If required, first disconnect the motor from the motor
control board.
• Make sure that the power cord does not show any signs of damage.
• After repair work always check that the integrated safety features are not overridden
or disconnected.
• If there is any visible damage to the machine casing do not hand-over the product to
the customer. First repair the machine casing.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 23 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 26

DD+DIS238.06E
Replacing batteries:
WARNING:
Battery can explode, causing chemical burns.
INSTRUCTION:
• Check that batteries are inserted with correct polarity.
• Only use batteries of the same type or an equivalent type as specified by the
manufacturer.
• Dispose of empty batteries in compliance with the specifications of the manufacturer.
• When removing lithium batteries from the equipment take appropriate measures to
avoid short circuit of the battery:
Either use tape to cover the two poles of the battery or put the battery back in its
original packing and secure the packing by tape.
Generic Safety Directions
Performing the electrical test according to national regulations:
WARNING:
Improper ground connections inside the device or too high leakage current may lead to
electric shocks.
• After any work at the power supply or at any component connected to mains voltage
inform the responsible organization
1
about the necessity of the electrical test
according to national regulations.
If specific national regulations do not exist: It is recommended to perform the
electrical test according to IEC 62353.
• Make sure, that all grounding connections to metallic covers and all grounding
connections inside the device are present.
NOTE:
On MedNet, GSO Library path "General Info => Agfa HealthCare => Publications =>
Service Manual" a form “IEC 62353:2007 Test Documentation” for CR Digitizers is
available, to be handed out to the responsible organization (Intranet Link
/ Extranet Link).
1
Responsible Organization:
Entity accountable for the use and maintenance of a medical equipment or a medical equipment system. The
accountable entity can be, for example, a hospital or an individual clinician.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 24 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 27

DD+DIS238.06E
Performing service activities at devices emitting laser radiation:
WARNING:
Laser radiation. Eye injury possible.
INSTRUCTION:
• Strictly observe the warning notes in the service manual of devices emitting laser
• Strictly observe the warning labels at the modules emitting laser light. For the
• Do not look into the laser beam.
Generic Safety Directions
radiation (See service manual chapter describing Safety Guidelines / General
Repair Instructions) and at the corresponding steps of instructions.
meaning of the labels refer to section 3.3 in this document.
• Do not open modules containing a laser. Only open modules containing a laser if
explicitly instructed to do so.
• Do not keep tools in the laser beam unless explicitly instructed to do so.
• Make yourself familiar with the path of the laser light and the conditions, when the
laser beam is switched on. Refer to the Functional Description in the
corresponding service manual.
• Do not operate modules with laser outside the device.
Sharp edges:
CAUTION:
Sharp edges inside the device: Cut or abrasion possible.
Be careful at maintenance and replacement of parts.
Cleaning optical elements:
CAUTION:
Image artifacts possible after cleaning optical elements.
When cleaning optical elements follow the service manual precisely.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 25 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 28

DD+DIS238.06E
Secured screws:
CAUTION:
Opening screws secured by red lacquer may misalign important device
adjustments:
Do not open screws that are secured by red lacquer.
Opening PCs and Workstations:
Warning:
Electrical shock and damage to the equipment possible.
• Only open the PC or workstation if explicitly stated in the service manual.
Generic Safety Directions
• Unplug before opening.
• Observe anti-static safety regulations.
Replacing fuses:
Warning:
Replacing fuses by wrong type may lead to fire hazard!
Use only fuses of the exact value and characteristics stated in the service manual or on
the device.
20 Safety Directions for remote Service Activities
Remote Service Activities:
Warning:
During remote service activities images can be lost.
Inform the customer prior to remote service activities to finish the current work and to
stop working on the system.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 26 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 29

DD+DIS238.06E
Generic Safety Directions
21 Safety Directions for Transport and Shipment of Spare Parts,
Accessories and Devices
• In compliance with transport regulations, all uninterruptible power supplies (UPS)
must be shipped with batteries disconnected.
• Use the original packing when returning spare parts, accessories or devices.
• Before returning any spare part with a built in lithium battery remove it and dispose the
batteries locally according to local waste regulations.
22 Safety Directions concerning Modifications
Modifications made in products/systems shipped by Agfa HealthCare must not be
implemented without written permission from Agfa HealthCare.
This applies in particular to changes which may affect the mechanical and/or electrical
safety or radiation-protection properties of a product (e.g. changing of safety distances,
removal of locks/instructions etc.).
23 Safety Directions concerning Hazardous Materials
'Hazardous materials' is the designation for substances which can ignite or explode or
which are toxic, injurious to health, corrosive or irritating. The “Hazardous Material”
instructions must be read and the required protective measures must be complied with
when performing work to avoid health risks.
Their properties together with the hazards and protective measures connected with them
are identified clearly by symbols and described by the instructions appertaining to the
hazardous substances.
24 Recycling
Agfa HealthCare has Recycling Passports available for all equipment. The Recycling
Passport explains whether hazardous materials, special components and batteries are
present, where they are located and how they can be removed at the end of the life cycle.
The Recycling Passports are meant to be used as information for waste treatment
partners and companies that want to recycle end-of-life Agfa equipment.
To get a copy of the required Agfa HealthCare Recycling Passport please contact your
local Sales organization.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 27 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 30

DD+DIS238.06E
25 Waste Disposal
On August 13, 2005, the European Directive on Waste Electrical and
Electronic Equipment (WEEE) 2002/96/EC, amended by Directive
2003/108/EC, came into force.
The directive on Waste Electrical and Electronic Equipment (WEEE) aims to
prevent the generation of electric and electronic waste and to promote the
reuse, recycling and other forms of recovery. It therefore requires the collection
of WEEE, recovery and reuse or recycling.
This directive has to be implemented into national law by the individual
European countries by August 13
Due to the implementation into national law, specific requirements can be
different within the European Member States.
th
2005.
Generic Safety Directions
This symbol on the product, or in the manual and in the warranty, and / or on
its packaging indicates that this product shall not be treated as household
waste.
For more detailed information about take-back and recycling of this product,
please contact your local Agfa service organization. By ensuring this product is
disposed of correctly, you will help prevent potential negative consequences
for the environment and human health, which could otherwise be caused by
inappropriate waste handling of this product. The recycling of materials will
help to conserve natural resources.
If your equipment or replaced spare parts contain batteries or accumulators
please dispose of these separately according to local regulations.
26 Erasing Protected Health Information (PHI)
AGFA HealthCare Field Service Personnel or its authorized affiliates are responsible for
the removal of Protected Health Information (PHI) patient data from devices, modules or
parts that are removed from the customer’s site. This also applies to the exchange of
spare parts, especially to parts that are returned to central warehouses for repair or
refurbishing. Examples for parts or modules that may contain Protected Health
Information (PHI) are: Computer hard disks, CD-ROMS, backup tapes, archive tapes.
The controlled version of this document resides on MedNet. Any printed copy of this document is uncontrolled.
Edition 1, Revision 3 Generic Safety Directions for HealthCare Imaging Products Page 28 of 28
07-2009 Agfa Company Confidential
DOCUMENT CONTROL NOTE:
Page 31

DD+DIS084.97E Pre-installation
Section 1
List of contents
1 INSPECTION OF PACKING.......................................................................... 1
2 UNPACKING INSTRUCTIONS...................................................................... 2
3 SPACE REQUIREMENTS............................................................................. 3
4 CHECKING THE ACCESSORY KIT.............................................................. 4
5 SPECIFICATIONS......................................................................................... 5
5.1 Environmental requirements..........................................................................................5
5.2 Dimensions and weight.................................................................................................. 5
5.3 Power requirements........................................................................................................ 6
5.4 Interface requirements ...................................................................................................6
6 AGENCY APPROVALS................................................................................. 7
01/28/98 DRYSTAR 3000 Section 1 / I
Page 32
DD+DIS084.97E Pre-installation
1 INSPECTION OF PACKING
Upon arrival, check inventory with the carrier.
• Inspect the packing material for obvious sig ns of damage such as crushed, punctured,
torn, broken, wet or ratt ling packages.
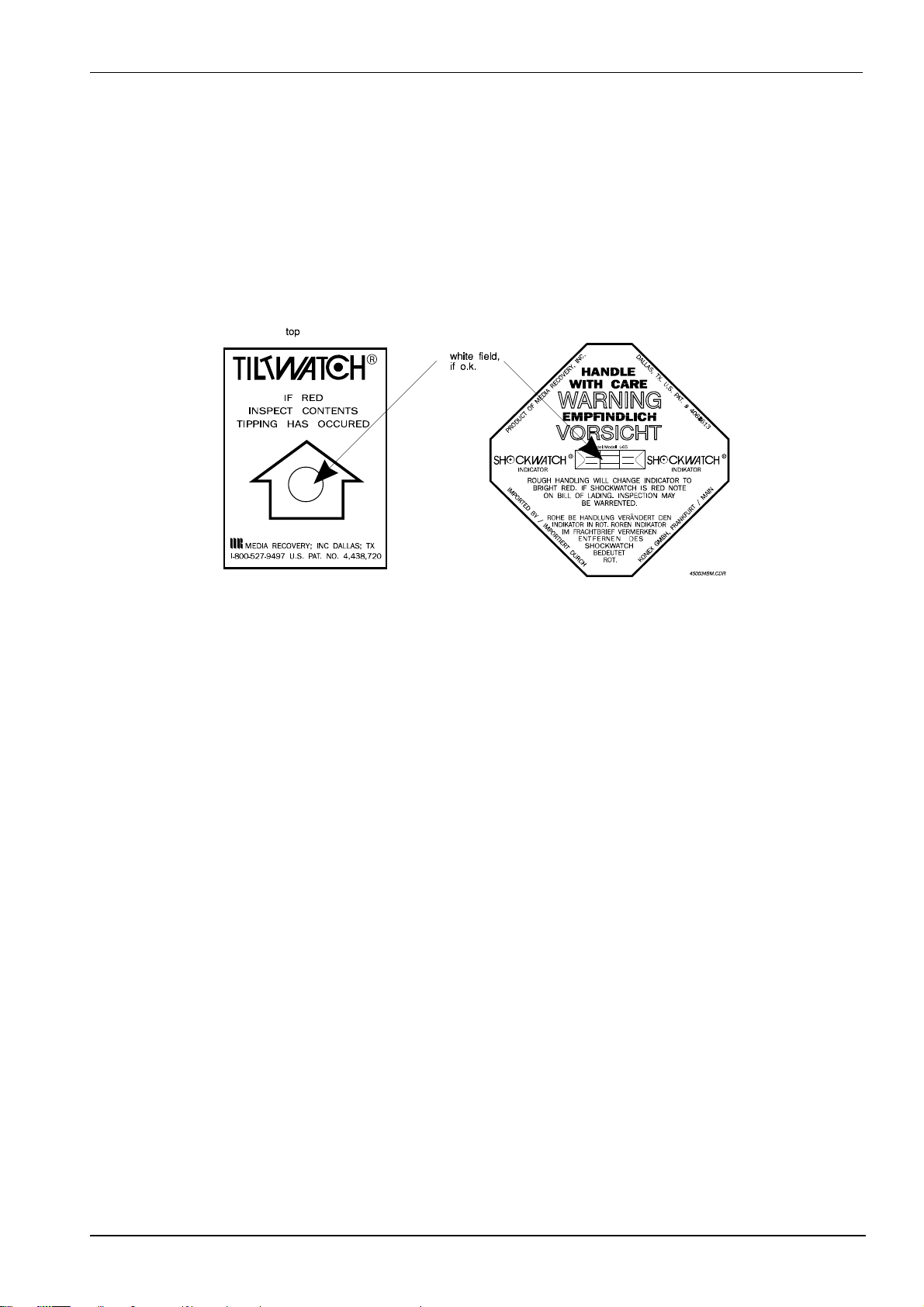
• Check the safety indicators on the machine pack ing.
The machine packing is provided with a "TILTWAT CH" indicat o r and a " SHO CKWATCH"
indicator. They are attached to the out side of the packing material and indicat e, if the
printer has been tilted, turned upside down or exposed to shocks dur ing transport.
Figure 1
If the machine was tilted, the cir cle in
the arrow head of the TILTWATCH
indicator changes from white to red.
If the machine was subjected to shocks the
square field in the middle of t he "SHOCKWATCH" indicator changes from white to
red.
• If damage is not evident, sign and stam p the bill of loading: conditions of cont ent unknown
- subject to inspection
• If damage is evident, contact your Purchasing Depar tment for action, have the carrier
indicate the damage on the fr eight bill, and sign all copies of the consig nm ent.
During formal inspection, you should:
• Open all packages within 15 days of receipt for a complet e inspection of the consignment.
• Report concealed damage to the carrier within 15 days of receipt or the carrier may not
accept liability.
01/28/98 DRYSTAR 3000 Section 1 / 1
Page 33
Pre-installation DD+DIS084.97E
2 UNPACKING INSTRUCTIONS
For unpacking refer to the unpacking instructions, enclosed i nsi de t he
packing, as shown in fi gure 2 bel ow !
Figure 2
Section 1 / 2 DRYSTAR 3000 01/28/98
Page 34
DD+DIS084.97E Pre-installation
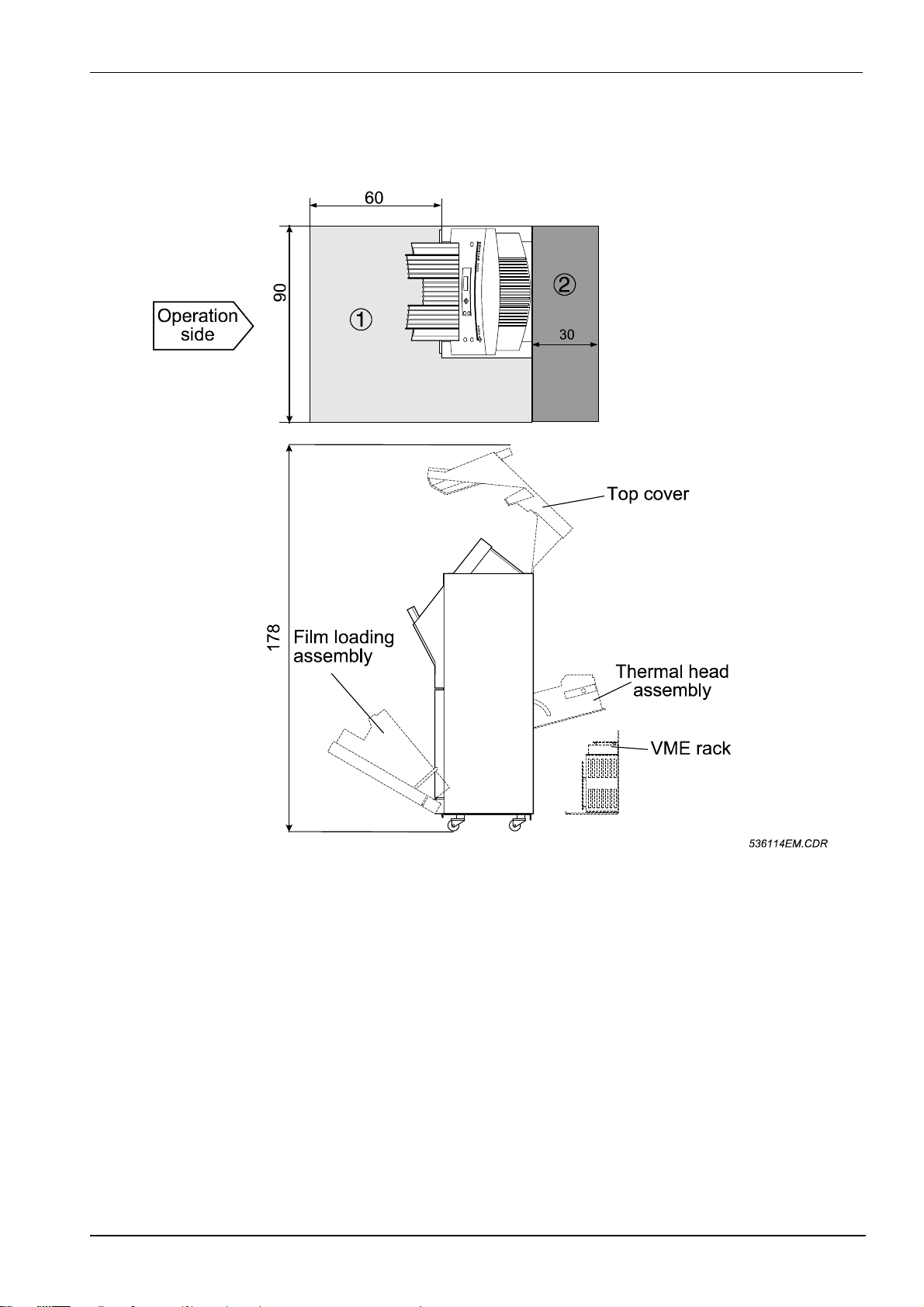
3 SPACE REQUIREMENTS
For easy access, it is recommended to respect the f ree space around the unit as shown in the
diagrams below.
Figure 3
Minimum space requirements for operation and servicing
01/28/98 DRYSTAR 3000 Section 1 / 3
Page 35

Pre-installation DD+DIS084.97E
Figure 4
Optimum space requirements for servicing
4 CHECKING THE ACCESSORY KIT
Compare the inventory of the accessory kit with the pack ing list. The packing list att ached to
the shipping carton specifies the exact contents of your accessory kit.
Section 1 / 4 DRYSTAR 3000 01/28/98
Page 36

DD+DIS084.97E Pre-installation
5 SPECIFICATIONS
5.1 Environmental requirements
You should install the printer in a location where the operating condit ions ar e within the lim it s
specified below.
Conditions Temperature Relative Humidity Air Pressure
Operating
Storage
Working-Mode Noise level Heat emission Max. permi ssi ble magn.
Standby
Printing
Throughput Film formats Film loading assembly
50 films (14 x 17") per hour on
an average
Dust (suspension) 0,2 mg/m³
Dust (sedimentation) 1,5 mg/(m²h)
+10° C to +35° C 10% - 80% 70 kPa to 106 kPa
-25° C to +55° C 10% - 100% 70 kPa to 106 kPa
field in the room
45 dB (A) max. 200 W 30 Gauss
55 dB (A) max. 650 W 30 Gauss
capacity
14 x 11"
14 x14"
14 x17"
100
5.2 Dimensions and weight
Dimensions
Printer With packing Without packing
Length (cm) 89,3 57
Width (cm ) 72,4 60
Height (cm) 160 134
Weight
Printer Floor load
With pack ing (kg) 160 58,4 N/cm² per area
Without packing (kg) 125 58,4 N/cm² per area
01/28/98 DRYSTAR 3000 Section 1 / 5
Page 37

Pre-installation DD+DIS084.97E
5.3 Power requirements
Ensure that adequate AC input is available. The printer’s power requirements are stated on
the serial number identifying label located on the rear panel. A grounded AC outlet, according
to one of the specifications list ed below, should be provided in the dir ect environm ent of the
printer.
Printer Drystar 3000 5361/100 Tolerance
Frequency (Hz) 50 60
Power input 3 wire (safety ground) single phase
Nominal voltage (V AC) 110 - 120 V AC 110 - 120 V AC +5% / - 5% of
rated voltage
Power consumption (W)
Standby:
Printing:
Fuses
Power supply (A):
Decurling unit (A):
15 A f (fixed fuse)
270
700
2 x 8 A ff
Leakage current < 3.5 mA
Protective conductor < 0.12 W to
5.4 Interface requirements
Depending on the printer configuration, the appropriate video input signal or net work
connection is to be provided prior to installation as shown in the table below:
Signal Printer configuration
Ethernet network node with Ethernet interface
Analog RGB or B/W video with analog video interface (VSI and SWB II)
Digital with MFRI interface
to protective ground
protective ground
< 3.5 mA
to protective ground
< 0.12 W to pr ot ect ive
ground
Section 1 / 6 DRYSTAR 3000 01/28/98
Page 38

DD+DIS084.97E Pre-installation
6 AGENCY APPROVALS
The printer has been tested and f ound t o comply with the following international standards
and regulations:
Safety standards:
• TÜV approval according to: IEC 601-1, IEC 601-1- 1, VDE 0750 Par t 1 (12.91):
Medical electrical equipment - General requir em ents for safety (Germany)
• UL approval according to: UL 2601-1:
Medical electrical equipment (USA)
• C-UL approval according to: CSA 22.2 No. 601.1 M90:
Medical electrical equipment (Canada)
Radio-interference regulations (interf erence suppression):
• FCC rules 47 CFR-part 15-subpart B, class A. (emission r ules - USA)
• EN60601-1-2 (1993)
• EN55011 class A; emission
• EN61000-3-3; Flicker, short term flicker PST<=1,0
• §N61000-3-2; Harmonic class A
USA regulations:
• FDA 510k requirements
• FDA par t 820 Good Manufacturing Practice for Medical Devices requirements
01/28/98 DRYSTAR 3000 Section 1 / 7
Page 39

DD+DIS084.97E Controls and connectors
Section 2
List of contents
1 LOCAL KEYPAD........................................................................................... 1
2 REAR PANEL................................................................................................ 2
3 VIDEO CONNECTION................................................................................... 3
4 NETWORK CONNECTION............................................................................ 4
5 EXTERNAL KEYPAD.................................................................................... 5
6 JAM CLEARANCE BUTTON......................................................................... 6
01/28/98 DRYSTAR 3000 Section 2 / I
Page 40
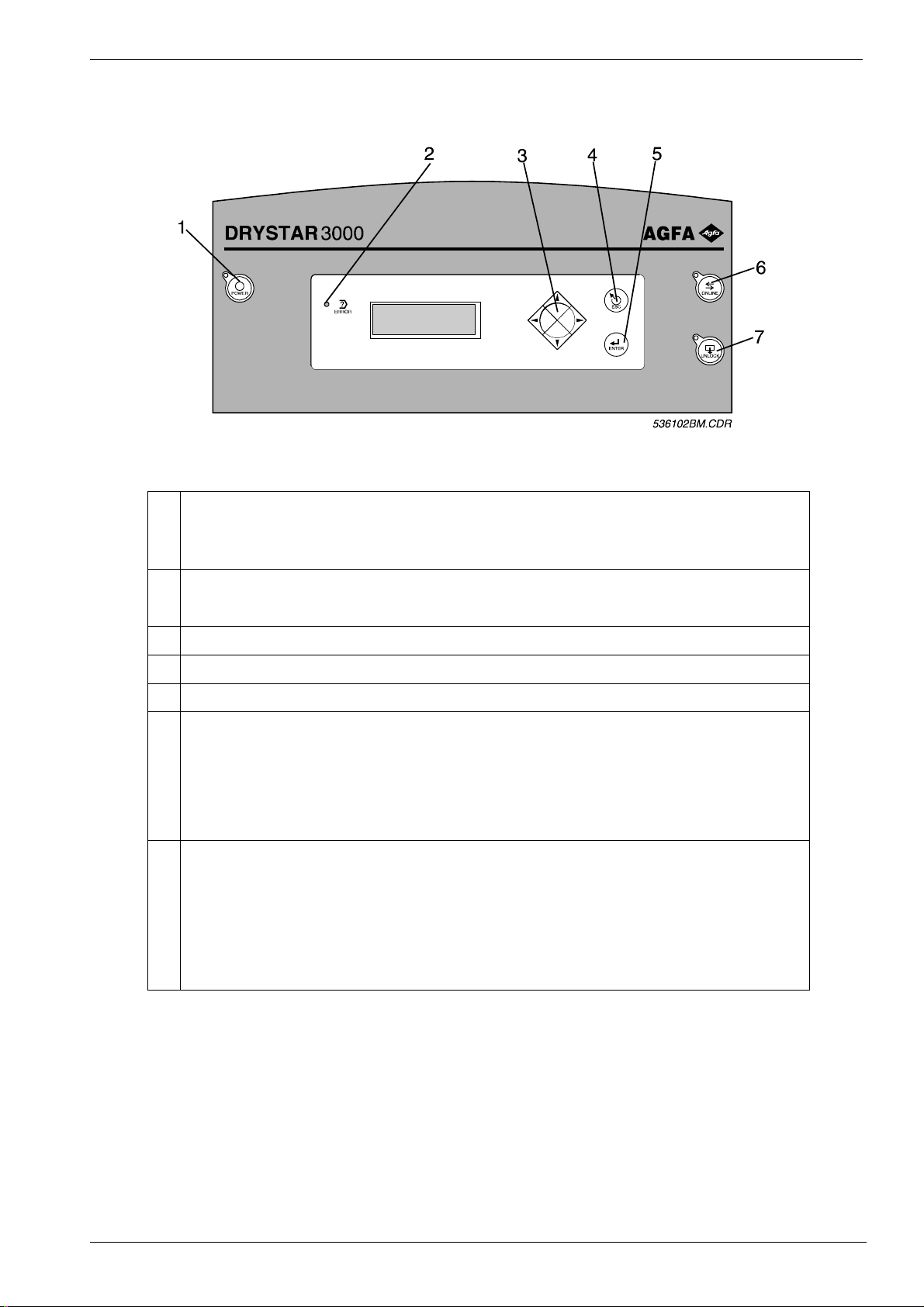
DD+DIS084.97E Controls and connectors
1 LOCAL KEYPAD
Figure 1
1 Power key, switches the printer on and off.
A delay circuit prevents the printer from voltage peaks. It may take up to 3 sec.
between switch-on and start-up of the printer .
2 Error LED, lights up when an error occurs.
Depending on the severity of the error a beeper is switched on, t oo.
3 Cursor key, to enter the local mode and scroll through the display screens.
4 ESC key, to leave the current display without saving.
5 ENTER key, to confirm the selection in the local keypad display.
6 ONLINE key, to switch the printer online or offline. The LED lights up if the
printer is online.
Online means, the printer is prepared to acquire diagnostic images.
Offline means, the printer cannot acquire diag nost ic im ages. Printing testimages
is possible however.
7 UNLOCK key, to open the front door. The LED lights up if the front door is open.
By pressing the unlock-key, the lock ing solenoid releases the two lids (front and
top - cover) for about 3 sec.
Note: In case the machine is switched off , the unlock key is disabled. Use a
screwdriver instead to open the lid. Refer to t he oper at ion instructions for more
information.
01/28/98 DRYSTAR 3000 Sect ion 2 / 1
Page 41

Controls and connectors DD+DIS084.97E
2 REAR PANEL
1
4
3
5
2
6
7
8
15
9
10
14
Figure 2, Example
13
12
11
536102AM.CDR
1
External Keypad Connectors, see page 5.
2*
Network Connection ( 10B2, Twisted pair, MAU), see page 4.
3*
Video Connection. See page 3.
4*
Video external Sync. See page 3.
5*
Video Sync Selection Receiver. See page 3.
6
Floppy drive To read and write 3,5“ DOS formatt ed floppies.
7*
RS232 or RS422 control port, optional. Used for host control connection.
8*
9*
10
Interground Bolt To be connected to the host device if necessar y. See sect ion 6. 1.
11
Service Port. RS232 (Sub-D 25 pin female) Connector for service PC.
12
Decurl Fuse Holder. It keeps two fuses for the decurl unit heater.
13
Mains Connector
14
Supplementary protective earth connector
15*
Connector plate for MFRI. 37 pin interface connector.
* optional
3 VIDEO CONNECTION
Section 2 / 2 DRYSTAR 3000 01/ 28/ 98
Page 42
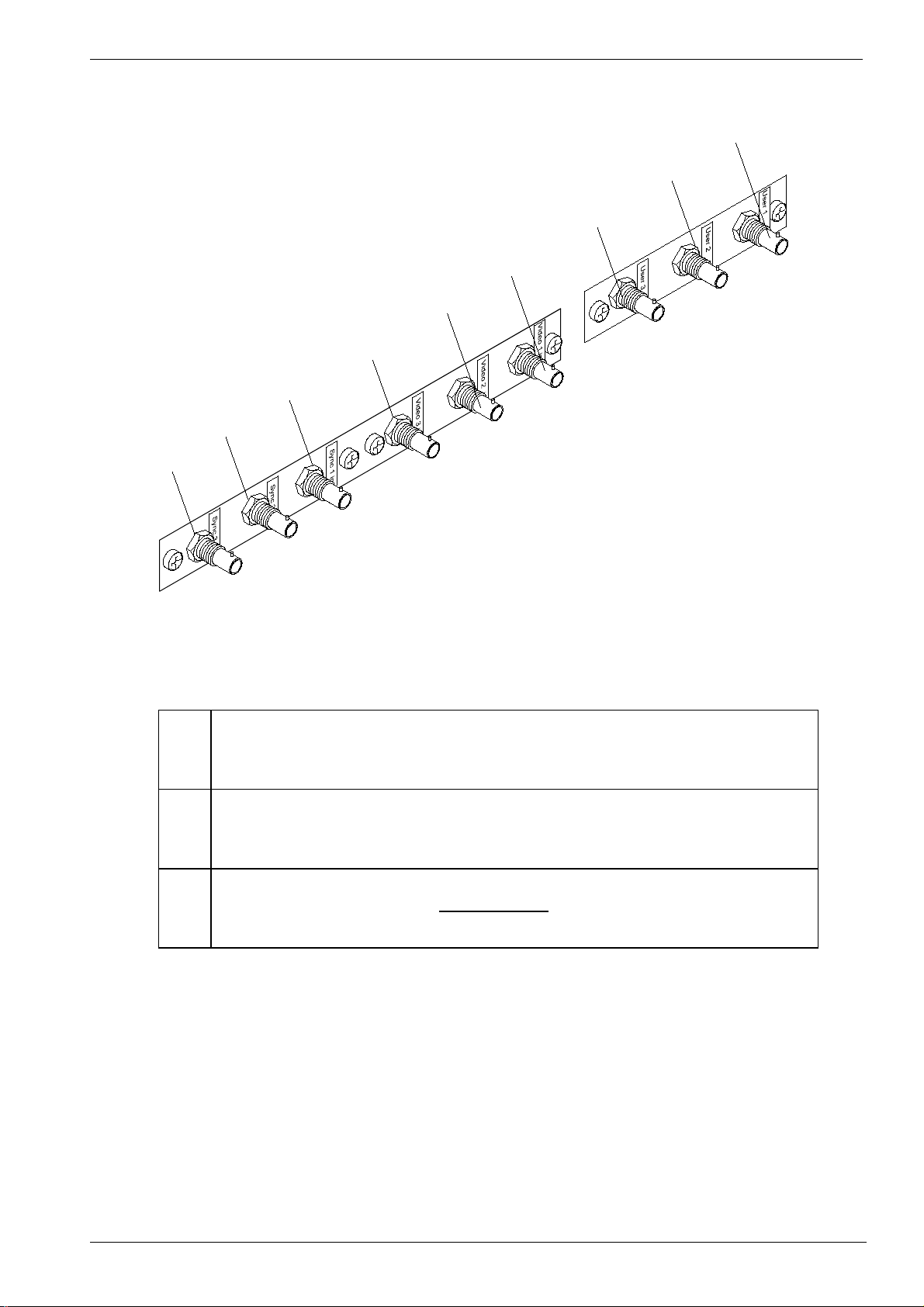
DD+DIS084.97E Controls and connectors
7
8
9
1
2
536102OM.CDR
3
4
5
6
Figure 3
1
Video inputs 1, 2 and 3. Fibre optic connector. Receives the video signal.
2
3
4
Sync 1, 2 and 3 input. Only used in case an external sync (synchronization)
connection is necessary.
5
6
7
Sync Selector 1, 2 and 3 input. Only used in case of an external sync
(synchronization) connection in combination with a VIDCOM III transmitter
8
box.
9
01/28/98 DRYSTAR 3000 Sect ion 2 / 3
Page 43

Controls and connectors DD+DIS084.97E
R
4 NETWORK CONNECTION
1
2
3
535802EM.CD
Figure 4
1
AUI Ether net (Sub-D 15 pin female) connection to an external t r ansceiver. *
2
10B2 50 Ω connection to coax cable Thin Ethernet . *
3
TP Thin Ethernet Twisted Pair connection.*
*Remark: No automatic „t ype of connection“ recognition. Jumperset t ing is necessary
according to the connection.
Section 2 / 4 DRYSTAR 3000 01/ 28/ 98
Page 44

DD+DIS084.97E Controls and connectors
5 EXTERNAL KEYPAD
FIGURE 5
Item Keyname Screen Function of the key
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
ESC
HELP
QUEUE
F1
INFO
F2
B&W/COL
F3
UP/DOWN
RIGHT/LEFT
SET UP
FORMAT
ERASE
PRINT
EXAM
COPY
STORE COL
STORE B&W
–
PLUS
–
All Returns to the previous screen without changes
All NOT YET IMPLEMENTED
Exam Group, Set,
To access destination(s) and queue(s)
Work, Destination
Work, Destination To view the destination specifications
Work Toggles between B/W and color acquisition
If applicable Scrolls or moves up/down
If applicable Moves left/right or page up/down
Exam Group, Set To access the set-up screen
Work , Patient ID To select a film format or to enter alphanumeric
characters
Work
Patient ID
Work + Copy (13)
Queue
Deletes the last captured, previous or selected
image
Deletes the last character
Deletes the predefined number of copies
Deletes the selected print job
Work Prints the current film and displays a new one
Work Closes an Exam folder
Work
Patient ID
Destination
To change the number of copies
To enter the selected character
Toggles the destination selection on/ off
If applicable Expose key or saves the entered color
information
If applicable Expose key or saves the entered B&W
information
– Connector for foot switch
Work, Queue Toggles emer gency priority on/off
– Intensity of LCD display
01/28/98 DRYSTAR 3000 Sect ion 2 / 5
Page 45

Controls and connectors DD+DIS084.97E
R
6 JAM CLEARANCE BUTTON
536102NM.CD
Figure 5
The button, located under the top cover at the left side of the printer, can be used to clear a
jam. The decurl motor M2 and the main drive mot or M1 are started simultaneously.
The decurl motor M2 drives the jammed film towards the decurl exit.
The main drive motor M1 drives the jamm ed film towards the film loading assem bly.
Remark: These two actions always are executed the same way, no matter whether the left or
right part of the button is hit.
Section 2 / 6 DRYSTAR 3000 01/ 28/ 98
Page 46

Page 47

DD+DIS170.99E Installation /Machine Start-up
Section 3
List of contents
1PRE-CHECK................................................................................................................... 2
1.1 Site inspection............................................................................................................................. 2
1.2 Installation checklist................................................................................................................... 2
1.3 Scope of delivery......................................................................................................................... 3
1.4 Removing the transport protection........................................................................................... 3
1.5 Mounting the flat belt of the main drive.................................................................................... 3
2INSTALLATION................................................................................................................ 3
2.1 Technical installation.................................................................................................................. 3
2.1.1 Standard delivery..........................................................................................................3
2.1.2 Mains connection..........................................................................................................3
2.1.3 Switch on device...........................................................................................................3
2.1.4 Loading films................................................................................................................ 4
2.1.5 Print SMPTE test image from local keypad .................................................................. 4
2.1.6 Check the configuration of the device (IMOS).............................................................. 5
2.1.7 Check Time and Date................................................................................................... 6
2.1.8 Fill out Site Info............................................................................................................. 7
2.1.9 Set the installation date................................................................................................ 7
2.1.10 Positioning of the printer............................................................................................... 7
2.2 Connectivity................................................................................................................................. 8
2.2.1 External cabling............................................................................................................ 8
2.2.2 Autolock of VSI........................................................................................................... 10
3SYSTEM INTEGRATION.................................................................................................. 10
3.1 Destination/Network.................................................................................................................. 10
3.2 Example for a simple Network .................................................................................................11
3.3 Connect the service host (optional)........................................................................................ 12
4APPLICATION............................................................................................................... 12
4.1 Introduction: Customization / NVE.......................................................................................... 12
4.2 Application / Customization Overview.................................................................................... 13
4.3 Language.................................................................................................................................... 14
4.4 Disk Space Quotas.................................................................................................................... 14
4.5 Edit keypads .............................................................................................................................. 15
4.6 Print a diagnostic image........................................................................................................... 17
5KICK OFF..................................................................................................................... 18
5.1 Backup........................................................................................................................................ 18
6ADDENDUM A: REMOVING THE TRANSPORT PROTECTION ............................................... 19
7A
DDENDUM B: MOUNTING THE DETACHED FLAT BELT OF THE MAIN DRIVE
(LEFT HAND MACHINE SIDE)........................................................................................... 23
7.1 Attachment of the flat belt on the flat belt pulley................................................................... 23
09.01.00 Type 5361 Section 3 / I
Page 48

Installation /Machine Start-up DD+DIS221.98E
1 Pre-Check
1.1 Site inspection
Take care that you have performed the necessary steps of the site inspection. A specif ic
readiness checklist concerning the site inspect ion t opics is included in the installation planning
section of this documentation (section 14). It is intended that you send the filled out readiness
checklist to the NSO in advance.
Thus, to your own advantage, it is ensured, that all necessar y prepar at ion work is done before
delivery and installation.
1.2 Installation checklist The following order of listed t asks i s mandat ory !
Action
1 Pre - Check
Ensure site inspection items
Unpack the printer
Check scope of delivery
Remove transport protection elements
The following order of listed t asks i s not mandat ory !
Action
2 Installation
Technical installation
Connect mains cable
Switch on
Load film
Print test image f rom local keypad
Check the configurat ion of the device (IMOS)
Compare with the delivered OGT file
Check time and date
Fill out site info
Set the installation date
Position the printer at its f inal locat ion
Task
Code
INS
Task
Code
INS 2.1
See
section
Add:A
See
section
14
1
1.3
Connectivity
Perform external connection (cables)
Perform autolock ( in case of VSI installed)
print diagnostic image f r om the host
3 System integration
Adjust network parameters (ART - Par am eters)
Connect service host (optional)
Section 3 / 2 5361 09.01.00
INS 2.2
SIC 3.
Page 49

DD+DIS170.99E Installation /Machine Start-up
4 Application
Adapt disk space quotas
Edit keypads
Print diagnostic image
5 Kick off
Backup
Train the user
1.3 Scope of delivery
Check the scope of delivery by means of the pack ing list, attached to the printer.
1.4 Removing the transport protection
To remove the transport protection elements refer to the addendum A of this section.
1.5 Mounting the flat belt of the mai n dri ve
For the mounting procedure of the main drive flat belt ref er to Addendum B of this section.
Do not connect the printer to the mains outlet before you finished all
actions described in the above mentioned Addenda A and B (refer to
item 1.4) !
APP 4
OTR 5
2 Installation
The installation sequence described in this part has been designed to make the installation as
easy as possible. The procedure below outlines the recommended installation steps.
All printers are configur ed at production site as defined by marketing .
2.1 Technical installation
2.1.1 Standard delivery
The printer is provided with an auto switching power supply. For this reason there are no
adaptations to perform t o align to local line voltages or freq uencies. There are no voltagespecific fuses to install.
default language is: - English
2.1.2 Mains connection
• Mains cable
Install the printer to the same g round as the host system for safet y reasons. If this is not
possible see section 6.1 for solutions
• Supplementary protective earth cable
This cable is plugged onto the supplementary earth connector (not included, has to be
ordered separately under ABC-Code: 79K67)
Apply this cable only in case the printer is connected to other non IEC 601 conf orming
equipment.
2.1.3 Switch on device
Switch on the device with the power switch in front of the printer.
09.01.00 5361 Section 3 / 3
Page 50

Installation /Machine Start-up DD+DIS221.98E
2.1.4 Loading films
Refer to the user manual to perform the correct action for
loading films.
You also can see the sequence of handling by imprinted
icons on top of the film loading assembly. To see these
icons, open the top cover and the cover of the f ilm loading
compartment (see Figure 1) .
Figure 1
2.1.5 Print SMPTE test image from local keypad
When all req uirements in the previous paragraphs are f ulfilled, a first test im age has to be
printed to verify the mechanical operation of the printer.
• Print an SMPTE testimage:
536103AZ.CDR
Figure 2: SMPTE testimage
?
ERROR
1
ESC
ENTER
3 x
?
2
ERROR
ERROR
?
ESC
ENTER
ESC
3
ENTER
?
ERROR
4
1 x
?
ERROR
ESC
ENTER
ESC
5
ENTER
wait ca. 2 minutes
?
6
ERROR
3 x
ESC
ENTER
536110AB.CDR
Figure 3: Print SMPTE testimag e at
the local keypad.
Section 3 / 4 5361 09.01.00
Page 51

DD+DIS221.98E Installation /Machine Start-up
2.1.6 Check the configuration of the device (IMOS)
2.1.6.1 Starting the I M O S Drystar 3000 service software
For to check the config ur at ion of the printer, perform the following steps to enter t he IMOS
Drystar 3000 service program:
• Connect your service PC locally to the printer
• Switch on your service PC.
• Switch on the printer.
• Select the Drystar 3000-Icon.
Figure 4
• The IMOS Drystar 3000 service program shows up with in a separate window as shown
below.
Figure 5
09.01.00 5361 Section 3 / 5
Page 52

Installation /Machine Start-up DD+DIS221.98E
2.1.6.2 Check configuration
In general, the delivered printer unit is pr econfigured, with all interfaces inst alled accor ding to
the order at OSD in Belgium. For t hi s reason t here i s no f urther conf iguration
necessary !
Configuration at product ion sit e affects the following items:
- 3 input s analog: VSI and switchboard 2 + opt. SSR1 (ext. sync.)
digital: MFRI + 3M (P 952)
USP (SPC 1)
AMDI
- 1 out put Drystar 3000
- 1 network APIP / DICOM* / Postscript*
*) optional
The window above shows a listing of a specific configuration. You may ensure t he correctness
of the configuration via the IMOS Drystar 3000 service program. In order t o do t his, compare
the attached OGT f ile (part of the delivery) with the actual config uration entries.
In case of discrepancies you have to reconfigure the Drystar 3000. Refer to section 6. 2 or 9
2.1.7 Check Time and Date
Check time and date in the IMOS Drystar 3000 application menu and adjust if necessary.
Figure 6
Note: Time and Date influences
• Patient ID: e.g. 804241430 = year 1998 Month 04
Day 24 Hour 14 Min 30
• The logbook time stamping
• The installation date
Figure 7: IMOS Window f or t i me and dat e adjust ment
Section 3 / 6 5361 09.01.00
Page 53
DD+DIS221.98E Installation /Machine Start-up
2.1.8 Fill out Site Info
• In IMOS Drystar 3000, go to "I nstall → Installation Report".
• Select "Site Info"
• Fill out the site specific inform ation. This will be part of the info count er header.
2.1.9 Set the installation date
• In IMOS Drystar 3000, go to "I nstall → Installation Report".
• Select "date of installation": Now automatically the cur r ent date is set as device
installation date.
Note: The date of installat ion can only be set once.
2.1.10 Positioning of the printer
When positioning the printer at ist final location, please ensure that the stoppers at the wheels
are locked.
Figure 8: Site info (Example)
Figure 9: Installation Date
09.01.00 5361 Section 3 / 7
Page 54

Installation /Machine Start-up DD+DIS221.98E
r
2.2 Connectivity
2.2.1 External cabling
• Refer to the corresponding interface release document available on DIGIMAIL (+49 89
6207 7777 analog / +49 89 69370160 ISDN), download area „interf aces / connectivity“ or
http://medical.agfa.com (AGFA Medical Homepage).
2.2.1.1 Video Connection. Monitor in the video chain
A
F
G
A
0
0
0
3
R
A
T
S
Y
R
D
VIDEO OUT
A
VIDEO IN
75 Ohm Terminator
VIDEO OUT
A
VIDEO IN
(Fibre)
VIDCOM
HOST
MONITOR
536103SZ.CDR
Figure 10
2.2.1.2 Video Connection: Monitor at the end of the video chain
A
F
G
A
0
0
0
3
R
A
T
S
Y
R
D
VIDEO OUT
A
VIDEO IN
VIDCOM
VIDEO IN
(Fibre)
A
Host
VIDEO OUT
536103RZ.cdr
MONITOR
Figure 11
• Make sure that the BNC-coax cables (A) used in the video chain are as short as
possible. For video signals above 100 MHz the coax-cables should not be longer
than 5 m.
• optical cables can have a length of up to 350m.
• For jumper settings of t he VSI board refer to IMOS Drystar 3000: REPAI R → VME
Boards
75 Ohm
Terminato
Section 3 / 8 5361 09.01.00
Page 55

DD+DIS221.98E Installation /Machine Start-up
2.2.1.3 Example for VIDCO M and VSI connection (here: color input)
Figure 12
09.01.00 5361 Section 3 / 9
Page 56
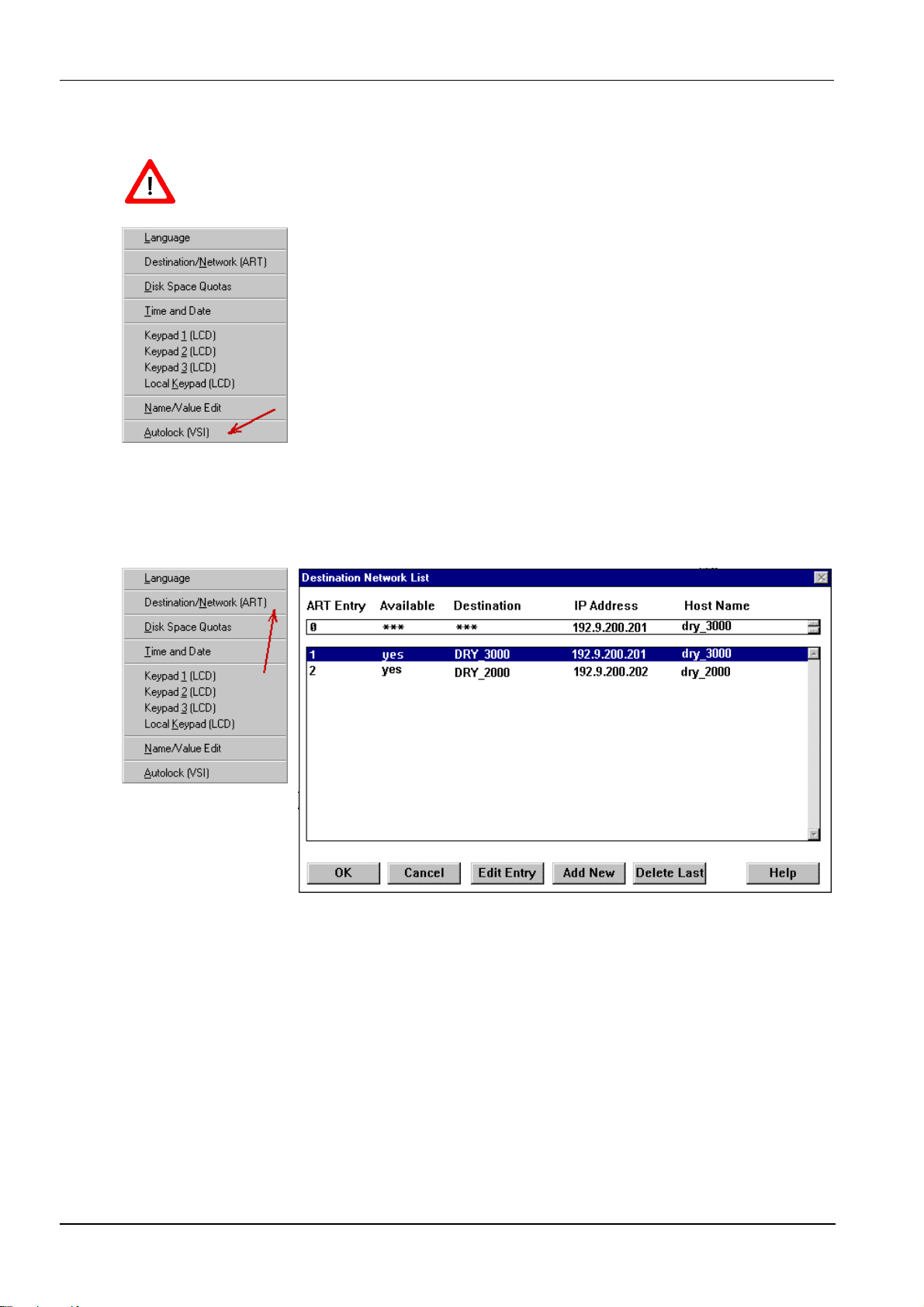
Installation /Machine Start-up DD+DIS221.98E
2.2.2 Autolock of VSI Autolock is only needed for analog connections
The autolock procedure is described in section 9, VSI.
Figure 13
3 System integration
3.1 Destination/Network
Figure 14
• The Destination Network List shows every configured Network Node with its corr esponding
parameters.
• To edit a new Network Node, press <ADD NEW>
Section 3 / 10 5361 09.01.00
Page 57

DD+DIS221.98E Installation /Machine Start-up
y
)
3.2 Example for a simple Network
0
0
0
3
R
A
T
S
Y
R
D
ART ENTRY 1
Destination name: DRY_3000
Host name: dry_3000 (= AOS shell prompt)
IP Address: 192.9.200.201
Port: 3027
Type: Drystar 3000 (Typ 53614)
Availability: Enabled
Input/Output: Local Printer
ART ENTRY 1
Capture images at Drys tar 3000
Print images at Drystar 3000
(in Drystar 3000)
=
5
6
3
0
1
3
Z
L
C
.
D
R
ART ENTRY 2
(in Drystar 3000)
Destination name: DRY_2000
Host name: dr
_2000 (= AOS shell prompt
IP Address: 192.9.200.202
Port: 3027
Type: Drystar 2000 (Typ 5358)
Availability: Enabled
Input/Output: Remote Printer
ART ENTRY 2
Capture images at D rystar 3000
Print images at Drystar 2000
=
Note 1
Note 2
For ART0 settings see screen in previous page (ART 0 = ART 1)
In case images captured on Drystar 2000 shall be print ed on t he Drystar 3000, too, the
Drystar 2000 settings have to be made accordingly.
09.01.00 5361 Section 3 / 11
Page 58

Installation /Machine Start-up DD+DIS221.98E
3.3 Connect the service host (optional)
• In case a service host (see Figure 15) was ordered with the device
(refer to OGT file, delivered with the device), make the connections as
described in the installation instructions delivered with the service host.
• Pass the corresponding RSC or GSC (RSC = Regonial Support
Center, GSC = Global Support Center) t he network parameters as
described in the service host installation instructions.
Figure 15
4 Application
4.1 Introduction: Customizati on / NVE
Application can also be called Customization. Customization is the adaptation of the printer to
the customers need. IMOS offer s for most customization steps comf or table shells.
Figure 17
Figure 16
Note: Name/Value Edit is a fast alternative for the advanced user,
to adjust specific parameter s of the printer directly.
For an overview of all important parameters see document
DD+DIS115.98E, NVF parameters MG3000 and Drystar.
Section 3 / 12 5361 09.01.00
Page 59

DD+DIS221.98E Installation /Machine Start-up
536103JZ.C
4.2 Application / Customization Overview
Customization is the adaptation of the system to t he customers needs. Following parts of the system
are concerned:
• The user display
• The harddisk
• The film
Enclosed a graphical overview of the most important custom ization items.
See next page for explanation of figures 1 to 11
User Display
INFO
QUEUE
HELP
ESC
1 2 3 4 5
SET UP FORMAT
F1
F2
Drystar 3000 US Room 2
<1> Dr. Miller
PRINT EXAM
ERASE
US Room 2 Dr. Miller
<1> 1/0 Kidn. pos.
B&W/COLB&W/COL
F3
IF 220
IS 80
IA 3
F4
6 7 8 9 0
STORE
COPY
COL
Dr. Miller
8x10 1/0 Kidn. Pos
Pt: US802041430
Copies: 1
To: DRY_2000
Queue: 12/16
STORE
B&W
Harddisk Drystar 3000
3
4
Partition F:
2
9
11
B&W Image
5
6
1
10
7
8
DR
Figure 18
Table with recommended adjustments during customi zat i on see next page
09.01.00 5361 Section 3 / 13
Page 60

Installation /Machine Start-up DD+DIS221.98E
Table with recommended adjustments during customizat ion
Adjustment Where to find
1 Time and Date Time and date (section 2.1.7)
2 Disk Space Quota Disk Space Quota (section 4.4)
3 Hospital Name and Location* Edit Keypad (section 4.5)
4 Exam group names* Edit Keypad (section 4.5)
5 Exam Set ID`s* Edit Keypad (section 4.5)
6 Black & White Layouts* Edit Keypad (section 4.5)
7 Contrast B&W* Edit Keypad (s ect ion 4. 5) Edit Films
8 Interpolation ( Shar pness) * Edit Keypad (section 4.5) Edit Sources
9 Film recovery after power failure Name Value Edit: MNU: parameter
b_cftbl_usr[1].no_recovery **
10 Set I CO N for film reduction
11 Activate annotations
* To make these parameter s act ive, no r eset r equired. Simply confirm with <ok>, leave the
current keypad screen and re-enter it .
**see DD+DIS115.98E; NVF Parameters
grey fields mean optional, i.e. not mandatory
Name Value Edit: DSP: parameter icon_pos **
Name Value Edit: MNU: parameter annotation **
4.3 Language
At the time being only English
4.4 Disk Space Quotas
Adapt the disk space quotas according t o the input image size and workload of the inputs.
Note: Input 4 is the local keypad. Do not adj ust input 2 below 10 MB
Figure 19
Section 3 / 14 5361 09.01.00
Page 61

DD+DIS221.98E Installation /Machine Start-up
4.5 Edit keypads
• Select the Keypad, you wish to customize.
Figure 20
• Select the desired exam set
Figure 21
• Select the next action to be performed via the 3 edit buttons at the bottom of the above
shown screen.
• Change the values for interpolation and taste look up table (and eventually other settings,
too) as described in section 5, application.
• Use <DELETE ALL> to delete all examination groups at once.
Selection: <Copy Exam Set>
• To shorten the amount of time for the customization you can copy specific completed
examination sets. After that you only have to modif y a few remaining parameters.
09.01.00 5361 Section 3 / 15
Page 62
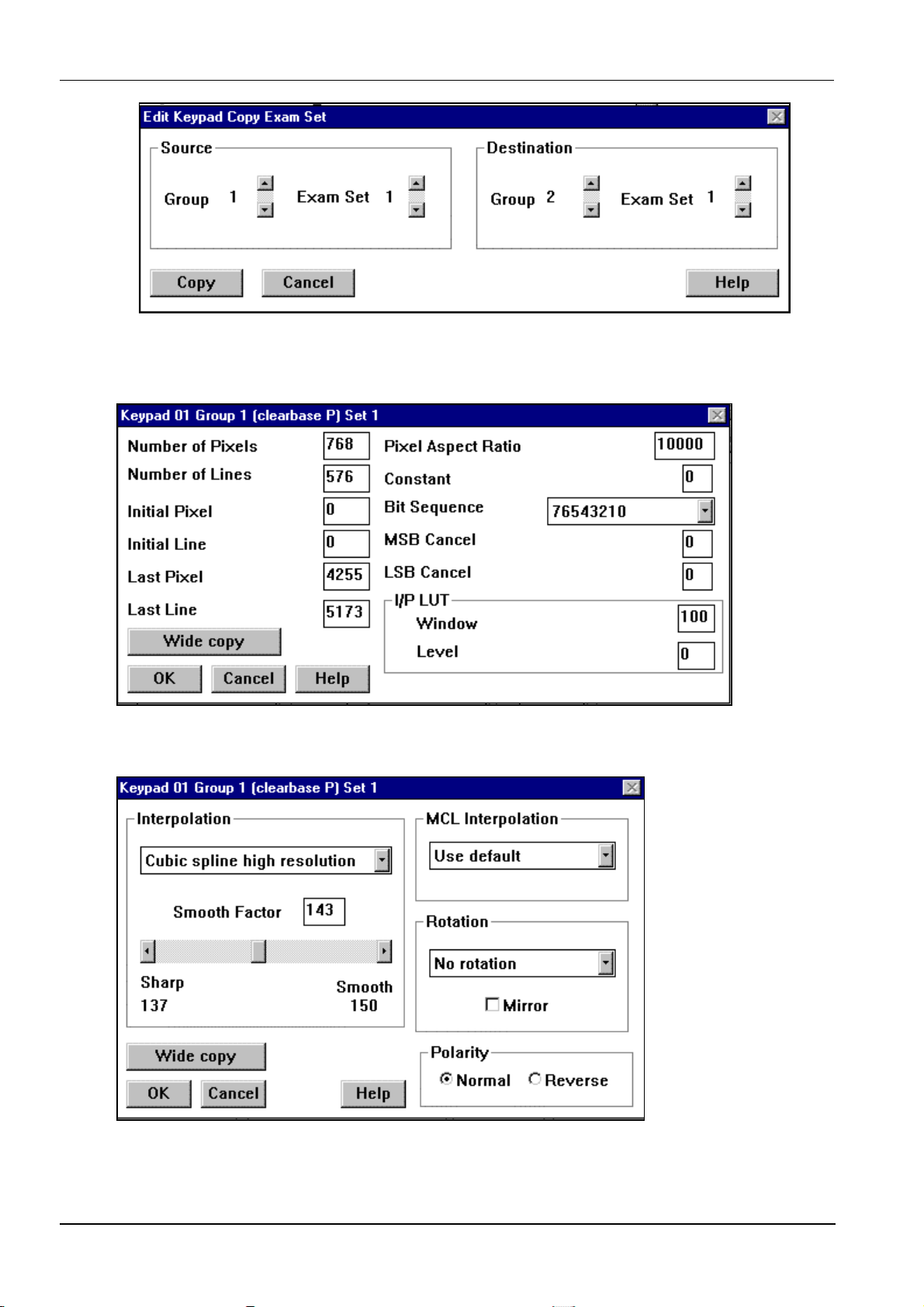
Installation /Machine Start-up DD+DIS221.98E
Figure 22
Selection: <EDIT SOURCES>
Figure 23
Selectio: <EDIT IMAGES>
Figure 24
Section 3 / 16 5361 09.01.00
Page 63

DD+DIS221.98E Installation /Machine Start-up
Selection: <EDIT FILMS>
The actions described above, which have to be performed for each keypad are identical for
each keypad.
4.6 Print a diagnostic image
• Print a diagnostic image from all connected host systems.
• Discuss the image quality with the customer
Figure 25
09.01.00 5361 Section 3 / 17
Page 64

Installation /Machine Start-up DD+DIS221.98E
5 Kick off
5.1 Backup
• In IMOS Drystar 3000, go to "I nstall" → "Backup".
• Select the destination and filename for the backup.
Figure 26
• Explain the operation of the device to the customer.
Section 3 / 18 5361 09.01.00
Page 65

DD+DIS221.98E Installation /Machine Start-up
6 Addendum A: Removing the transport protection
1. This document informs you what should be done after Drystar 3000 is unpack ed.
Do not connect the printer to the mains outlet before you finished all
handling described in this document !
• Take the long screw driver attached to the
accessories (see scope of delivery).
• Unlock the top cover and the cover of the
film loading compartm ent by insert ing the
screw driver in the hole betweeen the grips
of these covers
• Open the top cover first
• Open the cover of the film loading
compartment
536103AM.BMP
• At the inside- right hand side - there is a
floppy holder
• Remove the tape but leave the floppies in
the holder as you normally do not need
these floppies during installation
09.01.00 5361 Section 3 / 19
Page 66

Installation /Machine Start-up DD+DIS221.98E
• With the top cover opened you get access
to the unlocking screws of the side covers
• Turn the screws on top of the left and rig ht
side covers 90 degrees counter clock wise
using a coin or a screw driver
• Lift the covers a few centimeters and then
remove them.
536103BM.BMP
536103CM.BMP
• Cut the plastic strip that secures the film
guiding plate and remove it.
• Cut the 2 plastic strips that secure the
decurl unit and remove them
536103DM.BMP
Section 3 / 20 5361 09.01.00
Page 67

DD+DIS221.98E Installation /Machine Start-up
• Cut the 2 plastic strips that secure the shaft
above the film loading compartment and
remove them.
536103EM.BMP
• Open the film loading compartm ent and
remove the buffer, which lies where
normally the film is put. (not shown on this
picture)
536103FM.BMP
536103GM.BMP
• At the right side of the printer there is a
locking plate on the densitometer module.
• Loose the screw and then remove the plate
Option: Mobile Kit
At the right hand side of the printer with a
mobile kit installed there is a black teflon ring
on the densitometer module.
Do not remove this ring!
09.01.00 5361 Section 3 / 21
Page 68

Installation /Machine Start-up DD+DIS221.98E
• On the left and right side there is a square
hole.
• In that hole there is on both sides a red
coloured blocking plate holding the ther mal
head compartment against the chassis
• Remove on each side the hexagonal screw
and remove the red blocking plate
• Open the thermal head compartment.
536103HM.BMP
• Remove the hexagonal screws on both
sides.
• Close the thermal head compartment and
lock it by turning the 2 screws with the ring
90 degrees clock wise.
536103IM.BMP
• Deposit the screw driver of the accessoryKit in the prepared holder at the lef t side.
• Install the side covers (take care that the
bottom of the side covers is well positioned
over the positioning pins).
• Proceed with the installation.
536103JM.BMP
Section 3 / 22 5361 09.01.00
Page 69

DD+DIS170.99E Installation /Machine Start-up
7 Addendum B:
Mounting the detached flat belt of the main drive
(left hand machine side)
Reason:
For first operation of the machine the flat bel t must be mount ed agai n.
flat belt pulley
If the printer is out of operation for a longer time period (about 6 weeks), the flat
belt of the main drive may suff er permanent pressure marks due t o the belt
tension.
These pressure marks on the flat belt result in irregularit ies of the film transport
system, and thus also in image artef acts (horizontal lines at a spacing of approx.
13 cm).
Therefore, at t he pr oduction line, the flat belt is detached again after the printer
has been calibrated and the belt tension has been mounted at t he belt pulley,
(see figure 1).
detached flat belt
Figure 1 (Addendum B)
7.1 Attachment of the flat belt on the flat belt pulley
• Undo the upper screw in the left side cover (by quarter rot ation).
• Lift the cover and remove it.
• Place the detached belt on the running face of the pulley, by slowly turning the flat belt
pulley by hand.
When the flat belt pulley is turned in order to mount the belt, the motor
reacts like a generator.
Therefore turn t he flat belt pulley only slowly to keep the generator
effect as low as possible.
• Carefully keep the flat belt pulley turning by hand, thus moving the belt to the centre of the
pulley running face.
Subsequent adjustment of the belt tension or calibration of the pr int er
is not necessary.
09.01.00 5361 Section 3 / 23
Page 70

Page 71

DD+DIS103.02E Theory of operation
Section 5
Table of contents
1 INTRODUCTION............................................................................................................... 1
2 FUNCTIONAL SEQUENCE ..............................................................................................4
2.1 Sequence of functions after switching on: SW component startup ................................................4
2.2 Sequence of functions during operation ............................................................................................5
2.2.1 Overall block diagram ......................................................................................................................5
2.2.2 Image Data Flow..............................................................................................................................6
2.3 Sequence of functions during the print cycle.....................................................................................7
3 DESCRIPTION OF THE PRINT PROCESS .....................................................................8
3.1 The principle of thermographic printing .............................................................................................8
3.1.1 General principles of image formation in the printer ........................................................................8
3.2 Thermal head .........................................................................................................................................8
3.2.1 Necessity of the TH-position adjustment .........................................................................................9
3.2.2 Necessity of the TH-pressure adjustment...................................................................................... 10
3.3 Compensation......................................................................................................................................11
3.4 Monomed films ....................................................................................................................................12
4 COMPONENTS............................................................................................................... 13
4.1 Thermal Head Assembly .....................................................................................................................13
4.2 Film Loading Assembly.......................................................................................................................14
4.3 Description of the friction clutches ...................................................................................................15
4.4 Horizontal Film Alignment ..................................................................................................................16
4.5 Main Drive.............................................................................................................................................17
4.6 Macrodensitometer (MDM)) ................................................................................................................19
4.6.1 Film to film Dmax calibration..........................................................................................................20
4.6.2 Film type detection.........................................................................................................................20
4.6.3 Film calibrations at software version 1.7X and 1.9X......................................................................21
4.7 Decurl Unit............................................................................................................................................22
5 HARDWARE OVERVIEW............................................................................................... 23
5.1 Overall Hardware Model ......................................................................................................................23
5.2 VME Bus ...............................................................................................................................................24
5.2.1 Introduction ....................................................................................................................................24
5.2.2 VME BUS Overview.......................................................................................................................24
5.3 ROCAN bus ..........................................................................................................................................26
5.3.1 Introduction ....................................................................................................................................26
5.3.2 Boards............................................................................................................................................26
5.4 Harddisk / Print queue.........................................................................................................................28
5.4.1 Organization of the harddisk ..........................................................................................................28
5.4.2 Organization of the print queue......................................................................................................28
6 SOFTWARE ARCHITECTURE....................................................................................... 29
7 FILE STRUCTURE / AOS SHELL ..................................................................................30
8 APPLICATION ................................................................................................................32
8.1 Overview of all steps of image processing .......................................................................................32
Revision 32 Type 5361 Chapter 5 / I
Page 72

Theory of operation DD+DIS103.02E
8.2 Taste look up table. Combination with Sensolut..............................................................................32
8.3 Window level ........................................................................................................................................33
8.4 INTERPOLATION..................................................................................................................................34
8.5 Printing Areas.......................................................................................................................................36
9 NETWORKING: GENERAL INTRODUCTION ............................................................... 37
9.1 APIP.......................................................................................................................................................39
9.2 DICOM ...................................................................................................................................................39
9.2.1 Introduction ....................................................................................................................................39
9.2.2 DICOM in Combination with APIP..................................................................................................40
9.3 Postscript..............................................................................................................................................42
Chapter 5 / II Type 5361 Revision 32
Page 73

DD+DIS221.98E Theory of operation
1 INTRODUCTION
The printer is used to produce f ilm s ( har d copies) for medical diagnostics. T he pr inter is a dry,
black&white thermo hard-copy system for monomed films. Blue, clear and opaque films can be
handled. Possible film form at s ar e 14 x 17“, 14 x 14“ and 14 x 11“
The printer consists of t wo f unct ional blocks: A
controller and a print engine.
The controller captures the incoming analog or
digital data via an input interface and stor es t hem
onto a hard disk, composes the dif ferent images
and generates the appropriate print engine
control signals.
The print engine receives the image data from
the controller, drives the film t hr ough the device
and makes black/white prints.
Drystar 3000: It can be divided in Controller (VME Compartment) and Print Engine
• The printer can be
equipped with an analog
video input (VSI) or a
digital image input (MFRI).
• It can be host controlled
for printing. It is able to
understand the host
control programs AMDI
(AGFA), USP (Siemens),
MMM (3M) and TCT
(Toshiba.
• In addition, the printer can
be integrated in a
network. Possible
network protocols can be
APIP (AGFA) or DICO M.
Sending images via
network in postscript
description is possible too.
A lot of other image
formats, like sunr ast er or
TIF via E-mail are also
supported.
System Overview Drystar 3000. Example for a network connection.
09.01.00 Type 5361 Section 5 / 1
Page 74

Theory of operation DD+DIS221.98E
t
5361500
3.c
d
Table with possible inputs and correspondi ng i m age i nt erf aces
Signal Interface Max. number
Analog video VSI 3 x B&W or Color*
Digital according to 3M specs MFRI 3*
Ethernet SW interfaces: APIP,
DICOM, FTP, PAP
Theoretically number of inputs without
limit**
and LP
*The maximum number of local inputs is limited to 3, analog or digital.
**In case of DICOM inputs, where to every input a certain configuration has to be assigned, the
maximum number of inputs is limited to 4.
Decurl Unit
• An internal keypad is provided
for command entry, to set up
To p C o v e r
Drum
Filmrun
Thermal Head
the printer, and to display user
and error messages. Up to
three external keypads, one
per local input, can be
connected.
Film Loading Assembly
VME Compartmen
• The system is based on an
automatic sheet handling
system. Films can be loaded
in full daylight.
Filmrun of the printer
• The film path through t he
printer is shown in the sketch
r
beneath.
Section 5 / 2 Type 5361 09.01.00
Page 75
DD+DIS221.98E Theory of operation
Technical data of the printer Drystar 3000 in comparison with other AGFA printers:
Drystar 3000 Drystar 2000 LR5200
Weight 125 kg 70 kg 475 kg
Power
Requirements
100-240 V, 50/60 Hz;
920 W
100-240 V, 50/60 Hz; 1120
W
300 W to 3 kW,
depending on S or P
Version
Throughput: 50/h to 70/h (14x17“)
Density: <0,25 (0,17)......>3.0
<0,25 (0,17).......>3.0 O.D. <0,1......>3,4 O.D.
O.D.
Printing principle: Direct t hermal Direct Thermal / Dye
diffusion
Photochemical via red
laser beam
Calibration: Automatic - manual or semi automatic
Resolution: 320 dpi 300 dpi 320 to 600 dpi
Grey levels 256 out of 4096 256 out of 4096 256 or 4096 out of 65535
Warm- up time: 5 minutes 5 minutes 1 up to approx. 20 min.
with processor
Lifetime of
> 30000 films > 30000 print passes Laser tube > 20.000 h
printhead:
Film Shelf life: 18 months 18 months 24 months
Archivability 30 years 30 years 30 years
Nr. of Inputs 3+x (APIP/DICOM/PS) 1+x (APIP/DICOM/PS) 3+x (APIP/DICOM/PS)
Access time in min. approx. 1 appr ox. 3 ( color) to 1 (b&w) approx. 1
Film formats 14x17, 14x14, 14x11 8x10 8x10, 11x14, 14x14,
14x17
Max. number of
image pixels on a
film and
corresponding
4256 x 5174 (14x17")
→ 22,02 MB
2730 x 2048 (8x10")
→ 5,59 MB x 3 colors =
16,77 MB
8512 x 10348 (10x17"
high res)
→ 88, 08MB
image information
Number of film
formats used
simultaneously
Applications All medical
1 (other formats have
to be installed; 30 min.
required)
applications
11 or 2
US, NM All medical applications.
High quality images
09.01.00 Type 5361 Section 5 / 3
Page 76
Theory of operation DD+DIS221.98E
2 Functional sequence
2.1 Sequence of functions after switching on: SW component startup
Description:
The monitor EPROM, located on the GEMINI CPU star t s up t he
MONITOR shell.
It looks on the harddisk for the operating system (BSP.PRG).
The program BSP.PRG executes the AUTOEXEC.BAT script,
which spawns the SYS (SYStem) program.
This is the base for all the drivers that are necessary for image
processing and film transport.
When all the drivers ar e loaded, the bootup is finished.
This whole bootup takes about 3 minutes.
Bootup Sequence
Section 5 / 4 Type 5361 09.01.00
Page 77
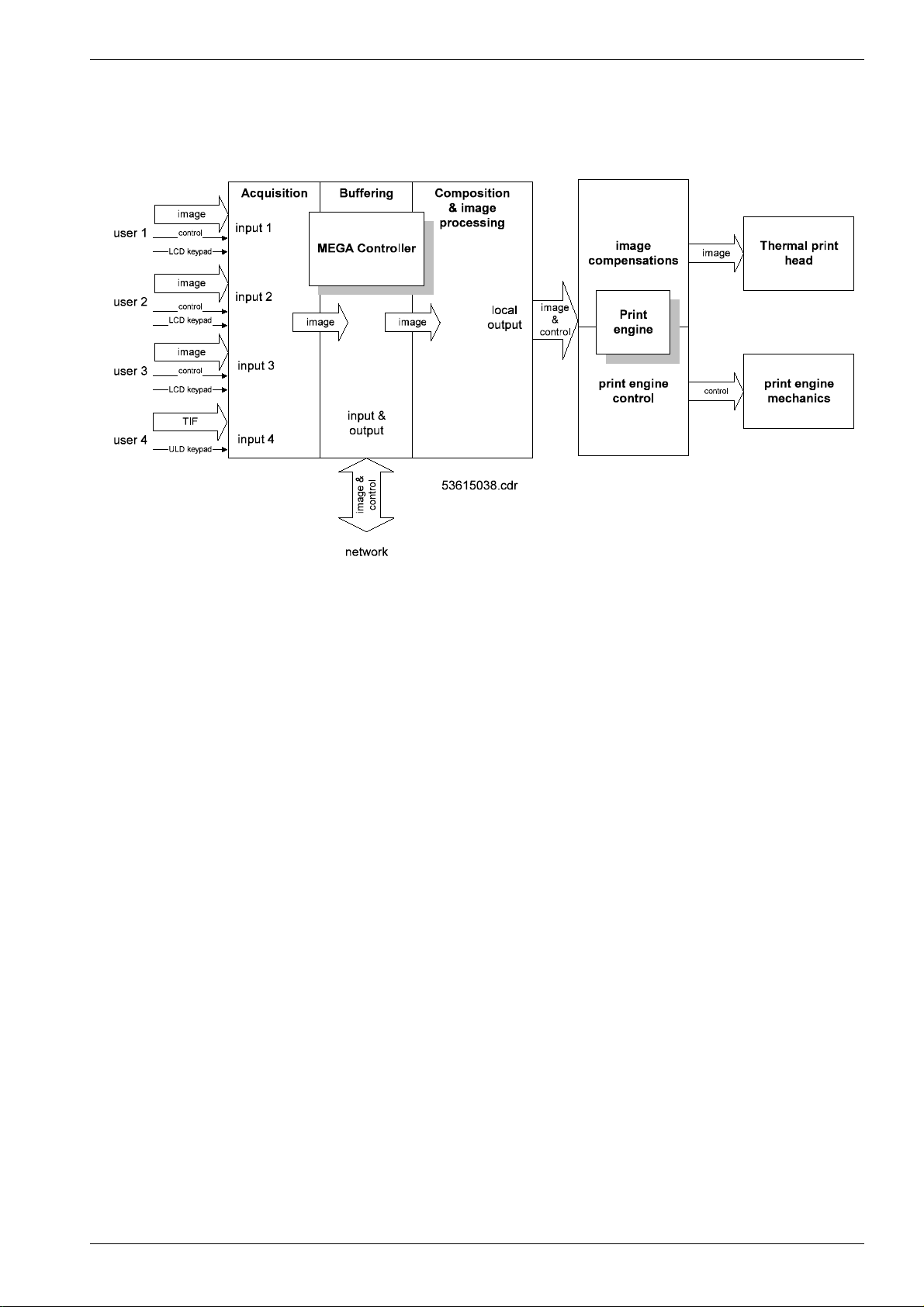
DD+DIS221.98E Theory of operation
2.2 Sequence of functions during operation
2.2.1 Overall block diagram
Functions of the controller:
• capturing of images
• buffering in image queue
• image is processed as define by the user
Functions of the Print engine
a) an image should be rendered independent of
• time, history
• place on the film
• neighbour pixels...
b) TH compensations
• control of engine mechanics
09.01.00 Type 5361 Section 5 / 5
Page 78
Theory of operation DD+DIS221.98E
2.2.2 Image Data Flow
Buffering Composition and CommunicationAcquisition
image
input
image
input
acquisition
acquisition
add
data set
parameters
add
data set
parameters
convert to
internal
format
convert to
internal
format
queue's on HD (F)
queue dest.1
queue dest. 2
convert to
internal
format
image from & to
netwerk
convert to
external
format
add defaults
for
undefined
parameters
image
processing
image
composition
53615039.cdr
image
composition
map on HD
(E)
Acquisition
• ACQ (ACQuisition), acquires images, from one of the inputs
• converts to internal format ( = API P) , using MNU data set parameters like image and film
parameters, ACOM (Accumulation Common Obj ect Module API for image conversion to the
internal format)
Buffering
BMD:
• controls input queues and disk quota for ‘users’,
• images are stored in a folder (film based or study based) and queued (FIFO) on t he har d-disk
partition F
image to
print
engine
• one queue for every destination (local printer, network pr inter)
• recovery of unfinished folders at start up
• each image is put in a 1/0 layout or is deleted (configurable)
Composition & Communication
a) DSP (DiSPatcher),
• reads out and decodes the control and images information from the queue
• adds defaults (dbmin, dbmax, layout, background...) for undefined fields
• organizes the image processing
b) NIP for image processing
c) ‘Image Composition on Disk’ manager uses ICI, to com pose the film-layout and stores it in the
ICM (Image composition map) on disk par t it ion E
d) By CPD images are sent to COS
Section 5 / 6 Type 5361 09.01.00
Page 79

DD+DIS221.98E Theory of operation
2.3 Sequence of functions during the print cycle
09.01.00 Type 5361 Section 5 / 7
Page 80

Theory of operation DD+DIS221.98E
3 DESCRIPTION OF THE PRINT PROCESS
3.1 The principle of thermographic printing
• The film is pressed to the print drum
by the TH
• The drum is rotated causing the film to
move along the TH
• Pixel values of the image are converted
into heating pulses in the therm al
elements of the TH
• The heating pulses are causing the
gray pixels rendering on the thermal
sensitive sheet
• The higher the temperature and the
longer the time an elementary part
(area) of the sheet is heated, the
higher will be the resulting optical
density of that part
3.1.1 General principles of i m age f ormat i on i n t he pri nt er
• The image formation in the pr int engine is based on the completely dry direct thermal technology.
Thermal because the image information is tr ansferred to the TM-film by means of heat and direct
because the heat producing component - the t hermal head(TH) is in direct contact with the TMfilm.
• The heat is generated in the heating elements of the thermal printhead. These heating elements
are arranged in a 14" wide one dimensional area with a spatial freq uency of 320 elements per
inch. The elements can be activated individually.
• During printing, the heating elem ent s ar e activated pixel by pixel and the thermal printhead is
brought into contact with the transpor t ed TM-film.
3.2 Thermal head
The thermal print head is manufactured using a
"thin film" technolog y and consists of the following
components:
• array of 4352 individual resistor elements on
one line, 14 inch, 320 elements per inch
• placed on a glaze bulb
• ceramic substrate supports the glaze bulb
• rigid base plate adds extra mechanical
strength
• electronic circuits for controlling t he heating
of each individual resistor
• TH is mounted on a heat-sink for dissipating
heat to the environment, so that the excess of
heat does not distort the printed image
Section 5 / 8 Type 5361 09.01.00
Page 81

DD+DIS221.98E Theory of operation
3.2.1 Necessity of the TH-position adjustment
The TH position influences the imag e
quality. Image artifacts like "woodiness",
transport and friction shocks and ar tifacts
caused by very small (< 10 µ!)
irregularities in the drum surface can be
noticed. The drawing beneath shows two
different T H posit ions:
Attention: TH position is determined by two pins and adjusted already in production.
The position adjustment f rom pin position to pin position is 0,2 mm (see drawing below)
TH position adjustment
09.01.00 Type 5361 Section 5 / 9
Page 82

Theory of operation DD+DIS221.98E
5
g
3.2.2 Necessity of the TH-pressure adjustment
• In the Drystar3000 printer, the image information is transferred to the film by means of heat which
is generated in the heating element s of the thermal printhead. Therefore, in order to guarantee a
reliable information transfer, a good thermal contact bet ween the T H and t he film is required.
• Since the heaterline of a TH is not perfect ly flat (see figure below), a good t her mal contact along
the thermal head is only possible if the TH is pressed har d enough against the film and the dr um.
• During production, the “TH-pressur e” is adjusted to 300 g/cm (≈ 2.5 g/elem ent ) . It cannot be
adjusted in the field.
• If on the other hand the pressure bet ween the T H and t he film becomes too high, the T H’s
passivation layer can be damaged by mechanical wear or by the passage of a dust particle
between the TH and the film. A too hig h pressure therefore can lead to the ear ly appearance of
TH-defects (like e. g. broken resistors).
mounted TH
unmounted TH
0,45
0,40
0,35
120µm
Flatness (mm)
0,30
0,25
0
5
10
53615015.cdr
Position alon
15
20
25
TH
30
3
Section 5 / 10 Type 5361 09.01.00
Page 83

DD+DIS221.98E Theory of operation
3.3 Compensation
During printing, the print engine must continuously compensate for a number of external (= non data
related) influences that otherwise would introduce density changes (and im age artifacts).
There is e.g. a very close and
strong relationship between the
system sensitometry and different
temperatures around the print head
(drawing see beneath).
The most important compensat ions ar e:
• The microdensitometer compensation which compensates f or the differences in heat
production of diff erent resistor elements in the therm al printhead. The scanning is done
with a resolution of 2000 dpi. Like that the obtained density for each resistor element is
known.
• The temperature compensation which compensates f or the changes in ambient and
heatsink temperature and for the heat accumulation in the thermal printhead during
printing (see drawing above).
• The sensitometric compensation which compensates for the differences in sensitometric
behaviour between different TM-production batches.
• The voltage drop compensation.
• The previous and neighbour compensation
• The copy-to-copy Dmax compensation.
09.01.00 Type 5361 Section 5 / 11
Page 84

Theory of operation DD+DIS221.98E
3.4 Monomed films
A cross section through a monomed sheet is repr esented below:
The image forming layer of AgOS is heat sensitive but not light sensitive.
By influence of heat (100° - 200° C) the silver (Ag) is reduced from AgO S:
heat (100°-200° C)
AgOS + reduction è Ag + (image information).......
Section 5 / 12 Type 5361 09.01.00
Page 85

DD+DIS221.98E Theory of operation
4 Components
4.1 Thermal Head Assembly
Function:
• Guarantee steady and continuous pressure of the T H t owards the film
• Film transport
Adjustments:
TH pressure adjustment
Note:
We disting uish 3 TH positions. In preprint
position the TH rollers are not yet leading
the film. As soon, as the film is in
position of the TH rollers, t hey press t he
film towards the drum and the following
film guide (sheet m etal).
09.01.00 Type 5361 Section 5 / 13
Page 86

Theory of operation DD+DIS221.98E
4.2 Film Loading Assembly
Function:
• Film separation
• Driving film to TH
• Horizontal film alignment (together with motor M8)è Motor M8 drives film towards stop in input
tray. See drawing beneath.
Adjustments: Vertical film alignment (position the st op further up / down).
Note: For the function of the clutch see description below: Function of the fricti on clutches.
Section 5 / 14 Type 5361 09.01.00
Page 87

DD+DIS221.98E Theory of operation
4.3 Description of the friction clutches
Friction Clutches: Example of function in input tray
• Altogether 3 friction clutches ar e built in (film loading assembly; vertical film alignment, sheet ta ke
over motor).
• Function 1: Opening of the rollers if motor drives backward.
• Function 2: Guarantee a fixed torque of 6 Ncm
09.01.00 Type 5361 Section 5 / 15
Page 88

Theory of operation DD+DIS221.98E
4.4 Horizontal Film Alignment
Functions:
• Horizontal film alignment
Adjustments:
After exchange of this unit, the horizontal position of the horizontal film alignment has to be adjusted.
Note: For the function of the clutch see description below: Function of the fricti on clutches.
Section 5 / 16 Type 5361 09.01.00
Page 89

DD+DIS221.98E Theory of operation
4.5 Main Drive
Functions:
• The maindrive drives the drum in constant speed.
• It calibrates after switch on (only if switch of f time > 15 min.) or if t he m achine was idle f or more
than 1 hour
• It also calibrates the drum speed during print ing
• Optical position encoder (3000 pulses per revolution) controls main drive motor
• Closed loop control
Adjustments: none
09.01.00 Type 5361 Section 5 / 17
Page 90

Theory of operation DD+DIS221.98E
Maindrive Control Principle
Section 5 / 18 Type 5361 09.01.00
Page 91

DD+DIS103.02E Theory of operation
4.6 Macrodensitometer (MDM))
A Macro Density Measurement system (MDM) is built-in to allow for constant image quality
concerning Dmin, Dmax, sensitometry and vertical banding.
Functions:
· Automatic film to film Dmax calibration (refer to 4.6.1, page 20)
· Automatic film type detection (after the printout!) (refer to 4.6.2, page 20)
· Film calibrations of the printer (refer to 4.6.3, page 21)
· reduction of the vertical macro density banding (density variation along the width of a TH)
(refer to 4.6.3, page 21)
· calibrating the MDM to an external density-meter, by means of a 'reference' film (refer to
4.6.3, page 21)
Adjustments:
No mechanical adjustments. For calibrations Refer to section 6.6, calibrations and adjustments.
Note: Details of the functions mentioned above depend on the SW release (SW Rel. 1.7X or 1.9X).
Refer to the table in section 4.6.3.
Revision 32 Type 5361 Chapter 5 / 19
Page 92

Theory of operation DD+DIS103.02E
4.6.1 Film to film Dmax calibration
· The 'film-to-film Dmax calibration' is automatically
performed after every print, if it is switched on. The
filmrun stops, the MDM carriage moves towards the film
and measures the maximum density at position 12 mm
from the right and 90 mm from the bottom.
· At SW Rel. 1.7X the film-to-film calibration is switched on
by default, at SW Rel. 1.9X it is switched off by default.
Refer to section 6.6 calibrations for more information.
· It keeps the 'border density' (from film to film) equal to a
'reference density' during printing.
~12 mm
~
Film to film
Dmax
~90 mm
~
calibration
measurement
· Every 'max.-density calibration' defines a new 'reference
density' (= new start point)
· The 'reference density' is defined by the average border
density of the first 5 printouts after the last max.-density
calibration
536105AE.CDR
· The film to film calibration reduces the average of a
'gradual density variation' over more than 5 printouts
4.6.2 Film type detection
~5 mm
~
Film type
detection
~90 mm
~
measurement
· With SW Rel. 1.7X the 'film type detection' is
automatically performed after every print. The filmrun
stops, the MDM carriage moves towards the film and
measures the fog level at position approx. 5 from the right
and 90 mm from the bottom.
· Like that after every print the film type blue base or clear
base is automatically detected.
· At SW Rel. 1.7X the film type detection is switched on by
default, at SW Rel. 1.9X it is switched off by default. This
cannot be altered with software parameters.
536105AF.CDR
Chapter 5 / 20 Type 5361 Revision 32
Page 93

DD+DIS103.02E Theory of operation
536110AO.C
536110
C
53616
6
C
536
0
C
4.6.3 Film calibrations at software version 1.7X and 1.9X
Calibration Purpose When to be performed How to be performed
in SW Rel. 1.7X
Registration *
DR
Reference film
Density Meter *
TH profile *
DR
AI.
Reduces the mechanical misregistration of the MDM carriage during
a film scan
To determine new reference values for
the MDM. This is done via an external
densitometer.
The default reference film density
536110AK.CDR
setting obtained at production is used in
case this adjustment is not performed.
Calibrate the MDM. Compensate its
long term drift. The MDM step wedge
reference film delivered with the printer
has to be entered and will be measured.
536110AK.CDR
To reduce vertical macro banding
(vertical density irregularities). Only
macro density compensation is possible
(in opposite to micro compensation in
production) due to the measurement
resolution of about 5 pixels.
· whenever a TH profile calibration is not satisfying
· first calibration in case of MDM exchange
· when the printer to printer density variation has to
be kept small (has to be done at both printers!)
· via local keypad
· via IMOS Drystar 3000
· via local keypad
· via IMOS Drystar 3000
· when an absolute density accuracy is important
(compensate for deviations of the reference film)
· when the system should be calibrated to the „user
densitometer“.
· at the maintenance, i.e. at least once a year · via local keypad
· via IMOS Drystar 3000
· at the maintenance, i.e. at least once a year
· whenever the vertical banding is not acceptable
· via local keypad
· via IMOS Drystar 3000
anymore.
· whenever the max. density variations of a
"maximum density test film" are > 0.2 O.D.
How to be performed
Remarks reg. SW Rel. 1.9X
in SW Rel. 1.9X
· via IMOS Drystar 3000 · At printers equipped with SW Rel. 1.9X the registration
calibration can only be performed via IMOS Drystar 3000.
· via IMOS Drystar 3000 · At printers equipped with SW Rel. 1.9X the reference film
calibration can only be performed via IMOS Drystar 3000.
· via IMOS Drystar 3000 · At printers equipped with SW Rel. 1.9X the MDM calibration
can only be performed via IMOS Drystar 3000.
· via local keypad
· via IMOS Drystar 3000
· In SW Rel. 1.9X automatic thermal head calibration is active
(by default). Requested every 5000 prints by means of a
warning message on the local display (i.e. it's only semiautomatic, as the customer has to initiate it)..
· Additionally, the user is able to perform a manual calibration.
· The automatic calibration cycle can only be altered or
switched off via NVF parameter.
Maximum density
DR
AP.
Set the Dmax of the printer to its default
(default = 3.0 O.D.). The lowest density
value of a test film has to be entered.
The printer will automatically correct its
thermal energy to reach Dmax = 3.0.
It also can be used for calibration of
opaque film (no internal densitometer
· With SW REL. 1.7X: After insertion of a new film
batch (new emulsion number) when the Dmax of
3.0 O.D. cannot be reached after 5 films
· With SW REL. 1.7X: Before every film sensito
calibration.
· via local keypad
· via IMOS Drystar 3000
functionality available for opaque)
Film Sensito *
To measure the film sensitometry and
to use these values for internal
reference, so that the printer behaves
· Whenever a new filmbatch was inserted · via local keypad
· via IMOS Drystar 3000
linear regarding density reproduction.
DR
AE.
11
*Note: These calibrations are made by a full width scan at about 15 mm from the bottom. Dust on the film will influence the result.
· via IMOS Drystar 3000 · In SW Rel. 1.9X the Dmax calibration is combined with
sensito calibration,. The target Dmax is 3.1.
· In SW Rel. 1.9X it still can be performed via IMOS Drystar
3000, it is however not required anymore.
· via local keypad
· via IMOS Drystar 3000
· In SW Rel. 1.9X the sensitometry is automatically initiated
after insertion of a new film package. This can be deactivated at the local display or via NVF parameter.
· In SW Rel. 1.9X, in case the reached Dmax level after a film
sensito calibration is below the target Dmax
(target Dmax = 3.10), a warning message is displayed
'Dmax= X.YZ'. In this case a new film sensito calibration is
recommended. In case the target Dmax cannot be reached,
printing can continue nevertheless.
Revision 32 Type 5361 Chapter 5 / 21
Page 94

Theory of operation DD+DIS103.02E
4.7 Decurl Unit
Function:
· Decurl of the film.
· Heating element 450 W
· Remote temperature sensor
· Temperature = 90°C
Adjustments: none
Decurl Temperature Control Principle:
Chapter 5 / 22 Type 5361 Revision 32
Page 95

DD+DIS221.98E Theory of operation
5 Hardware overview
5.1 Overall Hardware Model
Description: The hardware components can be split up in
• VME BUS and
• ROCAN Bus.
The Gemini CPU is the main CPU. The BER interface, located on the Ge m ini CPU is t he interface
between VME Bus and ROCAN Bus.
The VME interfaces are responsible for image acquisition (VSI / MFRI) and image processing (COS).
The ROCAN interfaces are r esponsible for the filmrun.
09.01.00 Type 5361 Section 5 / 23
Page 96

Theory of operation DD+DIS221.98E
5.2 VME Bus
5.2.1 Introduction
• Versa M
odule Eurocard Bus
• 32 bit BUS
• Developed by Motorola, Signetics, Mostek and
Thompson CSF
5.2.2 VME BUS Overview
• Widely used in industrial, milit ar y, industrial
applications
• Supply voltage: 5 V Boards
Board
Gemini CPU board
NIP Neuron Image
Processor. Image
Interpolation
BER Basic Ethernet and
ROCAN Interface
Description
• 68040 microprocessor, 25 MHz
• Up to 128MB RAM (32 MB default)
• Real time clock
• SCSI bus interface
• VME bus controller
• Socket for 2 nd 68040 microprocessor
• NIP board installed by default
• Gemini CPU plug-on board
• Performs the image scaling (reduction / magnification)
• Gemini CPU plug-on board
• drives internal hard disk 2 Gbyte
• drives external floppy
• drives external ethernet
• has the function of the rocan interface to the VME
• drives external serial port (2x)
RAM 24MB-DRAM-
• Gemini CPU plug-on board
Section 5 / 24 Type 5361 09.01.00
Page 97

DD+DIS221.98E Theory of operation
Memory
COS1 Compensation
and Superslicing.
Compensation for TH
VSI Video Synapsis
Interface with SWB2
SWB2 Switchboard2.
and AFR Analog Fibre
Receiver
MFRI Multi format
receiver interface
according to 3M Specs
SCN2 Serial control
interface (node) RS232 /
RS422. Host control
interface.
• Keeps the program
• Buffer between image interface an harddisk
• Performs the compensations f or t he TH
Cable connections:
− VME interface (backplane) → COS1
− COS1 50 pins flatcable →AIC1
− AIC1 64 pin flatcable →TH
• AFR converts fiber optical (analog) data to analog data
• SWB multiplexes up to 4 analog sour ces ( e.g. 3 x color + 1 b&w) to the
VSI
• The VSI converts the analog date to raw, digital im age data
• The VSI is a selfadjusting video interf ace
• Captures digital images and converts them to r aw, dig it al image data
• Has a built in RS422 interface for host cont r ol connection
• Interface for host contr ol connect ion.
• RS232 used for connections up to 18 m
• RS422 used for distances up to 1000 m
GPCI Generic Parallel
Control Interface. Host
control interface.
MAU2 Media access
unit. Part2 of Ethernet
Interface (BER!)
Backplane
BIO3*
Basic I
Power supply*
nput / Output
• Interface for host contr ol connect ion.
• By a host specific EPROM the host control signals are converted to
APIP
• Ethernet interface
• Jumper setting necessary to switch from 10B2 connection to twisted
pair.
• VME interconnection board
• Distributes the 5V to the diff e r ent VME boards
• Global wiring board for optional interf aces: (SCN, GPCI, MAU)
• VME fan control, shut down when temp 50° C (warning when 45°C)
• Reset logic for rocan interf ace
• Power supply remote voltage control thermal head
• Power supply remote voltage decurl unit
• Switch mode power supply
• Autorange 110 - 240 V / 50 - 60 Hz
• Output voltages: +5V / 36 A; +12 V / 4A; -12V / 2A; +24V / 6A; 60V /
400 W (decur l) , Vth from 17 till 22 V / 25 A max; 8,5V / 2A (ext. keypad
+ ROCAN)
DTS*
• Contains thermal sensor to measure temperat ure around drum and
VME rack. Two of these boards built in altog ether.
*These boards cannot be assigned to the VME Bus explicitely. They are part of the ROCAN Bus, too.
09.01.00 Type 5361 Section 5 / 25
Page 98

Theory of operation DD+DIS221.98E
5.3 ROCAN bus
5.3.1 Introduction
ROCAN = ROAM (AGFA R&D Department) C
ontroller Area Network
General Purpose: Data communication in distributed systems
CAN technical aspects:
• max. speed 1MBs/40m
• Mail oriented protocol: Every node sends and receives mails. Mails can have Priorities.
• Multimaster: Every node is a bus master.
• max. dataframe=8byte
• Systemwide data consistency
• Applications: Production, cars, buildings, eng ines
5.3.2 Boards
PMC1
KEY3
53615037.cdr
to VME
BIO3
Power
BER
Board Description
PMC1 Precision Motor
Control Board
MDM1
DSH1 AIC1
• Drum motor control, optical position encoder, quadrature decoding,
calibration, compensation of gear ration
• Decurl motor control, decurl temperat ur e cont rol, active/passive
protection, decurl heater element cont r ol through power supply
• Pick up motor control, input f eeder control
• Sheet take over motor control, sheet alignment solenoid
• Vertical alignment motor
• Cover locking solenoid
• Thermal head fan control, cooling
• Thermal head position control, home, print , preprint
• Al input sensors, paper position, empty cassette
• 3 external user keypads
• Safety circuit, cover switch, decurl overtemperature switch
Section 5 / 26 Type 5361 09.01.00
Page 99

DD+DIS221.98E Theory of operation
KEY3 Internal Keypad
AIC1 Analog Interface with
ROCAN
MDM1 Macrodensitometer
with DSH1.
• LCD display with LED backlight
• 4 lines, 20 characters/line
• Remote power on/off switch standby
• 4 cursors
• 4 control keys (ESC, ENTER, ONLINE, UNLOCK)
• 3 yellow LED's
• 1 green power on LED
• buzzer
• Thermal head voltage control, Vth measurem ent
• Thermal head heatsink temperatur e sensor s ( 5x) r eading
• thermal head resistance measurement
• Drum environment temperature measurement
• Data interface between COS and TH. Converts differential data to
single ended. Data multiplexing.
• DSH board: lamp control, light measurem ent system ( int egrator),
interface with MDM board
• MDM board: Stepper motor control, AD converter, ROCAN bus
interface
09.01.00 Type 5361 Section 5 / 27
Page 100

Theory of operation DD+DIS221.98E
5.4 Harddisk / Print queue
5.4.1 Organization of t he harddisk
5.4.2 Organization of t he print queue
The organization of the print queue works with the FIFO principle: first job in t he queue is the first job
out. In a network, every destination has its own queue. In t he queue there are always several output
jobs activated at the same time. The q ueue is locat ed on t he F: partition.
Job status
P Defines printable jobs that are: ready for printing or
currently printed
W Stands for warning which means that the respective output station is not r eady, e. g. because of
wrong setting. The job can be converted into P j obs as soon as the output is ready.
E Means that there was an error during the processing. The job cannot be completed. The data is
lost and can be deleted.
R Means that this is a remote job, sent via ether net from another user. This j ob can only be deleted
from the user it was sent fr om .
Emergency (+)
Normally, films are always added to the end of a q ueue waiting for the output. If there is an
emergency film which requires immediate exposure of a film, this can be moved up to t he first
position of the queue by pressing the em er gency key on the keypad. Further emergency films are
positioned behind the former emer gency film. The active jobs in the queue, ( P- jobs) however, remain
active and will be processed before the first emergency film.
Job owner
- <Space> self
- 1; 2; or 3 one of the other inputs (not self)
- N network
Film type
b = blue based, i. e. blue acceptor should be inserted
c = clear based, idem for clear
o= opaque, idem for opaque
Section 5 / 28 Type 5361 09.01.00
 Loading...
Loading...