Page 1

Isolibrium™ Support Surface
Integrated with InTouch® FL27 (2131 / 2141 / 2151 / 2152)
version 4.0
2971
2972
Operations Manual
International - EN | FR
2017/08 C.3
2971-209-001 REV C
www.stryker.com
Page 2

sample text
Page 3
Symbols
~
Refer to instruction manual/booklet
Operating instructions/Consult instructions for use
General warning
Caution
Warning; electricity
Alternating current
Catalogue number
English
EN
IPX4
87VL
Serial number
Manufacturer
Date of manufacture
Safe working load
Mass of equipment
Protective earth terminal
Protection from liquid splash
Defibrillation proof type B applied part
Medical Equipment recognized by UL LLC with respect to electric shock, fire, and mechanical
hazards in accordance with ANSI/AAMI ES60601-1:2005/(R)2012, A1:2012, C1:2009/(R)2012,
A2:2010/(R)2012, and CAN/CSA-C22.2 No. 60601-1 (2008)
In accordance with European Directive 2012/19/EU on Waste Electrical and Electronic
Equipment, this symbol indicates that the product must not be disposed of as unsorted
municipal waste, but should be collected separately. Contact your local distributor for disposal
information.
www.stryker.com 2971-209-001 REV C
Page 4

English
EN
Symbols
Wash by hand
Do not tumble dry
Do not dry-clean
Do not iron
Allow to completely air dry
Chlorinated bleach
For US Patents see www.stryker.com/patents
Pressure redistribution
Moisture management, Low Air Loss
Lateral rotation
Shear
Turn assist
2971-209-001 REV C www.stryker.com
Page 5

Isolibrium™ Support Surface
Integrated with InTouch® FL27 (2131 / 2141 / 2151 / 2152)
version 4.0
2971
2972
Operations Manual
2017/08 C.3
2971-209-001 REV C
www.stryker.com
Page 6

sample text
Page 7
Symbols
~
Refer to instruction manual/booklet
Operating instructions/Consult instructions for use
General warning
Caution
Warning; electricity
Alternating current
Catalogue number
English
EN
IPX4
87VL
Serial number
Manufacturer
Date of manufacture
Safe working load
Mass of equipment
Protective earth terminal
Protection from liquid splash
Defibrillation proof type B applied part
Medical Equipment recognized by UL LLC with respect to electric shock, fire, and mechanical
hazards in accordance with ANSI/AAMI ES60601-1:2005/(R)2012, A1:2012, C1:2009/(R)2012,
A2:2010/(R)2012, and CAN/CSA-C22.2 No. 60601-1 (2008)
In accordance with European Directive 2012/19/EU on Waste Electrical and Electronic
Equipment, this symbol indicates that the product must not be disposed of as unsorted
municipal waste, but should be collected separately. Contact your local distributor for disposal
information.
www.stryker.com 2971-209-001 REV C
Page 8

English
EN
Symbols
Wash by hand
Do not tumble dry
Do not dry-clean
Do not iron
Allow to completely air dry
Chlorinated bleach
For US Patents see www.stryker.com/patents
Pressure redistribution
Moisture management, Low Air Loss
Lateral rotation
Shear
Turn assist
2971-209-001 REV C www.stryker.com
Page 9

Table of Contents
Warning/Caution/Note Definition ............................................................................................................... 1-2
Summary of safety precautions........................................................................................................... 1-2
Introduction........................................................................................................................................... 1-4
Product description........................................................................................................................... 1-4
Intended use ................................................................................................................................... 1-4
Expected service life......................................................................................................................... 1-4
Contraindications ............................................................................................................................. 1-5
Specifications.................................................................................................................................. 1-5
Contact information .......................................................................................................................... 1-6
Serial number location ...................................................................................................................... 1-6
Date of manufacture......................................................................................................................... 1-7
Product illustration............................................................................................................................ 1-8
Isolibrium features............................................................................................................................ 1-8
Installation .......................................................................................................................................... 1-10
Operation ........................................................................................................................................... 1-13
Applying the linens ......................................................................................................................... 1-13
Positioning a patient on the support surface......................................................................................... 1-13
Managing incontinence and drainage ................................................................................................. 1-14
Activating and resetting CPR ............................................................................................................ 1-14
Relocating the support surface ......................................................................................................... 1-15
Transferring of patient from one patient support platform to another ......................................................... 1-16
Storing the product ......................................................................................................................... 1-16
Cleaning............................................................................................................................................. 1-17
Disinfecting......................................................................................................................................... 1-18
Preventive maintenance ........................................................................................................................ 1-19
Cover replacement, top ................................................................................................................... 1-20
Quick reference replacement parts.......................................................................................................... 1-22
EMC Information .................................................................................................................................. 1-23
Warranty ............................................................................................................................................ 1-27
Warranty exclusion and damage limitations ......................................................................................... 1-27
To obtain parts and service .............................................................................................................. 1-27
Return authorization........................................................................................................................ 1-27
Damaged product........................................................................................................................... 1-28
International warranty clause ............................................................................................................ 1-28
English
EN
www.stryker.com 2971-209-001 REV C 1-1
Page 10

Warning/Caution/Note Definition
English
EN
The words WARNING, CAUTION, and NOTE carry special meanings and should be carefully reviewed.
WARNING
Alerts the reader about a situation which, if not avoided, could result in death or serious injury. It may also describe
potential serious adverse reactions and safety hazards.
CAUTION
Alerts the reader of a potentially hazardous situation which, if not avoided, may result in minor or moderate injury to the
user or patient or damage to the product or other property. This includes special care necessary for the safe and
effective use of the device and the care necessary to avoid damage to a device that may occur as a result of use or
misuse.
Note: Provides special information to make maintenance easier or important instructions clearer.
Summary of safety precautions
Carefully read and strictly follow the warnings and cautions listed on this page. Service only by qualified personnel.
WARNING
• Risk of Shock. This product must only be connected to a supply mains with protective earth. Grounding reliability
can be achieved only when a hospital grade receptacle is used.
• Explosion risk. This product is not suitable for use in the presence of flammable anesthetic mixture with air or with
oxygen or nitrous oxide other than nasal, mask type, or half bed-length tent type.
• Do not move the support surface without assistance.
• Do not operate the support surface without the Isolibrium top and bottom covers and linens.
• Always position the support surface on the InTouch bed frame to that the support surface does not contact the
headboard.
• Do not use when gaps between the support surface and the bed frame are present to prevent the risk of
entrapment. Only use the Isolibrium support surface with InTouch bed frames.
• Do not attach the power cord to any moving parts of the InTouch bed frame. Improper routing of the power cord
could cause the risk of electric shock, pinching the power cord, or interfere with bed functions.
• Always confirm proper functionality of the scale system after you install the support surface to prevent product
malfunction.
• Do not use extension cords with Isolibrium. It is only intended to be powered by InTouch with the power cord
supplied.
• Always use the power source provided by the InTouch foot end auxiliary power outlet with Isolibrium.
• Always make sure that the operator has access to the CPR straps.
• Do not stick needles into a support surface through the support surface cover. Holes may allow body fluids to enter
the inside (inner core) of the support surface and could cause cross-contamination, product damage, or product
malfunction.
• Do not use fitted sheets with this support surface.
• Always center the patient on the support surface. Align the patient’s head toward the headboard before starting
functions. Check the patient frequently to make sure that you maintain the proper positioning.
• Always make sure that the tubing and wiring that is connected to the patient is long enough, stable, and secure
during Lateral Rotation or Turn Assist.
• Always raise all of the InTouch bed siderails before starting Turn Assist or Lateral Rotation functions.
1-2 2971-209-001 REV C www.stryker.com
Page 11

Warning/Caution/Note Definition
Summary of safety precautions (Continued)
WARNING (CONTINUED)
• Do not exceed the safe working load of the Isolibrium support surface. Excess weight could cause unpredictable
safety and performance of this system.
• Always use extra caution when reading radiology images taken of a patient on a support surface because internal
components can cause artifacts and distort readings.
• Always monitor the patient condition at regular intervals for patient safety.
• Do not use the support surface as a transfer device to avoid the risk of patient injury.
• Do not immerse the support surface or foot box in cleaning or disinfectant solutions.
• Do not allow fluid to pool on the support surface. Fluids may cause degradation of components and may cause
unpredictable safety and performance of this product.
• Always inspect the support surface covers (top and bottom) for tears, punctures, excessive wear, and misaligned
zippers as you clean the covers. If the cover is compromised, remove the support surface from service immediately
and replace the cover to prevent cross-contamination.
• Always perform preventive maintenance based on the usage level of the product. An increase in usage which may
include more frequent cleaning and disinfection, may adversely affect the life of the support surface.
• Always disinfect the support surface between patients, to avoid the risk of cross-contamination and infection.
• Always make sure that you wipe each product with clean water and thoroughly dry each product after cleaning or
disinfecting. Some cleaning and disinfecting agents are corrosive in nature and may cause damage to the product if
you use them improperly. If you do not properly rinse and dry the product, a corrosive residue may be left on the
surface of the product that could cause premature degradation of critical components. Failure to follow these
cleaning or disinfecting instructions may void your warranty.
• Always unplug the product power cord before cleaning or disinfecting to avoid the risk of shock.
• Do not service or perform maintenance while the product is in use.
English
EN
CAUTION
• Improper usage of the product can cause injury to the patient or operator. Operate the product only as described in
this manual.
• Do not modify the product or any components of the product. Modifying the product can cause unpredictable
operation resulting in injury to patient or operator. Modifying the product also voids its warranty.
• Do not allow sharp objects to come into contact with the support surface that could puncture, tear, or cut the cover.
• Do not allow sharp edges from the X-ray plate to come in contact with the support surface cover. The
recommendation is you cover the X-ray plate with a pillow case or other device before placement under the patient.
If damaged, remove the support surface cover from service immediately to prevent cross contamination.
• Do not iron, dry-clean, or tumble dry the support surface covers.
• Do not power wash the support surface.
• Always completely dry the support surface covers before storing, adding linens, or placing a patient on the surface.
• Do not over expose the covers to higher concentration disinfectant solutions as these may degrade the covers.
• Do not use accelerated hydrogen peroxides or quaternaries that contain glycol ethers as they may damage the
cover or the CPR strap print.
www.stryker.com 2971-209-001 REV C 1-3
Page 12

Introduction
English
EN
This manual assists you with the operation or maintenance of your Stryker product. Read this manual before operating
or maintaining this product. Set methods and procedures to educate and train your staff on the safe operation or
maintenance of this product.
CAUTION
• Improper usage of the product can cause injury to the patient or operator. Operate the product only as described in
this manual.
• Do not modify the product or any components of the product. Modifying the product can cause unpredictable
operation resulting in injury to patient or operator. Modifying the product also voids its warranty.
Notes
• This manual is a permanent part of the product and should remain with the product even if the product is sold.
• Stryker continually seeks advancements in product design and quality. This manual contains the most current
product information available at the time of printing. There may be minor discrepancies between your product and
this manual. If you have any questions, contact Stryker Customer Service or Technical Support at 1-800-327-0770.
Product description
Isolibrium™, Stryker model 2971/2972, is a powered support surface with features that provide Pressure Redistribution,
Low Air Loss, and Lateral Rotation. Isolibrium is for use with the InTouch® (revisions 4.0 or higher software) bed frame.
Isolibrium and InTouch are an integrated bed and surface system that caregivers use to assist in patient care.
Intended use
The Isolibrium support surface assists in the prevention and treatment of all pressure injury stages (including stages 1,
2, 3, 4, Unstageable and Deep Tissue Pressure Injury). Before implementing Isolibrium, a healthcare professional
should complete a clinical evaluation of risk factors and skin assessments. The Isolibrium patient-specific immersion to
assist in managing microclimate of the patient skin, patient repositioning, early mobility, and the prevention of pulmonary
complications.
The InTouch and Isolibrium system is for use by patients in an acute care setting. The acute care settings may include
critical care, step down, progressive care, medical / surgical, sub-acute care, and post anesthesia care unit (PACU), or
other locations as prescribed. The patient, accessories, and any extra loading must not exceed the safe working load of
InTouch or Isolibrium.
The operators for the system are healthcare professionals (nurses, nurse aids, and doctors) who can use all system
operations (for example: bed motion functions, nurse call, siderail communications, bed exit, or therapy options).
Patients and bystanders can use the bed motion functions, nurse call, and siderail communications but are restricted
from the use of the powered support surface functions. Trained professionals install, service, and calibrate the InTouch
and Isolibrium system.
Expected service life
Isolibrium support surface has a five year expected service life under normal use, conditions, and with appropriate
periodic maintenance.
Isolibrium covers (top and bottom) have a two year expected service life under normal use, and conditions.
1-4 2971-209-001 REV C www.stryker.com
Page 13

Introduction
Contraindications
Stryker promotes the clinical assessment of each patient and appropriate usage by the operator.
The InTouch and Isolibrium system is not intended:
• for use with patients 35 inches or less in height
• for use with patients that weigh less than 50 pounds
• for use with a full oxygen tent
• to support more than one individual at a time
• for use with a patient less than two years old
• in a home healthcare environment
• as a sterile product
• for use in the presence of flammable anesthetic mixture with air rich in oxygen or nitrous oxide
Note: When Isolibrium is on the InTouch bed, a half-oxygen tent within the sidreails is allowable. Do not enclose
any part of the support surface foot box with the half-oxygen tent.
Isolibrium is not intended for use on patients with the following:
• unstable fractures
• unstable spinal cord injuries
Isolibrium Lateral Rotation and Turn Assist features are for use only after you complete a clinical assessment. This is to
make sure that no adverse reaction would result in patients with the following:
English
EN
• spinal cord injury
• skeletal traction
• significant hemoptysis
• bleeding disorders
• rib fractures
• for whom the techniques cause increased dyspnea or wheezing
• hemodynamic instable
• increased intracranial pressure
• skeletal traction
Specifications
Model 2971/2972 Isolibrium
Length 84 in. 213,4 cm
Width 35 in. 88,9 cm
Thickness 8 - 10.5 in. 20.3 - 26.7 cm
Weight 90 lb 40.8 kg
Therapeutic weight 50 lb to 350 lb 22.7 kg to 158.7 kg
460 lb 208.6 kg
Safe working load
Isolibrium (Sum of the patient and
accessory weight)
www.stryker.com 2971-209-001 REV C 1-5
Page 14

Introduction
86 °F
(30 °C)
50 °F
(10 °C)
140 °F
(60 °C)
-40 °F
(-40 °C)
75%
30%
95%
10%
1060 hPa
700 hPa
1060 hPa
500 hPa
English
EN
Specifications (Continued)
Electrical requirements
Power cord
Low Air Loss flow rate
Leakage current 300 uA Maximum
Noise level 50 dBA maximum
Stryker reserves the right to change specifications without notice.
Environmental conditions
Ambient temperature
Relative humidity (non-condensing)
ANSI/AAMI ES60601-1:2005/(R)2012
and A1:2012, C1:2009/(R)2012 and
A2:2010/(R)2012
CAN/CSA-C22.2 No. 60601-1 (2008)
3 foot, 16 AWG cord with hospital grade plug for use with the InTouch foot end
auxiliary power outlet
100 LPM
Operation Storage and transportation
120VAC 50/60Hz, 2.5A
Atmospheric pressure
See the InTouch manual for bed specifications.
Contact information
Contact Stryker Customer Service or Technical Support at: 1-800-327-0770.
Stryker Medical
3800 E. Centre Avenue
Portage, MI 49002
USA
To view your operations or maintenance manual online, see https://techweb.stryker.com/.
Have the serial number (A) of your Stryker product available when calling Stryker Customer Service or Technical
Support. Include the serial number in all written communication.
Serial number location
The serial number (A) for your support surface is located on the patient right side of the foot box on the specification
label. Unzip the support surface top cover (see Cover replacement, top on page 1-20).
1-6 2971-209-001 REV C www.stryker.com
Page 15
Introduction
A
Serial number location (Continued)
English
EN
Date of manufacture
The year of manufacture is the first four digits of the serial number.
www.stryker.com 2971-209-001 REV C 1-7
Page 16
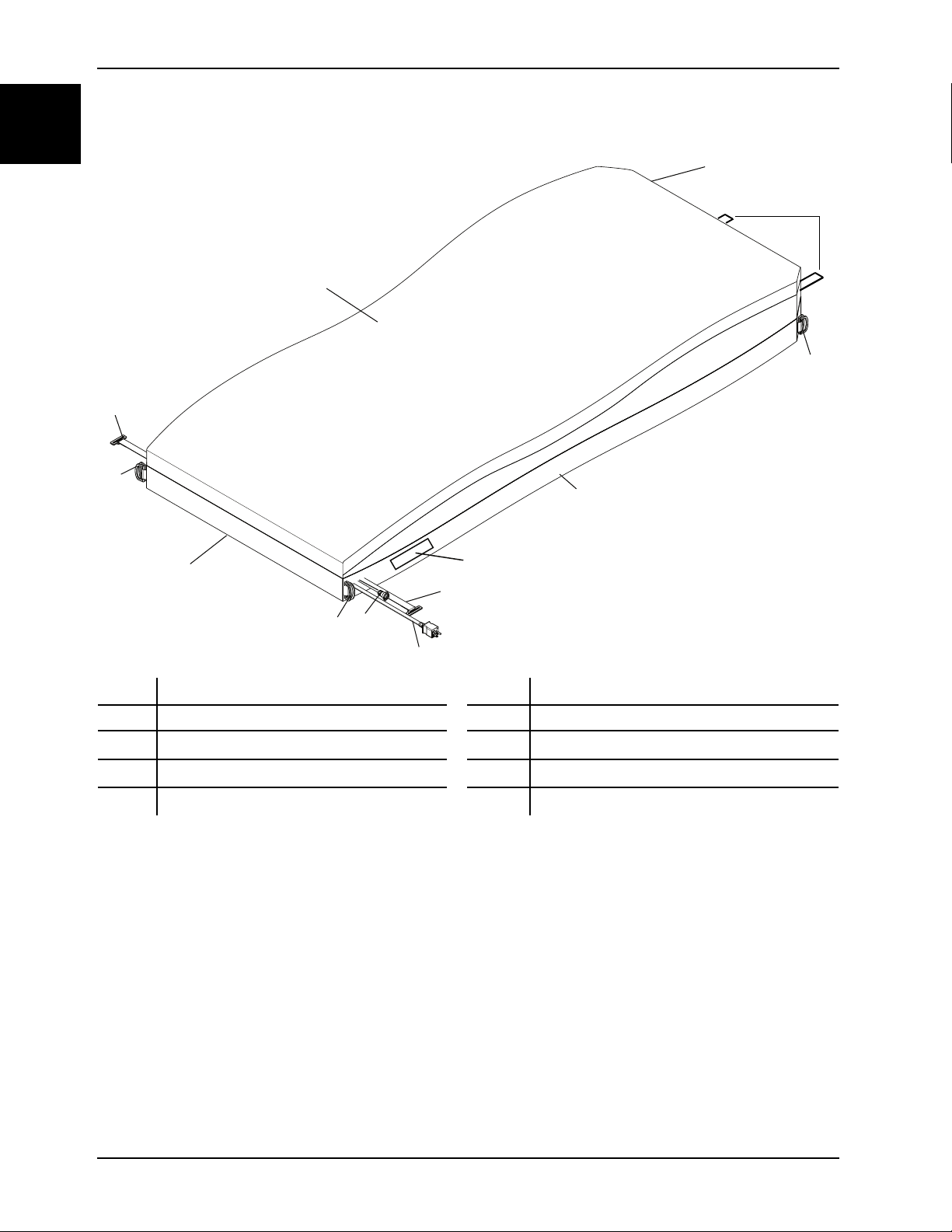
Introduction
C
A
B
D
E
F
G
H
D
J
K
C
C
English
EN
Product illustration
A Head end F Integration cable
B Foot end
C
D
E Power cord K Retainer straps (D-rings not shown)
Note: To view the power cord label, the operator must position the cord by rotating it until the label is legible from
a distance of up to 1m.
D-rings H
CPR straps
G
J
Isolibrium features
Physical Concepts Pressure Redistribution The ability of a support surface to distribute load over the
contact areas of the human body.
Pressure Redistribution setpoint Allows the operator to input patient weight range resulting
in automatic change of zoned sensor technology allowing
immersion and envelopment.
1-8 2971-209-001 REV C www.stryker.com
Handles
Top support surface cover
Bottom support surface cover
Page 17

Introduction
Isolibrium features (Continued)
Lateral Rotation Therapy
BackSmart® Turn Assist A feature of a support surface that provides a single
Max Inflate Allows maximum firmness inflation for use when boosting
Moisture management
Low Air Loss (LAL)
HeelGel™
Patient Specific Immersion™ Assists in managing microclimate of the patient skin,
1
Intelli-Gel® hollow column configuration licensed by EdiZONE LLC of Alpine, UT
1
Technology A feature of the support surface to address pressure
A feature of a support surface that provides rotation
about a longitudinal axis as characterized by degree of
patient turn, duration, and frequency.
position turn about a longitudinal axis.
a patient and on lateral transfer, Promotes patient ease
with dangling, egress, and ingress.
The top surface cover draws excess moisture away from
the patients skin and support surface interface.
Provides air flow to assist in the management of
(microclimate) the skin.
redistribution in the heel area.
patient repositioning, early mobility, and prevention of
pulmonary complications.
English
EN
www.stryker.com 2971-209-001 REV C 1-9
Page 18
Installation
English
EN
The product must reach the ambient operating temperature before installation or operation to avoid the risk of
permanent damage to this product. Upon initial installation, reset the CPR.
WARNING
• Risk of Shock. This product must only be connected to a supply mains with protective earth. Grounding reliability
can be achieved only when a hospital grade receptacle is used.
• Explosion risk. This product is not suitable for use in the presence of flammable anesthetic mixture with air or with
oxygen or nitrous oxide other than nasal, mask type, or half bed-length tent type.
• Do not move the support surface without assistance.
• Do not operate the support surface without the Isolibrium top and bottom covers and linens.
• Always position the support surface on the InTouch bed frame to that the support surface does not contact the
headboard.
• Do not use when gaps between the support surface and the bed frame are present to prevent the risk of
entrapment. Only use the Isolibrium support surface with InTouch bed frames.
• Do not use fitted sheets with this support surface.
• Do not attach the power cord to any moving parts of the InTouch bed frame. Improper routing of the power cord
could cause the risk of electric shock, pinching the power cord, or interfere with bed functions.
• Always confirm proper functionality of the scale system after you install the support surface to prevent product
malfunction.
• Do not use extension cords with Isolibrium. It is only intended to be powered by InTouch with the power cord
supplied.
• Always use the power source provided by the InTouch foot end auxiliary power outlet with Isolibrium.
Note: Do not plug into the power source until the assembly is complete.
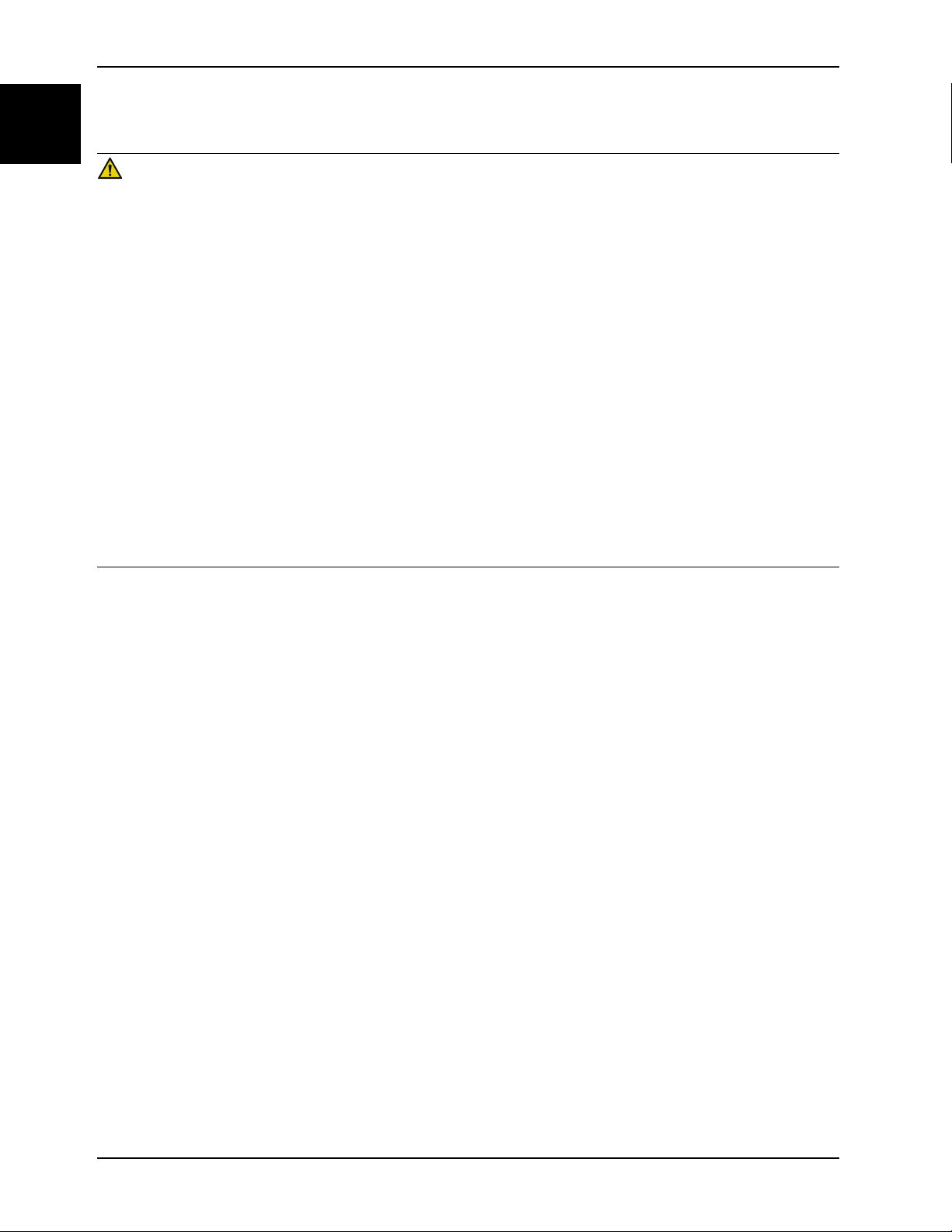
Prerequisite: Minimum of two operators are required to install the support surface.
1. Put the InTouch bed into the flat position before installing the support surface.
2. If powered, unplug the InTouch from the wall outlet and turn the battery switch to off.
3. Remove the InTouch headboard and footboard from the bed.
4. Position an operator at each end of the support surface and lift onto the InTouch bed frame.
1-10 2971-209-001 REV C www.stryker.com
Page 19
Installation
A
B
B
C
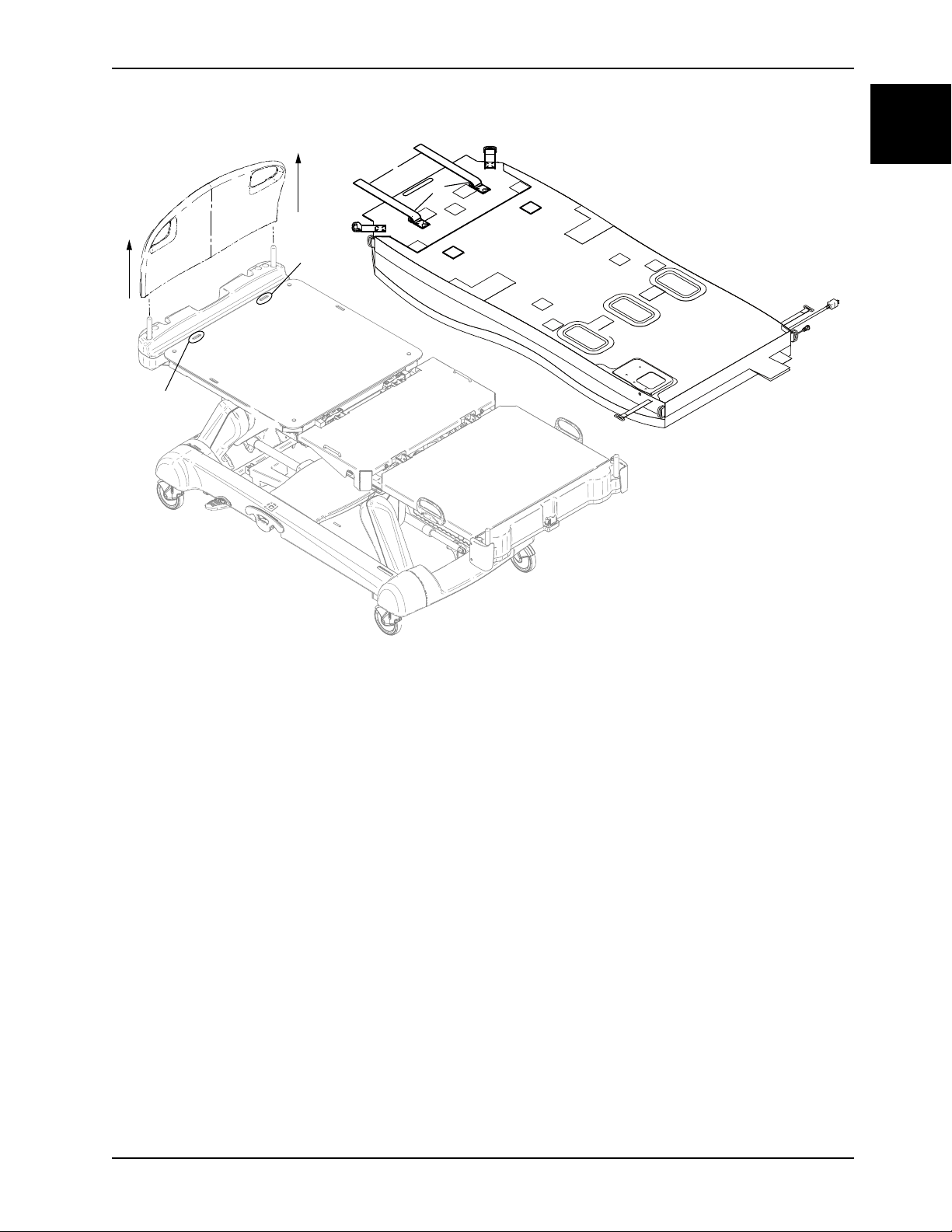
5. Thread the retainer straps (A), down through the InTouch Fowler retainer holes (B) (Figure 1-1 on page 1-11).
English
EN
Figure 1-1: Insert retainer straps
Note: The reverse side of the support surface is shown for reference. Make sure that the support surface is
installed with the top cover facing up.
6. Put the retainer strap through both D-Rings (C), both sides.
7. Thread the retainer strap through the bottom D-Ring, both sides.
8. Pull the retainer strap until the loop around the Fowler is tight, both sides.
9. Insert the loose end of the retainer straps behind the Fowler.
10. Reinstall the InTouch headboard and footboard.
11. Position the support surface against the footboard. Allow a small air gap at the headboard.
12. Connect the integration cable from the support surface foot end to the InTouch cable adapter (B) (Figure 1-2 on
13. Turn the locking collars clockwise to secure the connection.
14. Place the linens on the support surface as loose as possible to avoid creating a hammocking effect and increasing
page 1-12).
interface pressures.
www.stryker.com 2971-209-001 REV C 1-11
Page 20

Installation
A B
English
EN
15. Plug the power cord for the support surface into the InTouch foot end auxiliary power outlet (A) (Figure 1-2 on
page 1-12).
Figure 1-2: InTouch foot end auxiliary power outlet and mattress connector
16. Make sure that the power cord is routed to the side of the bed and not out the foot end of the InTouch.
17. Plug in the InTouch power cord into a properly grounded hospital grade three-prong power outlet.
18. Calibrate the bed following the procedures in the InTouch maintenance manual.
Notes
• Isolibrium is only operational when the power cord for the InTouch bed is in a 120v wall outlet.
• When the support surface is plugged into the InTouch bed, the InTouch footboard will respond. See the InTouch
manual for more information.
1-12 2971-209-001 REV C www.stryker.com
Page 21
Operation
A
B
C
Applying the linens
WARNING
• Always make sure that the operator has access to the CPR straps.
• Do not stick needles into a support surface through the support surface cover. Holes may allow body fluids to enter
the inside (inner core) of the support surface and could cause cross-contamination, product damage, or product
malfunction.
• Do not use fitted sheets with this support surface.
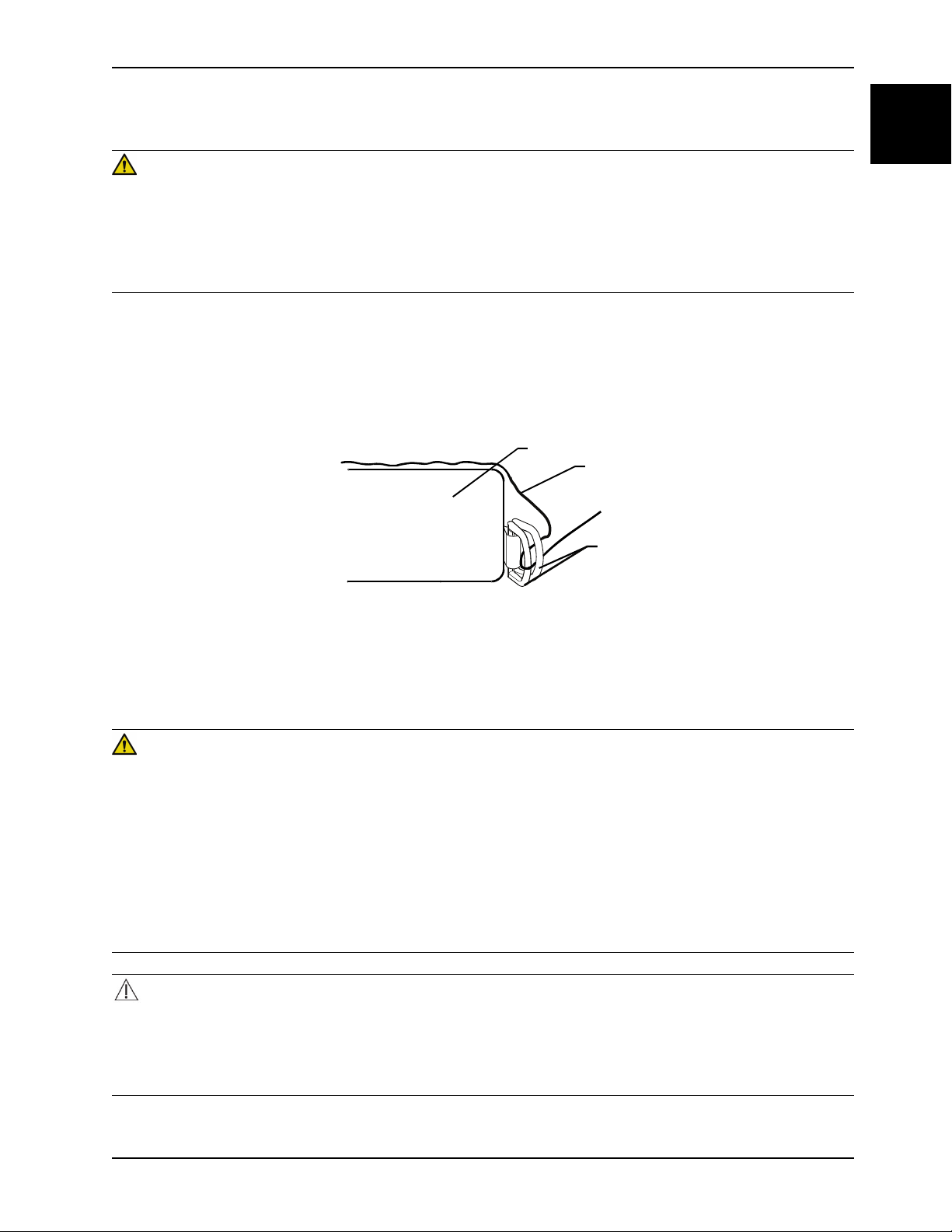
To apply the linens:
1. Apply the linens using the “D” rings for the flat sheet.
2. Thread the four linen corners through the “D” rings (C) attached to the bottom cover to secure the linens (A) to the
support surface (B).
English
EN
Figure 1-3: Apply linens
Note: Do not pull the linens tight. Keep the linens loose and as smooth as possible on top of the support surface to
effectively use the Turn Assist and Lateral Rotation functions.
Positioning a patient on the support surface
WARNING
• Always center the patient on the support surface. Align the patient’s head toward the headboard before starting
functions. Check the patient frequently to make sure that you maintain the proper positioning.
• Always make sure that the tubing and wiring that is connected to the patient is long enough, stable, and secure
during Lateral Rotation or Turn Assist.
• Always raise all of the InTouch bed siderails before starting Turn Assist or Lateral Rotation functions.
• Do not exceed the safe working load of the Isolibrium support surface. Excess weight could cause unpredictable
safety and performance of this system.
• Always use extra caution when reading radiology images taken of a patient on a support surface because internal
components can cause artifacts and distort readings.
CAUTION
• Do not allow sharp objects to come into contact with the support surface that could puncture, tear, or cut the cover.
• Do not allow sharp edges from the X-ray plate to come in contact with the support surface cover. The
recommendation is you cover the X-ray plate with a pillow case or other device before placement under the patient.
If damaged, remove the support surface cover from service immediately to prevent cross contamination.
www.stryker.com 2971-209-001 REV C 1-13
Page 22

Operation
English
EN
Positioning a patient on the support surface (Continued)
To position the patient:
1. Max Inflate the support surface.
2. Position the patient in the center of the support surface, align the patient’s head toward the head board (Figure 1-
4 on page 1-14).
3. Check the patient frequently during Lateral Rotation for proper positioning and support surface inflation (Figure 1-
5 on page 1-14 and Figure 1-6 on page 1-14).
Figure 1-4: Center the patient
Figure 1-5: Correct patient positioning
Figure 1-6: Incorrect patient positioning
Managing incontinence and drainage
WARNING
Always monitor the patient condition at regular intervals for patient safety.
You can use disposable diapers or incontinence pads to manage incontinence. Always provide appropriate skin care
after each incontinence episode.
Activating and resetting CPR
When you activate for CPR, the system deflates and the InTouch control display will change.
WARNING
Always make sure that the operator has access to the CPR straps.
To activate for CPR, pull the red CPR strap straight out from the support surface (Figure 1-7 on page 1-15). The CPR
straps are located on the left and right side of the foot end of the support surface. You can activate either one or both
CPR straps.
1-14 2971-209-001 REV C www.stryker.com
Page 23

Operation
Activating and resetting CPR (Continued)
Figure 1-7: CPR activation
English
EN
Figure 1-8: CPR reset
To reset for CPR, pull down on red CPR strap (Figure 1-8 on page 1-15). The support surface will begin to reinflate.
Relocating the support surface
Relocating the support surface is for the purpose of cleaning or moving to another InTouch bed frame.
WARNING
• Do not move the support surface without assistance.
• Do not use the support surface as a transfer device to avoid the risk of patient injury.
Prerequisite: Minimum of two operators are required to relocate the support surface and setup this product.
1. Make sure that there is not a patient on the support surface.
2. Remove the linens.
3. Unplug the power cord for the support surface from the foot end auxiliary power outlet.
4. Unplug the support surface connector.
5. Remove the headboard and footboard
6. Position an operator at each end of the support surface and remove the support surface and relocate.
Note: Use all four handles when relocating the support surface.
Note: See (Installation on page 1-10) of the support surface.
7. Reinstall the InTouch headboard and footboard.
www.stryker.com 2971-209-001 REV C 1-15
Page 24

Operation
English
EN
Transferring of patient from one patient support platform to another
WARNING
• Do not use the support surface as a transfer device to avoid the risk of patient injury.
• Do not stick needles into a support surface through the support surface cover. Holes may allow body fluids to enter
the inside (inner core) of the support surface and could cause cross-contamination, product damage, or product
malfunction.
• Do not exceed the safe working load of the InTouch bed frame when supporting both the patient and the support
surface. Excess weight could cause unpredictable safety and performance of this product.
To transfer the patient from one patient support surface to another:
Prerequisite: Follow hospital protocols required to transfer a patient from one surface to another.
1. Position one patient support platform alongside the other patient support platform while minimizing the gap between
the two platforms.
2. Set the brakes to on for both patient support platforms.
3. Activate maximum inflation of the support surface until the inflation level is reached. See the InTouch Starting and
Stopping Max Inflate section of the manual for use instructions.
4. Adjust the patient support platform heights so that they are level with one another.
5. Transfer the patient following all applicable safety rules and institution protocols for patient and operator safety.
6. Turn off Max Inflate.
Storing the product
Before storing the product, deflate the support surface by activating CPR.
1-16 2971-209-001 REV C www.stryker.com
Page 25

Cleaning
WARNING
• Always unplug the support surface power cord before cleaning or disinfecting to avoid the risk of shock.
• Do not immerse the support surface or foot box in cleaning or disinfectant solutions.
• Do not allow fluid to pool on the support surface. Fluids may cause degradation of components and may cause
unpredictable safety and performance of this product.
• Always inspect the support surface covers (top and bottom) for tears, punctures, excessive wear, and misaligned
zippers as you clean the covers. If the cover is compromised, remove the support surface from service immediately
and replace the cover to prevent cross-contamination.
• Always perform preventive maintenance based on the usage level of the product. An increase in usage which may
include more frequent cleaning and disinfection, may adversely affect the life of the support surface.
• Always make sure that you wipe each product with clean water and thoroughly dry each product after cleaning or
disinfecting. Some cleaning and disinfecting agents are corrosive in nature and may cause damage to the product if
you use them improperly. If you do not properly rinse and dry the product, a corrosive residue may be left on the
surface of the product that could cause premature degradation of critical components. Failure to follow these
cleaning or disinfecting instructions may void your warranty.
CAUTION
• Do not iron, dry-clean, or tumble dry the support surface covers.
• Do not power wash the support surface.
• Always completely dry the support surface covers before storing, adding linens, or placing a patient on the surface.
English
EN
Prerequisite: Minimum of two operators are required to clean the bottom support surface cover.
Always follow hospital protocol for cleaning and disinfecting.
To clean the support surface covers between patient uses, follow these steps in order:
1. If powered, unplug the InTouch from the wall outlet and turn the battery switch to off.
2. Unplug the support surface power cord.
3. Disconnect the support surface integration cable.
4. Using a clean, soft, damp cloth, wipe the support surface covers with a mild soap and water solution to remove
foreign material.
5. Wipe the support surface covers with a clean, dry cloth to remove any excess liquid or cleaning agent.
Note: To clean the bottom cover of the support surface, raise the head of bed and foot end to help fold the head
end and foot end of the support surface.
6. Thoroughly rinse and dry the support surface covers.
www.stryker.com 2971-209-001 REV C 1-17
Page 26

Disinfecting
English
EN
WARNING
• Always disinfect the support surface between patients, to avoid the risk of cross-contamination and infection.
• Always make sure that you wipe each product with clean water and thoroughly dry each product after cleaning or
disinfecting. Some cleaning and disinfecting agents are corrosive in nature and may cause damage to the product if
you use them improperly. If you do not properly rinse and dry the product, a corrosive residue may be left on the
surface of the product that could cause premature degradation of critical components. Failure to follow these
cleaning or disinfecting instructions may void your warranty.
• Do not allow fluid to pool on the support surface. Fluids may cause degradation of components and may cause
unpredictable safety and performance of this product.
• Always unplug the product power cord before cleaning or disinfecting to avoid the risk of shock.
CAUTION
• Always completely dry the support surface covers before storing, adding linens, or placing a patient on the surface.
• Do not over expose the covers to higher concentration disinfectant solutions as these may degrade the covers.
• Do not use accelerated hydrogen peroxides or quaternaries that contain glycol ethers as they may damage the
cover or the CPR strap print.
Prerequisite: Minimum of two operators are required to disinfect the support surface.
Suggested disinfectants:
• Quaternaries
• Phenolic disinfectants
• Chlorinated bleach solution (5.25% bleach diluted 1 part bleach to 100 parts water)
• Chlorinated bleach solution for 2972 Endurance cover is 10,000ppm
• 70% Isopropyl Alcohol
To disinfect the support surface covers after each patient use, follow these steps in order:
1. Unplug the InTouch power cord.
2. Unplug the support surface.
3. Disconnect the integration cable.
4. Thoroughly clean and dry the support surface covers (see Cleaning on page 1-17) before disinfectants are applied.
5. Apply recommended disinfectant solution with a damp cloth or pre-soaked wipes (do not soak the support surface).
Notes
• Make sure that you follow the disinfectant manufacturer’s instructions for appropriate contact time and rinsing
requirements.
• To disinfect the bottom cover of the support surface, raise the head of bed and foot end to help fold the head
end and foot end of the support surface.
6. Rinse the support surface covers with a clean, damp cloth to remove excess disinfectant.
7. Wipe the support surface covers with a clean, dry cloth to remove any excess liquid or disinfectant.
8. Allow the support surface covers to dry completely before returning to service.
1-18 2971-209-001 REV C www.stryker.com
Page 27

Preventive maintenance
WARNING
Do not service or perform maintenance while the product is in use.
At a minimum, check all items listed during annual preventive maintenance for all Stryker Medical products. You may
need to perform preventive maintenance checks more frequently based on your level of product usage. Service only by
qualified personnel.
Remove product from service before you perform preventive maintenance inspection.
Note: Clean and disinfect the exterior of the support surface before inspection, if applicable.
Inspect the following items:
Zipper, snaps, and covers (top, bottom, and fire barrier) are free of tears, cuts, holes, or any other damage
Note: If excessive wear is observed on the cover or the fire barrier, it is strongly recommended to replace the
cover.
Support surface cover labels are legible, have proper adherence, and free of damage.
Handles are free of rips or cracks
Power cord, integration cable, and plug are free of damage
**Perform a functional test of the Isolibrium functions
**Perform system diagnostics and confirm there are no errors
*Internal components for signs of staining from fluid ingress or contamination by fully unzipping the cover
Pods are not leaking or cracking
Hose connections to the manifold and sensor tubes (both ends) are seated fully
Foam is free from large tears or large gouges
Turn bladders are free of excessive wear and function properly
Low Air Loss fans rotate freely and are free of debris and dust
Left and Right CPR releases function properly
Both foot box cooling fans rotate freely and are free of debris and dust
Pump spring mount has visible clearance from the foot box bottom
Listen for abnormal pump vibration and noise level
Clean the foot box intake metal screen
Clean the mesh on the bottom cover
Clean the fan filter screen on the foot box
Standoffs (feet of the foot box) are present
Cable tie is present on the power cord
Current leakage not more than 200 µA
Ground impedance not more than 100 mΩ (Figure 1-9 on page 1-19)
English
EN
Note: The ground lug can be accessed through the slit in the cover for the CPR, patient left.
Figure 1-9: Ground lug (patient left side)
www.stryker.com 2971-209-001 REV C 1-19
Page 28

Preventive maintenance
English
EN
Notes
• *A zipper pull tool or equivalent is required to access the internal components of the support surface.
• **You cannot perform these preventive maintenance checks without the InTouch footboard. See the InTouch
integrated Models 2131 and 2141, software version 4.0 or higher manual.
• The zipper overlaps at the end and may appear to be misaligned.
Replace the following items every two years:
• Cover assembly, top and bottom
Product serial number:
Completed by:
Date:
Cover replacement, top
Tools required:
• Zipper pull tool or equivalent
Procedure:
1. Apply the InTouch brakes.
2. Unplug the support surface power cord.
3. Remove the headboard and footboards.
4. Lower all the siderails.
5. Unsnap the two secure snaps (C) at the foot end corners (Figure 1-10 on page 1-21).
6. Unsnap the four corner retainers (B) (Figure 1-10 on page 1-21).
7. Insert a zipper pull tool or equivalent through the hole in the zipper slider to unlock.
1-20 2971-209-001 REV C www.stryker.com
Page 29

Preventive maintenance
A
B
C
Cover replacement, top (Continued)
8. Unzip the support surface top cover from the bottom using the zipper pull tool or equivalent (A) (Figure 1-10 on
page 1-21).
Note: The zipper starts on the patient right side near the foot end.
English
EN
Figure 1-10: Top cover
9. Remove and discard the top cover.
10. Reverse to install the new cover.
Notes
• After installation, remove the zipper pull tool or equivalent from the zipper.
• Connect the corner retainers by placing the strap below each D-ring and snaps to the cover. Repeat for the
other three corners.
• Make sure that the watershed covers the zipper.
11. Verify proper operation before returning the product to service.
www.stryker.com 2971-209-001 REV C 1-21
Page 30

Quick reference replacement parts
English
EN
These parts are currently available for purchase. Call Stryker Customer Service: 1-800-327-0770 for availability and
pricing.
Part name Part number
Board, main 2971-022-140
Board, sensor 2971-021-033
Cover, bottom
Cover, top
Cover, bottom (Alternative Dartex)
Cover, top (Alternative Dartex)
Fan, foot box
Fan, LAL 2971-021-189
Integration cable 2971-022-133
Kit, power cord 2971-700-003
Kit, pump assembly 2971-700-001
Kit, resonator assembly 2971-700-002
Kit, valve manifold
Pods assembly 2971-021-003
2971-700-008
2971-021-004
2972-700-001
2972-021-004
2971-022-188
2971-700-007
Power supply 2971-022-171
Kit, solenoid valve, SLI, BLI, BLD, BRI, BRD, SRD, SRI, SLD
Kit, solenoid valve, TRI, TRD, EX, TRI, TU 2971-700-005
Kit, solenoid valve, IN 2971-700-006
2971-700-004
1-22 2971-209-001 REV C www.stryker.com
Page 31

EMC Information
Guidance and Manufacturer’s declaration - Electromagnetic Immunity
Isolibrium is suitable for use in the electromagnetic environment specified below. The customer or the user of
Isolibrium should make sure that it is used in such an environment.
Immunity Test
Electrostatic Discharge
(ESD)
IEC 61000-4-2
IEC 60601 Test Level
+6 kV contact
+8 kV air
Compliance Level
+6 kV contact
+8 kV air
Electromagnetic
Environment Guidance
Floors should be wood,
concrete, or ceramic tile. If
floors are covered with
synthetic material, the
relative humidity should be
at least 30%.
Electrostatic fast transient/
burst
+2 kV for power supply
lines
+2 kV for power supply
lines
Main power quality should
be that of a typical
commercial or hospital
IEC 61000-4-4 *
Surge
IEC 61000-4-5 *
Voltage dips, voltage
variations and short
interruptions on power
supply input lines
IEC 61000-4-11 *
+1 kV for input/ output
lines
+1 kV lines to lines
+2 kV lines to earth
<5% U
(>95% dip in UT)
T
for 0.5 cycle
40% U
(60% dip in UT)
T
for 5 cycles
+1 kV for input/ output
lines
+1 kV lines to lines
+2 kV lines to earth
<5% U
(>95% dip in UT)
T
for 0.5 cycle
40% U
(60% dip in UT)
T
for 5 cycles
environment.
Main power quality should
be that of a typical
commercial or hospital
environment.
Main power quality should
be that of a typical
commercial and/or hospital
environment. If the user of
Isolibrium requires
continued operation during
power main interruptions, it
70% U
(30% dip in UT)
T
for 25 cycles
<5% U
(>95% dip in UT)
T
70% U
(30% dip in UT)
T
for 25 cycles
<5% U
(>95% dip in UT)
T
is recommended that the
device be powered from an
uninterrupted power supply
or a battery.
English
EN
Power frequency (50/60 Hz)
magnetic field
IEC 61000-4-8
for 5 sec
3 A/m 3 A/m
for 5 sec
Power frequency magnetic
fields should be at levels
characteristic of a typical
location in a typical
commercial and/or hospital
environment.
Note: U
is the a.c. mains voltage before applications of the test level.
T
Recommended separation distances between portable and mobile RF communications equipment and
Isolibrium.
Isolibrium is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled.
The customer or the user of Isolibrium can help prevent electromagnetic interferences by maintaining a minimum
distance between portable and mobile RF communications equipment (transmitters) and Isolibrium as recommended
below, according to the maximum output power of the communications equipment.
www.stryker.com 2971-209-001 REV C 1-23
Page 32

EMC Information
English
EN
(Continued)
Rated maximum output power of
transmitter
W
150 kHz to 80 MHz
d=[3.5/V1]√P
0.01 0.12 0.12 0.23
0.1 0.38 0.38 0.73
1
10 3.8 3.8 7.3
100 12 12 23
For transmitters rated at a maximum output power not listed above, the recommended separation distance d in
meters (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the
maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer.
Note: At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies.
Note: These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and
reflection from structures, objects and people.
1.2 1.2 2.3
Separation distance according to frequency of transmitter
m
80 MHz to 800 MHz
d=[3.5/E1]√P
800 MHz to 2.5 GHz
d=[7/E1]√P
Isolibrium is suited for use in the electromagnetic environment specified below. The customer or the user of
Isolibrium should make sure that it is used in such an environment.
Immunity Test
Conducted RF IEC
61000- 4-6 *
Radiated RF
IEC 61000-4-3
IEC 60601 Test Level
3 Vrms
150 kHz to 80 MHz
3 V/m
80 MHz to 2.5 GHz
Compliance Level Electromagnetic Environment - Guidance
Portable and mobile RF communications
equipment should be used no closer to any
part of Isolibrium, including cables, than
the recommended separation distance
calculated from the equation appropriate for
the frequency of the transmitter.
Recommended Separation Distance
d=1.2√P
3 Vrms
3 V/m
where P is the maximum output power rating
of the transmitter in watts (W) according to
the transmitter manufacturer and d is the
recommended separation distance in
Field strengths from fixed RF transmitters,
as determined by an electromagnetic site
d=1.2√P
80 MHz to 800 MHz
d=2.3√P
800 MHz to 2.5 GHz
meters (m).
1-24 2971-209-001 REV C www.stryker.com
Page 33

EMC Information
(Continued)
survey,ashould be less than the compliance
level in each frequency range.
Interference may occur in the vicinity of
equipment marked with the following symbol:
Note: At 80 MHz and 800 MHz, the higher frequency range applies.
Note: These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and
reflection from structures, objects and people.
a
Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land
mobile radios, amateur radio, AM and FM radio broadcast, and TV broadcast cannot be predicted theoretically with
accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey
should be considered. If the measured field strength in the location in which Isolibrium is used exceeds the
applicable RF compliance level above, Isolibrium should be observed to verify normal operation. If abnormal
performance is observed, additional measures may be necessary, such as reorienting or relocating Isolibrium.
b
Over the frequency range 150 kHz to 80 MHz, field strengths are less than 3 V/m.
English
EN
b
www.stryker.com 2971-209-001 REV C 1-25
Page 34

EMC Information
English
EN
(Continued)
Guidance and Manufacturer’s declaration - Electromagnetic Emissions
Isolibrium is intended for use in an electromagnetic environment specified below. The customer or the user of
Isolibrium should make sure that it is used in such an environment.
Emissions Test
RF Emissions
CISPR 11
RF Emissions
CISPR 11
Harmonic Emissions
IEC 61000-3-2
Voltage Fluctuations
Flicker Emissions
IEC 61000-3-3
Compliance
Group 1
Class A
Class A
Complies
Isolibrium uses RF energy only for its internal
function. Therefore, its RF emissions are very low
and are not likely to cause any interference in
nearby electronic equipment.
Isolibrium is suitable for use in all establishments
other than domestic and those directly connected
to the public low voltage power supply network that
supplies buildings used for domestic purposes.
WARNING
This equipment/system is intended for use by healthcare professionals only. This equipment/system may cause radio
interference or may disrupt the operation of nearby equipment. It may be necessary to take mitigation measures, such
as reorienting or relocating Isolibrium or shielding the location.
Electromagnetic Environment
1-26 2971-209-001 REV C www.stryker.com
Page 35

Warranty
Stryker Medical, a division of Stryker Corporation (“Stryker”), warrants that its Model 2971/2972 Isolibrium™ product
will be free from defects in material and workmanship. This Stryker warranty covers only the following items of the
Stryker Isolibrium product (each known individually as a “Part” and collectively as the “Product” or “Isolibrium
product”) during normal use* as follows:
2 year parts, labor, and travel warranty with 1 preventive maintenance inspection per year.
• Support Surface (Air Pods, Turning Bladders and Foam Crib) Warranty Period: 2 years
• Cover and Fire Barrier Assembly Warranty Period: 2 years
• Pump Warranty Period: 2 years
The above noted Warranty periods apply only to the original purchaser of the Isolibrium product and begin on the date
of delivery to such original purchaser.
If Stryker determines, in its sole discretion, that one or more parts is defective within the above noted warranty periods,
then Stryker may, at its option, either repair or replace the Isolibrium product or part.
The warranty set forth above does not include or cover the following:
• Abnormal wear and tear on the product, or wear which indicates that the product was not properly maintained in
accordance with this Operations/Maintenance Manual, or which product has been subject to unusual stress; or
• Product that has been misused, modified, refurbished or repaired without the prior written consent of Stryker; or
• Damage or product failure due to causes beyond Stryker’s control, including but not limited to, abuse, theft, fire,
flood, wind, lightning, freezing, clogging of support surface pores dues to tobacco smoke, unusual atmosphere
conditions, or material degradation due to exposure to moisture; or
• Damage which is determined to have resulted through the use of the product for patient transfer or transport; or
• Product which serial numbers or other identification marks have been removed or destroyed.
English
EN
*”Normal use” is defined as use of the product in typical or normal use settings in a hospital or medical facility under
normal conditions. Damage to the product which arises from abnormal use, such as damage to the product that may be
caused by needle punctures, burns, chemicals, negligent use, improper care, or improper cleaning (as such is detailed
in this Operations/Maintenance Manual) or staining resulting from such abnormal uses are exempt from the above note
warranty coverage.
Warranty exclusion and damage limitations
The express warranty set forth herein is the only warranty applicable to the product. Any and all other warranties,
whether express or implied, including any implied warranty of merchantability or fitness for a particular purpose
are expressly excluded by Stryker. In no event shall Stryker be liable for incidental or consequential damages.
To obtain parts and service
Stryker products are supported by a nationwide network of dedicated Stryker Field Service Representatives. These
representatives are factory trained, available locally, and carry a substantial spare parts inventory to minimize repair
time. Simply call your local representative or call Stryker Customer Service at 1-800-327-0770.
Return authorization
Product cannot be returned without prior approval from the Stryker Customer Service Department. An authorization
number will be provided which must be printed on the returned product. Stryker reserves the right to charge shipping
and restocking fees on returned product. Special, modified, or discontinued products are not subject to return.
www.stryker.com 2971-209-001 REV C 1-27
Page 36

Warranty
English
EN
Damaged product
ICC Regulations require that claims for damaged product must be made within fifteen (15) days of receipt of the product.
Do not accept damaged shipments unless such damage is noted on the delivery receipt at the time of receipt. Upon
prompt notification, Stryker will file a freight claim with the appropriate carrier for damages incurred. Claims will be
limited in amount to the actual replacement cost. In the event that this information is not received by Stryker within the
fifteen (15) day period following the delivery of the product, or the damage was not noted on the delivery receipt at the
time of receipt, the customer will be responsible for payment of the original invoice in full within thirty (30) days of
receipt. Claims for any incomplete shipments must be made within thirty (30) days of invoice.
International warranty clause
This warranty reflects U.S. domestic policy. Warranty outside the U.S. may vary by country. Contact your local Stryker
Medical representative for additional information.
1-28 2971-209-001 REV C www.stryker.com
Page 37

Surface de soutien Isolibrium™
Intégré avec InTouch® FL27 (2131 / 2141 / 2151 / 2152)
version 4.0
2971
2972
Manuel d’utilisation
2017/08 C.3
2971-209-001 REV C
www.stryker.com
Page 38

sample text
Page 39

Symboles
~
Consulter le manuel d’utilisation/notice
Mode d’emploi/Consulter le mode d’emploi
Avertissement général
Mise en garde
Avertissement ; électricité
Courant alternatif
Numéro de référence
Numéro de série
Fabricant
Date de fabrication
Français
FR
IPX4
87VL
Charge maximum admissible
Poids de l’équipement
Borne de protection
Protection contre les projections de liquides
Pièce appliquée de type B résistant à la défibrillation
Équipement médical reconnu par l’organisme de contrôle UL LLC concernant le choc
électrique, l’incendie et les dangers mécaniques conformément aux normes ANSI/AAMI
ES60601-1:2005/(R)2012, A1:2012, C1:2009/(R)2012, A2:2010/(R)2012 et CAN/CSA-C22.2
No. 60601-1 (2008)
Conformément à la directive européenne 2012/19/UE relative aux déchets d’équipements
électriques et électroniques, ce symbole indique que le produit ne doit pas être éliminé avec
les déchets municipaux non triés mais faire l’objet d’une collecte sélective. Pour des
informations sur la mise au rebut, contacter le distributeur local.
www.stryker.com 2971-209-001 REV C
Page 40

Français
FR
Symboles
Nettoyer à la main
Ne pas sécher au sèche-linge
Ne pas nettoyer à sec
Ne pas repasser
Laisser sécher complètement à l’air
Eau de Javel
Pour les brevets américains, consulter www.stryker.com/patents
Redistribution de la pression
Gestion de l’humidité, faible perte d’air
Rotation latérale
Cisaillement
Retournement assisté
2971-209-001 REV C www.stryker.com
Page 41

Table des matières
Définition de « Avertissement », « Mise en garde » et « Remarque » .................................................................. 2-2
Résumé des mesures de sécurité ........................................................................................................ 2-2
Introduction........................................................................................................................................... 2-5
Description du produit ....................................................................................................................... 2-5
Utilisation ....................................................................................................................................... 2-5
Durée de vie prévue.......................................................................................................................... 2-6
Contre-indications ............................................................................................................................ 2-6
Caractéristiques techniques ............................................................................................................... 2-6
Informations de contact ..................................................................................................................... 2-7
Emplacement du numéro de série........................................................................................................ 2-8
Date de fabrication ........................................................................................................................... 2-8
Illustration du produit......................................................................................................................... 2-9
Fonctions de l’Isolibrium .................................................................................................................... 2-9
Installation .......................................................................................................................................... 2-11
Fonctionnement ................................................................................................................................... 2-14
Mise en place des draps.................................................................................................................. 2-14
Installation d’un patient sur la surface d’appui...................................................................................... 2-14
Prise en charge de l’incontinence et du drainage.................................................................................. 2-15
Activation et réinitialisation de la RCP................................................................................................. 2-16
Déplacement de la surface d’appui.................................................................................................... 2-16
Transfert d’un patient entre deux plateformes d’appui ........................................................................... 2-17
Stockage du produit........................................................................................................................ 2-17
Nettoyage........................................................................................................................................... 2-18
Désinfection........................................................................................................................................ 2-19
Maintenance préventive......................................................................................................................... 2-20
Remplacement de la housse supérieure.............................................................................................. 2-21
Référence rapide des pièces de rechange ................................................................................................ 2-23
Informations CEM................................................................................................................................. 2-24
Garantie ............................................................................................................................................. 2-28
Exclusion de garantie et limitations des dommages ............................................................................... 2-28
Pièces de rechange et service technique ............................................................................................ 2-28
Autorisation de retour...................................................................................................................... 2-29
Produit endommagé........................................................................................................................ 2-29
Clause de garantie internationale ...................................................................................................... 2-29
Français
FR
www.stryker.com 2971-209-001 REV C 2-1
Page 42

Définition de « Avertissement », « Mise en garde » et
« Remarque »
Les termes AVERTISSEMENT, MISE EN GARDE et REMARQUE ont une signification particulière et doivent faire l’objet
d’une lecture attentive.
Français
FR
AVERTISSEMENT
Avertit le lecteur d’une situation qui, si elle n’est pas évitée, pourrait entraîner la mort ou des blessures graves. Peut
également attirer l’attention sur l’existence potentielle d’effets indésirables graves ou de risques d’accident.
MISE EN GARDE
Avertit le lecteur d’une situation potentiellement dangereuse qui, si elle n’est pas évitée, peut causer des blessures
mineures ou modérées à l’utilisateur ou au patient ou endommager le matériel en question ou d’autres biens. Couvre
notamment les précautions à prendre afin d’assurer l’utilisation sûre et efficace du dispositif et d’éviter les dommages
qui pourraient découler de l’usage ou du mésusage du matériel.
Remarque : Fournit des informations spécifiques destinées à faciliter l’entretien ou à clarifier des instructions
importantes.
Résumé des mesures de sécurité
Lire attentivement et respecter strictement les avertissements et les mises en garde indiqués sur cette page. Toute
réparation doit être effectuée exclusivement par du personnel qualifié.
AVERTISSEMENT
• Risque de choc électrique. Ce produit doit être uniquement branché à une alimentation électrique dotée d’un
raccordement à la terre. La fiabilité de la mise à la terre ne peut être assurée que si une prise secteur aux normes
hospitalières est utilisée.
• Risque d’explosion. Ce produit ne convient pas pour une utilisation en présence de mélange anesthésique
inflammable avec un air ambiant ou avec de l’oxygène ou de l’oxyde nitreux destiné à une utilisation autre pour le
nez, pour des masques ou des tentes sur moitié de lit.
• Ne pas déplacer la surface d'appui sans assistance.
• Ne pas utiliser la surface d'appui sans les housses supérieure et inférieure de l’Isolibrium et sans draps.
• Toujours positionner la surface d'appui sur le châssis du lit InTouch de manière à ce qu’elle ne touche pas la tête
de lit.
• Ne pas utiliser en cas d’espace entre la surface d'appui et le châssis du lit afin d’éviter tout risque de prise au
piège. Utiliser uniquement la surface d’appui Isolibrium avec des châssis de lit InTouch.
• Ne pas fixer le cordon d’alimentation électrique aux parties mobiles du châssis du lit InTouch. L’acheminement
incorrect du cordon d'alimentation électrique peut entraîner un risque de choc électrique, le pincement du cordon
d'alimentation électrique ou gêner le fonctionnement du lit.
• Pour éviter tout dysfonctionnement du produit, toujours vérifier le fonctionnement correct du système de pesée
après l’installation de la surface d'appui.
• Ne pas utiliser des rallonges de câble avec l’Isolibrium. Elle doit uniquement être alimentée par le cordon
d'alimentation électrique fourni avec le lit InTouch.
• Toujours alimenter l’Isolibrium avec l’alimentation électrique fournie par la prise électrique auxiliaire du côté pieds
du lit InTouch.
• Veiller toujours à ce que l’opérateur puisse accéder aux sangles de CPR.
• Ne pas planter d’aiguille dans une surface d’appui à travers la housse. La formation de petits trous risque de
provoquer l’infiltration de fluides corporels à l’intérieur de la surface d’appui (dans le corps interne), ce qui pourrait
entraîner une contamination croisée ou un dysfonctionnement du produit, ou endommager ce dernier.
2-2 2971-209-001 REV C www.stryker.com
Page 43

Définition de « Avertissement », « Mise en garde » et «
Remarque »
Résumé des mesures de sécurité (Suite)
AVERTISSEMENT (SUITE)
• Ne pas installer de draps-housses sur cette surface d’appui.
• Toujours centrer le patient sur la surface d’appui. Placer la tête du patient en direction de la tête de lit avant de
commencer les opérations. Vérifier souvent la position correcte du patient.
• Toujours vérifier que les tubes et câbles reliés au patient sont suffisamment longs, stables et solidement raccordés
pendant la rotation latérale ou le retournement assisté.
• Toujours relever les barrières du lit InTouch avant de commencer une rotation latérale ou un retournement assisté.
• Ne pas dépasser la charge maximale admissible de la surface d’appui Isolibrium. Une charge excessive pourrait
rendre imprévisible la sécurité et les performances de ce système.
• Toujours procéder avec précaution lors de la lecture des radiographies prises avec un patient sur la surface
d’appui car les composants internes peuvent causer des artéfacts et altérer les résultats.
• Toujours contrôler l’état du patient à des intervalles réguliers pour la sécurité du patient.
• Afin de ne pas blesser le patient, ne pas utiliser la surface d’appui comme un système de transfert.
• Ne pas immerger la surface d’appui ou la boîte côté pieds dans des solutions nettoyantes ou désinfectantes.
• Ne pas laisser de fluides s’accumuler sur la surface d’appui. Les fluides peuvent endommager les composants et
rendre imprévisible la sécurité et les performances de ce produit.
• Lors du nettoyage, toujours inspecter les housses (inférieure et supérieure) de la surface d’appui pour vérifier
l’absence de déchirures, perçages, usure excessive et désynchronisation de la fermeture à glissière. Si la housse
est endommagée, cesser immédiatement l’utilisation de la surface d’appui et remplacer la housse afin d’éviter
toute contamination croisée.
• Toujours effectuer une maintenance préventive en fonction du niveau d’utilisation du produit. Une augmentation de
l’utilisation (pouvant inclure un nettoyage et une désinfection fréquents) risque de diminuer la durée de vie de la
surface d’appui.
• Toujours désinfecter la surface d’appui entre chaque patient afin d’éviter tout risque de contamination croisée et
d’infection.
• Toujours veiller à bien rincer chaque produit à l’eau claire et à sécher complètement chaque produit après le
nettoyage ou la désinfection. Certains produits de nettoyage et de désinfection sont de nature corrosive et peuvent
endommager le produit s’ils ne sont pas utilisés convenablement. Si le produit n’est pas rincé et séché
correctement, un résidu corrosif peut rester sur la surface du produit et peut entraîner une corrosion prématurée
des composants essentiels. Le non-respect de ces instructions de nettoyage ou de désinfection peut annuler la
garantie.
• Toujours débrancher le cordon d’alimentation électrique du produit avant le nettoyage ou la désinfection afin
d’éviter tout risque de choc électrique.
• Ne pas effectuer l’entretien ni la maintenance du produit lorsqu’il est en cours d’utilisation.
Français
FR
MISE EN GARDE
• L’utilisation incorrecte du produit est susceptible de causer des blessures au patient ou à l’utilisateur. Utiliser le
produit uniquement de la manière décrite dans ce manuel.
• Ne pas modifier le produit ni aucun de ses composants. Toute modification du produit peut entraîner un
fonctionnement imprévisible, susceptible de causer des blessures au patient ou à l’utilisateur. La garantie du
produit serait en outre invalidée par toute modification du produit.
• Eviter tout contact d’objets tranchants avec la surface d’appui car ils pourraient percer, déchirer ou couper la
housse.
www.stryker.com 2971-209-001 REV C 2-3
Page 44

Français
FR
Définition de « Avertissement », « Mise en garde » et «
Remarque »
Résumé des mesures de sécurité (Suite)
MISE EN GARDE (SUITE)
• Eviter tout contact des bords tranchants de la plaque pour rayons X avec la housse de la surface d’appui. Il est
recommandé de recouvrir la plaque à rayons X d’une taie d’oreiller (ou autre) avant de la placer sous le patient. En
cas d’endommagement, retirer immédiatement la housse de la surface d’appui pour éviter une contamination
croisée.
• Ne pas repasser, ni nettoyer à sec, ni sécher au sèche-linge les housses de la surface d’appui.
• Ne pas passer la surface d’appui en laveuse à pression.
• Toujours sécher complètement les housses de la surface d’appui avant de les stocker, de poser des draps ou
d’installer un patient sur la surface d’appui.
• Ne pas appliquer de doses excessives de solutions de désinfection à forte concentration sur les housses car elles
risquent de les endommager.
• Ne pas utiliser de peroxydes d’hydrogène accélérés ni de mélanges quaternaires contenant des éthers glycoliques
car ils peuvent endommager la housse ou le marquage des sangles de CPR.
2-4 2971-209-001 REV C www.stryker.com
Page 45

Introduction
Ce manuel vous aide à utiliser ou entretenir votre produit Stryker. Lire ce manuel avant d’utiliser ce produit ou d’en
effectuer la maintenance. Il convient d'établir des procédures et techniques visant à éduquer et à former le personnel
quant au fonctionnement et à l’entretien sécuritaires de ce produit.
MISE EN GARDE
• L’utilisation incorrecte du produit est susceptible de causer des blessures au patient ou à l’utilisateur. Utiliser le
produit uniquement de la manière décrite dans ce manuel.
• Ne pas modifier le produit ni aucun de ses composants. Toute modification du produit peut entraîner un
fonctionnement imprévisible, susceptible de causer des blessures au patient ou à l’utilisateur. La garantie du
produit serait en outre invalidée par toute modification du produit.
Remarques :
• Ce manuel doit être considéré comme faisant partie du produit et doit l’accompagner à tout moment, même en cas
de vente ultérieure du produit.
• Stryker cherche continuellement à améliorer le design et la qualité de ses produits. Ce manuel contient les
informations produit les plus récentes disponibles au moment de l’impression. Il peut y avoir de légères divergences
entre le produit et ce manuel. Pour toute question, contacter le service clientèle ou le support technique de Stryker
au +1-800-327-0770.
Description du produit
L’Isolibrium Stryker 2971/2972 est une surface d’appui électrisée dotée de fonctions assurant une redistribution de la
pression, une faible perte d’air et une rotation latérale. L’Isolibrium est destinée à une utilisation avec le châssis de lit
InTouch® (version logicielle 4.0 ou supérieure). Isolibrium et InTouch consistent en un lit intégré et une surface aidant
les soignants pendant la délivrance de soins aux patients.
Utilisation
Français
FR
La surface d’appui Isolibrium aide à la prévention des plaies de pression. Toutes les catégories ou stades de plaies de
pression, y compris I, II, III, IV, immesurables et lésion tissulaire profonde. Avant la mise en place de l’Isolibrium, un
professionnel de santé doit effectuer une évaluation clinique des facteurs de risque et une évaluation dermatologique.
Le support de patient Isolibrium facilite la gestion du microclimat de la peau du patient, le repositionnement du patient,
le déplacement anticipé et la prévention des complications pulmonaires.
Les systèmes InTouch et Isolibrium doivent être utilisés par des patients en unités de soins intensifs. Les unités de
soins intensifs peuvent inclure les soins intensifs, les soins courants, les soins gradués, les soins médicaux/chirurgicaux,
les soins pour affections subaiguës et l’unité de soins post-anesthésie (USPA). D’autres unités peuvent être prescrites.
Le patient, les accessoires et les éventuelles charges supplémentaires ne doivent pas dépasser la charge maximale
admissible du système InTouch ou Isolibrium.
Les opérateurs du système sont des professionnels de santé (infirmières, aides-soignants et médecins) autorisés à
utiliser les fonctions telles que : les fonctions de déplacement du lit, l’appel d’une infirmière, les fonctions de
communication intégrées des barrières, la sortie du lit ou les options de traitement. Les patients et les visiteurs peuvent
utiliser les fonctions de déplacement du lit, l’appel d’une infirmière et les fonctions de communication intégrées des
barrières. Ils ne peuvent cependant pas utiliser les fonctions électrisées de la surface d’appui. Les professionnels
formés sont chargés de l’installation, de l’entretien et de l’étalonnage des systèmes InTouch et Isolibrium.
www.stryker.com 2971-209-001 REV C 2-5
Page 46

Introduction
Durée de vie prévue
La surface de soutien Isolibrium possède une durée de vie prévue de cinq ans dans des conditions d’utilisation
normales et en respectant la maintenance périodique appropriée.
Français
FR
Les housses Isolibrium (supérieur et inférieure) possèdent une durée de vie prévue de deux ans dans des conditions
d’utilisation normales.
Contre-indications
Stryker recommande l’évaluation clinique de chaque patient et une utilisation adaptée par l’opérateur.
Les systèmes InTouch et Isolibrium ne sont pas prévus pour :
• une utilisation pour des patients mesurant 88,9 cm ou moins,
• une utilisation pour des patients pesant moins de 22,7 kg,
• une utilisation avec une tente intégrale à oxygène,
• le soutien de plusieurs personnes en même temps,
• le soutien d’un patient âgé de moins de deux ans,
• une utilisation pour des soins de santé à domicile,
• une utilisation en tant que produit stérile,
• une utilisation en présence d’un mélange anesthésique inflammable avec de l’air, de l’oxygène ou de l’oxyde
nitreux.
Remarque : Lorsque l'Isolibrium est installé sur le lit InTouch, il est possible de poser une tente à oxygène sur la
moitié du lit à l'intérieur des barrières. N'inclure aucune partie de la boîte côté pieds de la surface de soutien sous
la tente à oxygène.
Isolibrium n’est pas destiné à une utilisation sur les patients présentant les cas suivants :
• Fractures instables
• Lésions de la moelle épinière instables
Les fonctions de rotation latérale et de retournement assisté de l’Isolibrium ne peuvent être utilisées qu’après une
évaluation clinique. Cela permet d’éviter toute réaction indésirable potentielle chez les patients présentant les cas
suivants :
• Lésion de la moelle épinière
• Traction squelettique
• Hémoptysie sévère
• Troubles du saignement
• Fractures de côte
• Aggravation d’une dyspnée ou d’une respiration sifflante, due aux techniques
• Instabilité hémodynamique
• Augmentation de la pression intracrânienne
• Traction squelettique
Caractéristiques techniques
Modèle
Longueur 84 po. 213,4 cm
2971/2972 Isolibrium
2-6 2971-209-001 REV C www.stryker.com
Page 47

Introduction
86 °F
(30 °C)
50 °F
(10 °C)
140 °F
(60 °C)
-40 °F
(-40 °C)
75%
30%
95%
10%
1060 hPa
700 hPa
1060 hPa
500 hPa
Caractéristiques techniques (Suite)
Largeur 35 po. 88,9 cm
Épaisseur
8 à 10,5 po. 20,3 à 26,7 cm
Poids 90 livres 40,8 kg
Poids thérapeutique 50 à 350 livres 22,7 à 158,7 kg
460 livres 208,6 kg
Charge maximale admissible
Isolibrium (poids total du patient et
des accessoires)
Spécifications électriques ANSI/AAMI ES60601-1:2005/(R)2012 et
120 V ca, 50/60Hz, 2,5 A
A1:2012, C1:2009/(R)2012 et A2:2010/
(R)2012
CAN/CSA-C22.2 No. 60601-1 (2008)
Cordon d’alimentation électrique Cordon de 0,9 m (3 pieds), 16 AWG avec fiche aux normes hospitalières pour
une utilisation sur une prise électrique auxiliaire côté pieds du lit InTouch
Débit à faible perte d’air
Courant de fuite
100 l/min
300 uA maximum
Niveau de bruit 50 dBA maximum
Stryker se réserve le droit de modifier ces caractéristiques sans préavis.
Conditions ambiantes Fonctionnement
Stockage et transport
Français
FR
Température ambiante
Humidité relative (sans
condensation)
Pression atmosphérique
Pour voir les caractéristiques techniques du lit, voir le manuel InTouch.
Informations de contact
Contacter le service clientèle ou le support technique de Stryker au +1-800-327-0770.
Stryker Medical
3800 E. Centre Avenue
Portage, MI 49002
États-Unis
www.stryker.com 2971-209-001 REV C 2-7
Page 48

Français
A
FR
Introduction
Informations de contact (Suite)
Pour consulter votre mode d'emploi ou votre manuel d'entretien en ligne, rendez-vous sur https://techweb.stryker.com/.
Avoir le numéro de série (A) du produit Stryker à disposition avant d’appeler le service clientèle ou le support technique
de Stryker. Inclure le numéro de série dans toutes les communications écrites.
Emplacement du numéro de série
Le numéro de série (A) de votre surface d’appui se trouve sur l’étiquette d’identification située sur la boîte côté pieds,
côté droit du patient. Ouvrir la fermeture à glissière de la housse supérieure de la surface d’appui (voir Remplacement
de la housse supérieure à la page 2-21).
Date de fabrication
Les quatre premiers chiffres du numéro de série correspondent à l'année de fabrication.
2-8 2971-209-001 REV C www.stryker.com
Page 49
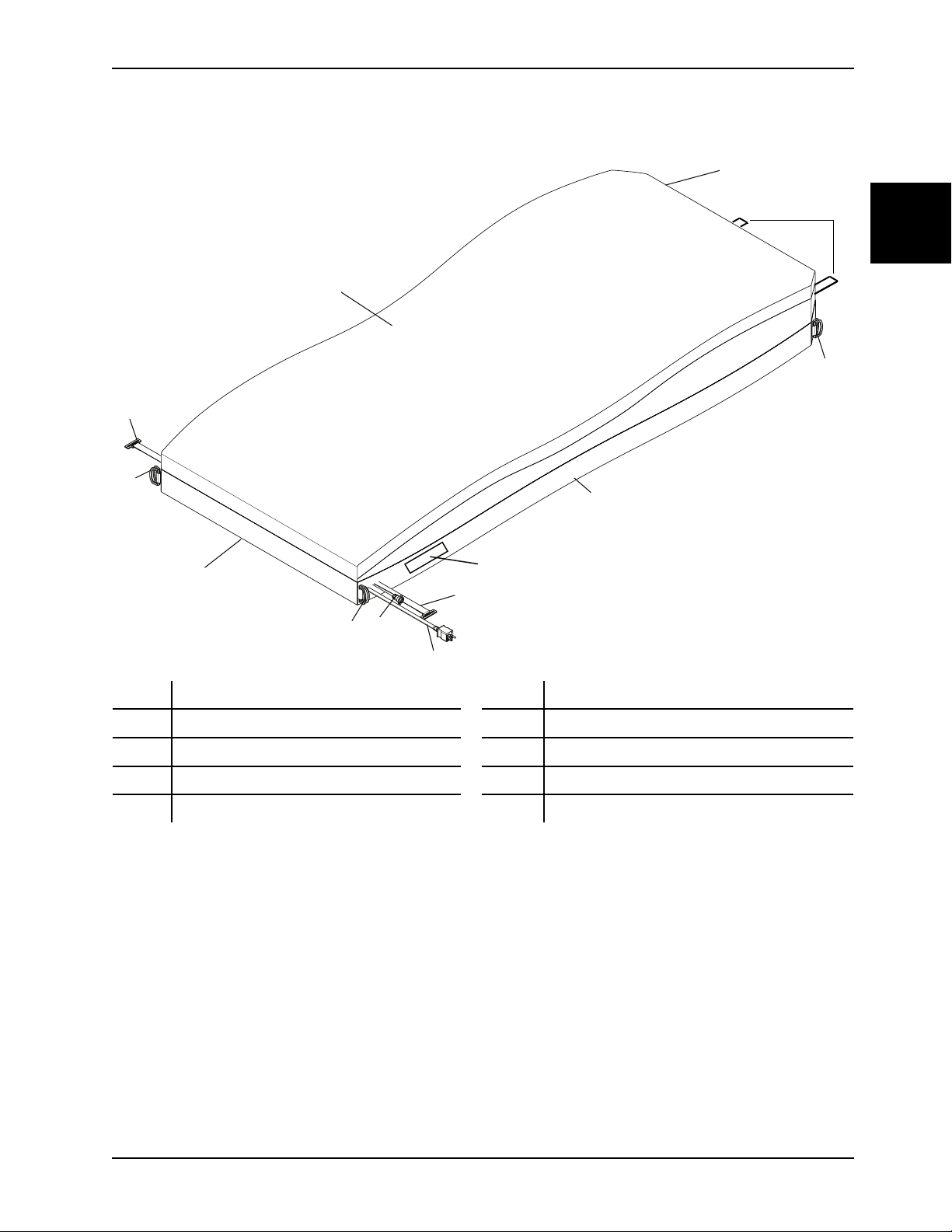
Illustration du produit
C
A
B
D
E
F
G
H
D
J
K
C
C
Introduction
Français
FR
A
B
C
D
E
Remarque : Pour voir l’étiquette du cordon d’alimentation électrique, l’opérateur doit tourner ce dernier jusqu’à
ce que l’étiquette soit lisible jusqu’à une distance d’1 mètre.
Côté tête
Côté pieds G Poignées
Anneaux en D H
Sangles de CPR
Cordon d’alimentation
Fonctions de l’Isolibrium
Redistribution de la pression selon les concepts
physiques
Réglage de la redistribution de la pression Permet à l’opérateur de définir la plage de poids du
www.stryker.com 2971-209-001 REV C 2-9
F
Câble d’intégration
Housse supérieure de la surface d’appui
J
K
Housse inférieure de la surface d’appui
Sangles de retenue (anneaux en D non illustrés)
La capacité d’une surface d’appui à répartir la charge
sur les zones de contact du corps humain.
patient. Celle-ci est utilisée par la technologie de
capteurs zonés pour la modification automatique du
support afin d’assurer l’immersion et l’enveloppement du
patient.
Page 50

Français
FR
Introduction
Fonctions de l’Isolibrium (Suite)
Traitement par rotation latérale Fonction de la surface d’appui qui effectue une rotation
autour d’un axe longitudinal selon un degré, une durée et
une fréquence de retournement du patient.
Retournement assisté BackSmart® Fonction de la surface d’appui qui effectue un
retournement simple autour d’un axe longitudinal.
Max Inflate (Gonflage maximum) Offre une fermeté optimale pour redresser un patient et
lors d’un transfert latéral. Facilite les mouvements du
patient pour se mettre au bord du lit jambes pendantes,
sortir du lit et entrer dans le lit.
Gestion de l’humidité La housse supérieure draine l’humidité excessive de la
peau du patient et du drap de la surface d’appui.
Low Air Loss (LAL) (Faible perte d’air [FPA])
Technologie HeelGel™
Patient Specific Immersion™ (Immersion spécifique au
patient)
1
Configuration à colonnes creuses Intelli-Gel® sous licence d’EdiZONE LLC d’Alpine, Utah, États-Unis
1
Assure une circulation de l’air pour aider à la régulation
(du microclimat) de la peau.
Une fonction de la surface d’appui, qui permet de
redistribuer la pression dans la région des talons.
Facilite la gestion du microclimat de la peau du patient,
le repositionnement du patient, le déplacement anticipé
et la prévention des complications pulmonaires.
2-10 2971-209-001 REV C www.stryker.com
Page 51

Installation
Afin d’éviter tout risque d’endommagement définitif de ce produit, il doit être porté à la température ambiante de
fonctionnement avant d’être installé et utilisé. Lors de l’installation initiale, réinitialiser la fonction RCP.
AVERTISSEMENT
• Risque de choc électrique. Ce produit doit être uniquement branché à une alimentation électrique dotée d’un
raccordement à la terre. La fiabilité de la mise à la terre ne peut être assurée que si une prise secteur aux normes
hospitalières est utilisée.
• Risque d’explosion. Ce produit ne convient pas pour une utilisation en présence de mélange anesthésique
inflammable avec un air ambiant ou avec de l’oxygène ou de l’oxyde nitreux destiné à une utilisation autre pour le
nez, pour des masques ou des tentes sur moitié de lit.
• Ne pas déplacer la surface d'appui sans assistance.
• Ne pas utiliser la surface d'appui sans les housses supérieure et inférieure de l’Isolibrium et sans draps.
• Toujours positionner la surface d'appui sur le châssis du lit InTouch de manière à ce qu’elle ne touche pas la tête
de lit.
• Ne pas utiliser en cas d’espace entre la surface d'appui et le châssis du lit afin d’éviter tout risque de prise au
piège. Utiliser uniquement la surface d’appui Isolibrium avec des châssis de lit InTouch.
• Ne pas installer de draps-housses sur cette surface d’appui.
• Ne pas fixer le cordon d’alimentation électrique aux parties mobiles du châssis du lit InTouch. L’acheminement
incorrect du cordon d'alimentation électrique peut entraîner un risque de choc électrique, le pincement du cordon
d'alimentation électrique ou gêner le fonctionnement du lit.
• Pour éviter tout dysfonctionnement du produit, toujours vérifier le fonctionnement correct du système de pesée
après l’installation de la surface d'appui.
• Ne pas utiliser des rallonges de câble avec l’Isolibrium. Elle doit uniquement être alimentée par le cordon
d'alimentation électrique fourni avec le lit InTouch.
• Toujours alimenter l’Isolibrium avec l’alimentation électrique fournie par la prise électrique auxiliaire du côté pieds
du lit InTouch.
Français
FR
Remarque : Ne pas procéder au branchement sur la source d’alimentation avant que le montage ne soit terminé.
Condition requise : Au minimum deux opérateurs sont requis pour installer la surface d’appui.
1. Mettre le lit InTouch à plat avant d’installer la surface d’appui.
2. S’il sous tension, débrancher le lit InTouch de la prise électrique murale et tourner l’interrupteur de la batterie sur
la position d’arrêt.
3. Retirer la tête de lit et le pied de lit du lit InTouch.
4. Positionner un opérateur à chaque bout de la surface d'appui et soulever cette dernière sur le châssis du lit
InTouch.
www.stryker.com 2971-209-001 REV C 2-11
Page 52

Français
A
B
B
C
FR
Installation
5. Enfiler les sangles de retenue (A), par les trous de retenue (B) du relève-buste du lit InTouch (Figure 2-1 à la page
2-12).
Figure 2-1 : Insertion des sangles de retenue
Remarque : La surface inférieure de la surface d’appui est illustrée pour référence. S’assurer que la surface
d’appui est installée avec la housse supérieure face en haut.
6. Passer la sangle de retenue dans les deux anneaux en D (C), des deux côtés.
7. Passer la sangle de retenue dans l’anneau en D inférieur, des deux côtés.
8. Tirer la sangle de retenue jusqu’à ce que la boucle autour du relève-buste soit serrée, des deux côtés.
9. Introduire l’extrémité libre des sangles de retenue derrière le relève-buste.
10. Repositionner la tête de lit et le pied de lit InTouch.
11. Placer la surface d'appui contre le pied de lit. Laisser un petit espace au niveau de la tête de lit.
12. Brancher le câble d’intégration du côté pieds de la surface d'appui sur l’adaptateur de câble (B) du lit InTouch
(Figure 2-2 à la page 2-13).
13. Tourner les bagues de blocage dans le sens horaire pour serrer le raccordement.
14. Placer les draps le plus lâche possible sur la surface d'appui pour éviter tout effet de hamac et éviter
l’augmentation des pressions sur la surface.
2-12 2971-209-001 REV C www.stryker.com
Page 53

Installation
A B
15. Brancher le cordon d'alimentation électrique de la surface d'appui à la prise électrique auxiliaire (A) côté pieds du
lit InTouch (Figure 2-2 à la page 2-13).
Figure 2-2 : Prise électrique auxiliaire côté pieds du lit InTouch et raccord de matelas
16. Vérifier que le cordon d’alimentation électrique est acheminé le long du lit et qu’il ne ressort pas du côté pieds du lit
InTouch.
17. Brancher le cordon d'alimentation électrique du lit InTouch à une prise électrique murale trois broches aux normes
hospitalières.
18. Étalonner le lit en suivant les procédures indiquées dans le manuel d’entretien du lit InTouch.
Français
FR
Remarques :
• L’Isolibrium ne fonctionne que lorsque le cordon d’alimentation électrique du lit InTouch est branché à une prise
murale 120 V.
• Lorsque la surface d'appui est branchée au lit InTouch, le pied de lit InTouch fonctionne. Pour plus d’informations,
voir le manuel InTouch.
www.stryker.com 2971-209-001 REV C 2-13
Page 54

Français
A
B
C
FR
Fonctionnement
Mise en place des draps
AVERTISSEMENT
• Veiller toujours à ce que l’opérateur puisse accéder aux sangles de CPR.
• Ne pas planter d’aiguille dans une surface d’appui à travers la housse. La formation de petits trous risque de
provoquer l’infiltration de fluides corporels à l’intérieur de la surface d’appui (dans le corps interne), ce qui pourrait
entraîner une contamination croisée ou un dysfonctionnement du produit, ou endommager ce dernier.
• Ne pas installer de draps-housses sur cette surface d’appui.
Pour mettre en place les draps :
1. Mettre les draps en utilisant les anneaux en D prévus pour drap plat.
2. Pour fixer les draps (A) à la surface d’appui (B), passer les quatre coins du drap dans les anneaux en D (C) fixés à
la surface d’appui.
Figure 2-3 : Mise en place des draps
Remarque : Ne pas trop serrer les draps. Les draps doivent être suffisamment lâches et lisses sur la surface d’appui
afin de pouvoir utiliser les fonctions de retournement assisté et de rotation latérale.
Installation d’un patient sur la surface d’appui
AVERTISSEMENT
• Toujours centrer le patient sur la surface d’appui. Placer la tête du patient en direction de la tête de lit avant de
commencer les opérations. Vérifier souvent la position correcte du patient.
• Toujours vérifier que les tubes et câbles reliés au patient sont suffisamment longs, stables et solidement raccordés
pendant la rotation latérale ou le retournement assisté.
• Toujours relever les barrières du lit InTouch avant de commencer une rotation latérale ou un retournement assisté.
• Ne pas dépasser la charge maximale admissible de la surface d’appui Isolibrium. Une charge excessive pourrait
rendre imprévisible la sécurité et les performances de ce système.
• Toujours procéder avec précaution lors de la lecture des radiographies prises avec un patient sur la surface
d’appui car les composants internes peuvent causer des artéfacts et altérer les résultats.
2-14 2971-209-001 REV C www.stryker.com
Page 55

Fonctionnement
Installation d’un patient sur la surface d’appui (Suite)
MISE EN GARDE
• Eviter tout contact d’objets tranchants avec la surface d’appui car ils pourraient percer, déchirer ou couper la
housse.
• Eviter tout contact des bords tranchants de la plaque pour rayons X avec la housse de la surface d’appui. Il est
recommandé de recouvrir la plaque à rayons X d’une taie d’oreiller (ou autre) avant de la placer sous le patient. En
cas d’endommagement, retirer immédiatement la housse de la surface d’appui pour éviter une contamination
croisée.
Pour positionner le patient :
1. Gonfler au maximum la surface d’appui.
2. Positionner le patient au centre de la surface d’appui et placer sa tête en direction de la tête de lit (Figure 2-4 à la
page 2-15).
3. Vérifier souvent la position correcte du patient pendant la rotation latérale et le gonflage de la surface d’appui
(Figure 2-5 à la page 2-15 et Figure 2-6 à la page 2-15).
Français
FR
Figure 2-4 : Centrer le patient
Figure 2-5 : Positionnement correct du patient
Figure 2-6 : Positionnement incorrect du patient
Prise en charge de l’incontinence et du drainage
AVERTISSEMENT
Toujours contrôler l’état du patient à des intervalles réguliers pour la sécurité du patient.
www.stryker.com 2971-209-001 REV C 2-15
Page 56

Fonctionnement
Prise en charge de l’incontinence et du drainage (Suite)
Il est possible d’utiliser des couches jetables ou des serviettes pour incontinent pour prendre en charge l’incontinence.
Toujours dispenser les soins cutanés appropriés après chaque épisode d’incontinence.
Français
FR
Activation et réinitialisation de la RCP
Lorsque vous activez la RCP, la surface d’appui se dégonfle et l’écran de commande InTouch change d’affichage.
AVERTISSEMENT
Veiller toujours à ce que l’opérateur puisse accéder aux sangles de CPR.
Pour activer la RCP, tirer sur la sangle de CPR rouge pour la faire sortir de la surface d’appui (Figure 2-7 à la page 2-
16). Les sangles de CPR se trouvent sur les côtés gauche et droit du côté pieds de la surface d’appui. Il est possible de
tirer une seule sangle CPR, ou les deux.
Figure 2-7 : Activation de la RCP
Figure 2-8 : Réinitialisation de la RCP
Pour réinitialiser la RCP, tirer la sangle CPR rouge vers le bas (Figure 2-8 à la page 2-16). La surface d’appui
commence à se regonfler.
Déplacement de la surface d’appui
Le déplacement de la surface d’appui est destiné à des fins de nettoyage ou d’installation sur un autre châssis de lit
InTouch.
AVERTISSEMENT
• Ne pas déplacer la surface d’appui sans assistance.
• Afin de ne pas blesser le patient, ne pas utiliser la surface d’appui comme un système de transfert.
Condition requise : Au minimum deux opérateurs sont requis pour déplacer et préparer la surface d’appui.
2-16 2971-209-001 REV C www.stryker.com
Page 57

Fonctionnement
Déplacement de la surface d’appui (Suite)
1. Vérifier qu’aucun patient n’est installé sur la surface d’appui.
2. Enlever les draps.
3. Débrancher le cordon d’alimentation électrique de la surface d’appui sur la prise électrique auxiliaire située côté
pieds.
4. Débrancher la fiche de la surface d’appui.
5. Retirer la tête de lit et le pied de lit.
6. Placer un opérateur de chaque côté de la surface d’appui, puis retirer cette dernière et la déplacer.
Remarque : Utiliser les quatre poignées lors du déplacement de la surface d’appui.
Remarque : Voir (Installation à la page 2-11) de la surface d’appui.
7. Repositionner la tête de lit et le pied de lit InTouch.
Transfert d’un patient entre deux plateformes d’appui
AVERTISSEMENT
• Afin de ne pas blesser le patient, ne pas utiliser la surface d’appui comme un système de transfert.
• Ne pas planter d’aiguille dans une surface d’appui à travers la housse. La formation de petits trous risque de
provoquer l’infiltration de fluides corporels à l’intérieur de la surface d’appui (dans le corps interne), ce qui pourrait
entraîner une contamination croisée ou un dysfonctionnement du produit, ou endommager ce dernier.
• Ne pas dépasser la charge maximale admissible du châssis du lit InTouch lorsque celui-ci soutient le patient et la
surface d’appui. Une charge excessive pourrait rendre imprévisible la sécurité et les performances de ce produit.
Français
FR
Pour transférer un patient entre deux surfaces d’appui :
Condition requise : Respecter le protocole hospitalier relatif au transfert de patient entre deux surfaces d’appui.
1. Placer une plateforme d’appui de patient le long d’une autre plateforme d’appui, en veillant à minimiser l’espace
entre elles.
2. Serrer les freins sur les deux plateformes d’appui de patient.
3. Activer le gonflage maximal sur la surface d’appui jusqu’à atteindre le niveau de gonflage. Voir la section
Démarrage et arrêt du gonflage maximal sur InTouch du manuel pour consulter les instructions d’utilisation.
4. Régler la hauteur de la plateforme d’appui de patient suivant la hauteur de l’autre plateforme.
5. Pour assurer la sécurité du patient et de l’opérateur, observer toutes les règles de sécurité et protocoles hospitaliers
en vigueur lors du transfert du patient.
6. Désactiver le gonflage maximal.
Stockage du produit
Avant de stocker le produit, dégonfler la surface d’appui en activant la fonction RCP.
www.stryker.com 2971-209-001 REV C 2-17
Page 58

Français
FR
Nettoyage
AVERTISSEMENT
• Toujours débrancher le cordon d’alimentation électrique de la surface d’appui avant le nettoyage ou la désinfection
afin d’éviter tout risque de choc électrique.
• Ne pas immerger la surface d’appui ou la boîte côté pieds dans des solutions nettoyantes ou désinfectantes.
• Ne pas laisser de fluides s’accumuler sur la surface d’appui. Les fluides peuvent endommager les composants et
rendre imprévisible la sécurité et les performances de ce produit.
• Lors du nettoyage, toujours inspecter les housses (inférieure et supérieure) de la surface d’appui pour vérifier
l’absence de déchirures, perçages, usure excessive et désynchronisation de la fermeture à glissière. Si la housse
est endommagée, cesser immédiatement l’utilisation de la surface d’appui et remplacer la housse afin d’éviter
toute contamination croisée.
• Toujours effectuer une maintenance préventive en fonction du niveau d’utilisation du produit. Une augmentation de
l’utilisation (pouvant inclure un nettoyage et une désinfection fréquents) risque de diminuer la durée de vie de la
surface d’appui.
• Toujours veiller à bien rincer chaque produit à l’eau claire et à sécher complètement chaque produit après le
nettoyage ou la désinfection. Certains produits de nettoyage et de désinfection sont de nature corrosive et peuvent
endommager le produit s’ils ne sont pas utilisés convenablement. Si le produit n’est pas rincé et séché
correctement, un résidu corrosif peut rester sur la surface du produit et peut entraîner une corrosion prématurée
des composants essentiels. Le non-respect de ces instructions de nettoyage ou de désinfection peut annuler la
garantie.
MISE EN GARDE
• Ne pas repasser, ni nettoyer à sec, ni sécher au sèche-linge les housses de la surface d’appui.
• Ne pas passer la surface d’appui en laveuse à pression.
• Toujours sécher complètement les housses de la surface d’appui avant de les stocker, de poser des draps ou
d’installer un patient sur la surface d’appui.
Condition requise : Au minimum deux opérateurs sont requis pour nettoyer la housse inférieure de la surface d’appui.
Toujours respecter le protocole hospitalier concernant le nettoyage et la désinfection.
Pour nettoyer les housses de la surface d’appui entre chaque patient, suivre la procédure suivante :
1. S’il sous tension, débrancher le lit InTouch de la prise électrique murale et tourner l’interrupteur de la batterie sur
la position d’arrêt.
2. Débrancher le cordon d’alimentation électrique de la surface d’appui.
3. Débrancher le câble d’intégration de la surface d’appui.
4. A l’aide d’un chiffon propre, doux et humide, essuyer les housses de la surface d’appui en utilisant un mélange
d’eau et de savon doux pour enlever tout corps étranger.
5. Essuyer les housses de la surface d’appui à l’aide d’un chiffon propre et sec afin d’enlever tout excès de liquide ou
d’agent nettoyant.
Remarque : Pour nettoyer la housse inférieure de la surface d’appui, soulever la tête et le pied du lit afin de plier
les côtés tête et pieds de la surface d’appui.
6. Rincer et sécher complètement les housses de la surface d’appui.
2-18 2971-209-001 REV C www.stryker.com
Page 59

Désinfection
AVERTISSEMENT
• Toujours désinfecter la surface d’appui entre chaque patient afin d’éviter tout risque de contamination croisée et
d’infection.
• Toujours veiller à bien rincer chaque produit à l’eau claire et à sécher complètement chaque produit après le
nettoyage ou la désinfection. Certains produits de nettoyage et de désinfection sont de nature corrosive et peuvent
endommager le produit s’ils ne sont pas utilisés convenablement. Si le produit n’est pas rincé et séché
correctement, un résidu corrosif peut rester sur la surface du produit et peut entraîner une corrosion prématurée
des composants essentiels. Le non-respect de ces instructions de nettoyage ou de désinfection peut annuler la
garantie.
• Ne pas laisser de fluides s’accumuler sur la surface d’appui. Les fluides peuvent endommager les composants et
rendre imprévisible la sécurité et les performances de ce produit.
• Toujours débrancher le cordon d’alimentation électrique du produit avant le nettoyage ou la désinfection afin
d’éviter tout risque de choc électrique.
MISE EN GARDE
• Toujours sécher complètement les housses de la surface d’appui avant de les stocker, de poser des draps ou
d’installer un patient sur la surface d’appui.
• Ne pas appliquer de doses excessives de solutions de désinfection à forte concentration sur les housses car elles
risquent de les endommager.
• Ne pas utiliser de peroxydes d’hydrogène accélérés ni de mélanges quaternaires contenant des éthers glycoliques
car ils peuvent endommager la housse ou le marquage des sangles de CPR.
Français
FR
Condition requise : Au minimum deux opérateurs sont requis pour désinfecter la surface d’appui.
Désinfectants recommandés :
• Mélanges quaternaires
• Désinfectants phénoliques
• Solution d’eau de Javel (1 dose d’eau de Javel 5,25 % diluée dans 100 doses d’eau)
• Solution d’eau de Javel for 2972 Endurance cover is 10,000ppm
• Alcool isopropylique à 70 %
Pour désinfecter les housses de la surface d’appui après chaque patient, suivre la procédure suivante :
1. Débrancher le cordon d’alimentation électrique du lit InTouch.
2. Débrancher la surface d’appui.
3. Débrancher le câble d’intégration.
4. Nettoyer et sécher complètement les housses de la surface d’appui (voir Nettoyage à la page 2-18) avant de les
désinfecter.
5. Appliquer les solutions désinfectantes recommandées à l’aide d’un chiffon humide ou des lingettes pré-imbibées
(ne pas imprégner la surface d’appui).
Remarques :
• Suivre les recommandations du fabricant du désinfectant concernant la durée d’application appropriée et les
spécifications de rinçage.
• Pour désinfecter la housse inférieure de la surface d’appui, soulever la tête et le pied du lit afin de plier les
côtés tête et pieds de la surface d’appui.
6. Rincer les housses de la surface d’appui à l’aide d’un chiffon propre humide afin d’enlever l’excès de désinfectant.
7. Essuyer les housses de la surface d’appui à l’aide d’un chiffon propre et sec afin d’enlever tout excès de liquide ou
de désinfectant.
8. Laisser sécher complètement les housses de la surface d’appui avant de les réutiliser.
www.stryker.com 2971-209-001 REV C 2-19
Page 60

Français
FR
Maintenance préventive
AVERTISSEMENT
Ne pas effectuer l’entretien ni la maintenance du produit lorsqu’il est en cours d’utilisation.
Au minimum, vérifier tous les éléments mentionnés pendant la maintenance préventive annuelle pour tous les produits
Stryker Medical. Il peut être nécessaire d’effectuer les vérifications de maintenance préventive plus fréquemment en
fonction du degré d’utilisation du produit. Toute réparation doit être effectuée exclusivement par du personnel qualifié.
Arrêter l’entretien du produit avant d’effectuer les vérifications de maintenance préventive.
Remarque : Nettoyer et désinfecter l’extérieur de la surface d’appui avant inspection, le cas échéant.
Inspecter les éléments suivants :
La fermeture à glissière, les loquets et housses (supérieure, inférieure et protection incendie). Elles ne doivent
présenter aucune déchirure, coupure, trou, ni aucun autre dommage.
Remarque : Si on observe une usure excessive de la housse ou de la protection incendie, il est fortement
recommandé de remplacer la housse.
Les étiquettes de la housse de la surface d’appui sont lisibles, bien collées et intactes.
Les poignées ne présentent pas d’accroc ni de fissures.
Le cordon d’alimentation électrique, le câble d’intégration et la fiche sont intacts.
**Effectuer un test de fonctionnement des fonctions de l’Isolibrium.
**Effectuer des diagnostics du système et confirmer l’absence d’anomalies.
*Rechercher sur les composants internes toute trace de contamination ou de tache due à l’infiltration de fluides
après avoir ouvert complètement la fermeture à glissière.
Les cellules ne présentent pas de fuites ni de fissures.
Les raccords des tubes du collecteur et du capteur (à chaque extrémité) sont complètement enfoncés.
La mousse ne présente pas de déchirures ni de fentes importantes.
Les poches de rotation ne présentent pas d’usure excessive et fonctionnent correctement.
Les ventilateurs à faible perte d’air (FPA) tournent librement et ne contiennent aucun débris ni poussière.
Les sangles d’activation de RCP gauche et droite fonctionnent correctement.
Les ventilateurs de refroidissement de la boîte côté pieds tournent librement et ne contiennent aucun débris ni
poussière.
Le ressort de la pompe est libre de tout obstacle depuis le fond de la boîte côté pieds.
Ecouter pour détecter toute anomalie de vibration de la pompe et de niveau de bruit.
Nettoyer la crépine métallique de la boîte côté pieds.
Nettoyer le filet sur la housse inférieure.
Nettoyer la crépine du filtre du ventilateur sur la boîte côté pieds.
Les entretoises (pieds de la boîte côté pieds) sont présentes.
Le serre-câble est fixé au cordon d’alimentation électrique.
Le courant de fuite ne dépasse pas 200 µA.
L’impédance de la mise à la terre ne dépasse pas 100 mΩ (Figure 2-9 à la page 2-21).
Remarque : La cosse de mise à la terre est accessible par la fente de la housse destinée à la fonction RCP, côté
gauche du patient.
2-20 2971-209-001 REV C www.stryker.com
Page 61

Maintenance préventive
Figure 2-9 : Cosse de mise à la terre (côté gauche du patient)
Remarques :
• *Une tirette de fermeture à glissière ou un dispositif équivalent est requis pour accéder aux composants internes de
la surface d’appui.
• **Il n’est pas possible d’effectuer ces vérifications de maintenance préventive sans le pied de lit InTouch. Voir le
manuel des modèles intégrés InTouch 2131 et 2141, logiciel version 4.0 ou supérieure.
• La fermeture à glissière se chevauche à son extrémité et peut apparaître désynchronisée.
Remplacer les composants suivants tous les deux ans :
• Ensemble de housses supérieure et inférieure
Français
FR
Numéro de série du produit :
Effectué par :
Date :
Remplacement de la housse supérieure
Outils requis :
• Tirette de fermeture à glissière ou dispositif équivalent
Procédure :
1. Serrer les freins du lit InTouch.
2. Débrancher le cordon d’alimentation électrique de la surface d’appui.
3. Retirer la tête de lit et le pied de lit.
4. Abaisser toutes les barrières.
5. Désenclencher les deux loquets de fixation (C) à chaque coin du côté pieds (Figure 2-10 à la page 2-22).
6. Désenclencher les attaches aux quatre coins (B) (Figure 2-10 à la page 2-22).
7. Introduire une tirette de fermeture à glissière ou un dispositif équivalent dans le trou du curseur de la fermeture à
glissière à débloquer.
www.stryker.com 2971-209-001 REV C 2-21
Page 62
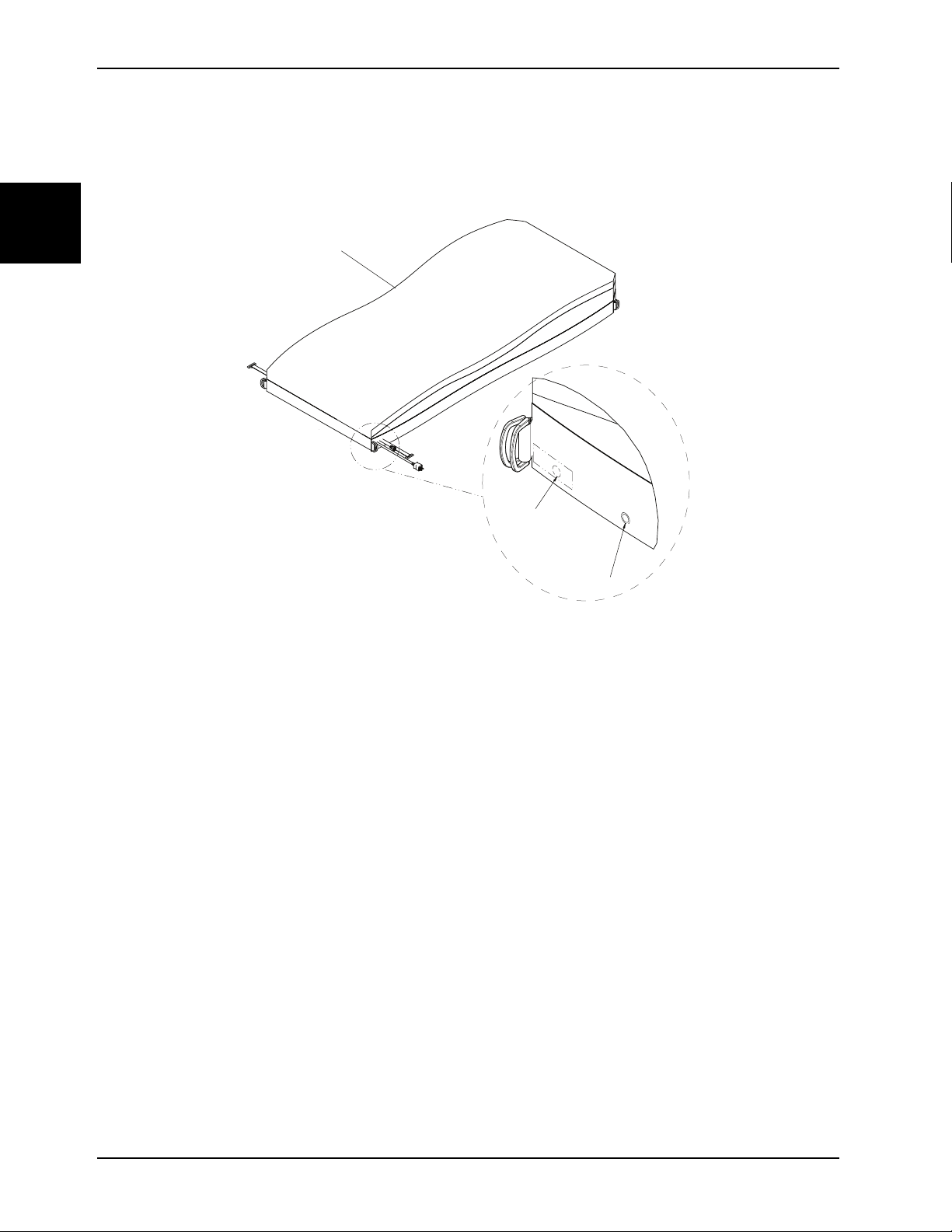
Français
A
B
C
FR
Maintenance préventive
Remplacement de la housse supérieure (Suite)
8. A l’aide de la tirette ou d’un dispositif équivalent (A), ouvrir la fermeture à glissière de la housse supérieure/
inférieure de la surface d’appui (Figure 2-10 à la page 2-22).
Remarque : La fermeture à glissière démarre sur le côté droit du patient près du côté pieds.
Figure 2-10 : Housse supérieure
9. Retirer et éliminer la housse supérieure.
10. Inverser la procédure pour installer une housse neuve.
Remarques :
• Après l’installation, retirer la tirette de fermeture à glissière ou un dispositif équivalent.
• Installer les attaches aux coins en plaçant la sangle sous chaque anneau en D et en la fixant à la housse.
Répéter la procédure pour les trois autres coins.
• Vérifier que le rabat recouvre la fermeture à glissière.
11. Vérifier le fonctionnement correct avant de remettre le produit en service.
2-22 2971-209-001 REV C www.stryker.com
Page 63

Référence rapide des pièces de rechange
Ces pièces sont actuellement disponibles à la vente. Appeler le service clientèle de Stryker au 1-800-327-0770 pour
connaître la disponibilité et les prix.
Dénomination de la pièce Nº de pièce
Carte mère
Circuit du capteur
Housse inférieure
Housse supérieure
Housse inférieure (Dartex de rechange)
Housse supérieure (Dartex de rechange)
Ventilateur de la boîte côté pieds
Ventilateur FPA 2971-021-189
Câble d’intégration
Kit de cordon d’alimentation électrique
Kit du bloc de pompe 2971-700-001
Kit du système de résonateur
Kit de collecteur de valves 2971-700-007
Assemblage de cellules 2971-021-003
Bloc d’alimentation électrique
Kit d’électrovanne, SLI, BLI, BLD, BRI, BRD, SRD, SRI, SLD
Kit d’électrovanne, TRI, TRD, EX, TRI, TU
2971-022-140
2971-021-033
2971-700-008
2971-021-004
2972-700-008
2972-021-004
2971-022-188
2971-022-133
2971-700-003
2971-700-002
2971-022-171
2971-700-004
2971-700-005
Français
FR
Kit d’électrovanne, IN
2971-700-006
www.stryker.com 2971-209-001 REV C 2-23
Page 64

Français
FR
Informations CEM
Directives et déclaration du fabricant – Immunité électromagnétique
La surface d’appui Isolibrium peut être utilisée dans l’environnement électromagnétique spécifié ci-dessous.
L’acheteur ou l’utilisateur de l’Isolibrium doit s’assurer qu’elle est utilisée dans un tel environnement.
Environnement
Test d’immunité Niveau d’essai IEC 60601 Niveau de conformité
Décharge électrostatique
(DES)
+6 kV contact
+8 kV air
+6 kV contact
+8 kV air
IEC 61000-4-2
Transitoires électriques
rapides/rafale
IEC 61000-4-4 *
Surtension
IEC 61000-4-5 *
+2 kV pour les lignes
électriques
+1 kV pour les lignes
entrée/sortie
+1 kV lignes à lignes
+2 kV lignes à la terre
+2 kV pour les lignes
électriques
+1 kV pour les lignes
entrée/sortie
+1 kV lignes à lignes
+2 kV lignes à la terre
électromagnétique –
Directives
Les sols doivent être en
bois, en béton ou en
carrelage céramique. Si
les sols sont recouverts
d’un matériau synthétique,
l’humidité relative doit être
d’au moins 30 %.
La qualité de la tension du
secteur doit être la qualité
habituelle
d’un environnement
commercial ou hospitalier.
La qualité de la tension du
secteur doit être la qualité
habituelle
d’un environnement
commercial ou hospitalier.
Baisses de tension,
variations de tension et
coupures brèves sur les
lignes d’alimentation
d’entrée
IEC 61000-4-11 *
Champ magnétique à la
fréquence du secteur (50/
60 Hz)
IEC 61000-4-8
<5 % U
U
pendant 0,5 cycle
40 % U
en U
pendant 5 cycles
(baisse >95 % en
T
)
T
(baisse de 60 %
T
)
T
<5 % U
U
(baisse >95 % en
T
)
T
pendant 0,5 cycle
40 % U
en U
(baisse de 60 %
T
)
T
pendant 5 cycles
La qualité de la tension du
secteur doit être la qualité
habituelle
d’un environnement
commercial et/ou
hospitalier. Si l’utilisateur
de l’Isolibrium nécessite le
maintien du
fonctionnement durant les
70 % U
en U
(baisse de 30 %
T
)
T
pendant 25 cycles
<5 % U
U
(baisse >95 % en
T
)
T
pendant 5 secondes
70 % U
en U
(baisse de 30 %
T
)
T
pendant 25 cycles
<5 % U
U
(baisse >95 % en
T
)
T
pendant 5 secondes
interruptions du secteur, il
est recommandé
d’alimenter le système à
l’aide d’un onduleur ou
d’une batterie.
3 A/m 3 A/m Les niveaux des champs
magnétiques à la
fréquence du secteur
doivent correspondre aux
niveaux caractéristiques
d’un emplacement type
dans un environnement
commercial et/ou
hospitalier type.
Remarque : U
2-24 2971-209-001 REV C www.stryker.com
représente la tension du secteur (CA) avant l’application du niveau d’essai.
T
Page 65

Informations CEM
Distances de séparation recommandées entre les équipements de communication RF portables et mobiles et
l’Isolibrium.
L’Isolibrium est destiné à être utilisé dans un environnement électromagnétique où les perturbations des RF
rayonnées sont contrôlées. L’acheteur ou l’utilisateur de l’Isolibrium peut contribuer à prévenir les interférences
électromagnétiques en respectant la distance minimum entre les équipements de communication RF portables et
mobiles (émetteurs) et l’Isolibrium, recommandée ci-dessous en fonction de la puissance de sortie maximum des
équipements de communication.
Français
FR
Puissance de sortie maximum
nominale de l’émetteur
W
0,01 0,12 0,12 0,23
0,1 0,38 0,38 0,73
1
10 3,8 3,8 7,3
100 12 12 23
Pour les émetteurs dont la puissance de sortie maximum nominale n’est pas indiquée ci-dessus, la distance de
séparation recommandée d en mètres (m) peut être déterminée en utilisant l’équation applicable à la fréquence de
l’émetteur, où P est la puissance de sortie maximum nominale en watts (W), selon le fabricant de l’émetteur.
Remarque : À 80 MHz et 800 MHz, la distance de séparation pour la gamme de fréquences la plus élevée s’applique.
Remarque : Ces directives peuvent ne pas convenir à toutes les situations. La propagation électromagnétique est
affectée par l’absorption et la réflexion des structures, objets et personnes.
Distance de séparation en fonction de la fréquence de l’émetteur
m
150 kHz à 80 MHz
d=[3,5/V1]√P
1,2 1,2 2,3
80 MHz à 800 MHz
d=[3,5/E1]√P
800 MHz à 2,5 GHz
d=[7/E1]√P
L’Isolibrium peut être utilisé dans l’environnement électromagnétique spécifié ci-dessous. L’acheteur ou l’utilisateur
de l’Isolibrium doit s’assurer qu’il est utilisé dans un tel environnement.
Test d’immunité
RF émise IEC 61000-
4-6 *
RF rayonnée
IEC 61000-4-3
www.stryker.com 2971-209-001 REV C 2-25
Niveau d’essai IEC
60601
3 Vrms
150 kHz à 80 MHz
3 V/m
80 MHz à 2,5 GHz
Niveau de
conformité
3 Vrms
3 V/m
Environnement électromagnétique –
Directives
Les équipements de communication RF
portables et mobiles ne doivent pas être
utilisés en deçà de la limite de distance
recommandée calculée avec l’équation
applicable à la fréquence de l’émetteur,
relativement à toute partie de l’Isolibrium, y
compris les câbles.
Distance de séparation recommandée
d=1,2√P
d=1,2√P
80 MHz à 800 MHz
d=2,3√P
800 MHz à 2,5 GHz
Page 66

Français
FR
Informations CEM
(Suite)
Où P correspond à la puissance nominale
de sortie maximum en watts (W) pour
l’émetteur, selon le fabricant de ce dernier,
et d correspond à la distance recommandée
Les intensités de champ produites par les
émetteurs RF fixes, telles que déterminées
par une étude de site électromagnétique
doivent être inférieures au niveau de
conformité de chaque gamme de
Des interférences peuvent survenir à
proximité des appareils porteurs du symbole
Remarque : À 80 MHz et à 800 MHz, la gamme de fréquences la plus élevée s’applique.
en mètres (m).
fréquences.
b
suivant :
a
Remarque : Ces directives peuvent ne pas convenir à toutes les situations. La propagation électromagnétique est
affectée par l’absorption et la réflexion des structures, objets et personnes.
a
Les intensités de champ des émetteurs fixes, telles que les stations de base pour les radiotéléphones (cellulaires/
sans fil) et les radios mobiles terrestres, la radio d’amateur, la radiodiffusion AM et FM, et la diffusion télévisuelle, ne
peuvent pas être prévues théoriquement avec exactitude. Pour évaluer l’environnement électromagnétique dû aux
émetteurs RF fixes, il convient d’envisager une étude de site électromagnétique. Si l’intensité de champ mesurée à
l’emplacement où l’Isolibrium est utilisé dépasse le niveau de conformité RF applicable indiqué ci-dessus, il convient
d’observer l’Isolibrium pour vérifier qu’il fonctionne normalement. Si des performances anormales sont observées,
des mesures supplémentaires peuvent être nécessaires, telles que la réorientation ou le déplacement de l’Isolibrium.
b
Dans la gamme de fréquences de 150 kHz à 80 MHz, les intensités de champ sont inférieures à 3 V/m.
2-26 2971-209-001 REV C www.stryker.com
Page 67

Informations CEM
(Suite)
Directives et déclaration du fabricant – Émissions électromagnétiques
La surface d’appui Isolibrium est destinée à être utilisée dans l’environnement électromagnétique spécifié cidessous. L’acheteur ou l’utilisateur de l’Isolibrium doit s’assurer qu’il est utilisé dans un tel environnement.
Test d’émissions Conformité
Émissions RF
CISPR 11
Groupe 1
L’Isolibrium utilise une énergie RF uniquement
pour ses fonctions internes. Par conséquent, ses
émissions RF sont très faibles et ne peuvent
vraisemblablement pas provoquer d’interférence
dans les équipements électroniques situés à
proximité.
Environnement électromagnétique
Français
FR
Émissions RF
CISPR 11
Émissions harmoniques
IEC 61000-3-2
Fluctuations de tension
Émissions de scintillement
IEC 61000-3-3
Classe A
Classe A
Conforme
L’Isolibrium convient à une utilisation dans tous les
établissements autres que les établissements
domestiques et ceux directement raccordés au
réseau basse tension public alimentant les
bâtiments à usage domestique.
AVERTISSEMENT
Cet équipement/système est destiné à une utilisation par un professionnel de santé uniquement. Cet équipement/
système peut provoquer des interférences radio ou perturber le fonctionnement des appareils situés à proximité. Il peut
être nécessaire d’adopter des mesures d’atténuation telles que la réorientation ou la relocalisation de la surface
d’appui Isolibrium, ou le blindage du lieu.
www.stryker.com 2971-209-001 REV C 2-27
Page 68

Français
FR
Garantie
Stryker Medical, une division de Stryker Corporation (« Stryker »), garantit que son produit Isolibrium™ 2971/2972est
exempt de tout défaut de matériau et de main d’œuvre. Cette garantie Stryker couvre uniquement les éléments suivants
du produit Stryker Isolibrium (chacun étant nommé individuellement « Pièce » et collectivement « Produit » ou « produit
Isolibrium ») dans le cadre d’une utilisation normale* tel que mentionné ci-après :
Garantie de 2 ans comprenant les frais de déplacement, de main-d'œuvre et de remplacement des pièces et 1
inspection annuelles de maintenance préventive.
• Période de garantie de la surface d’appui (cellules d’air, poches de rotation et matelas en mousse) : 2 ans
• Période de garantie des housses et de la protection incendie : 2 ans
• Période de garantie de la pompe : 2 ans
Les périodes de garantie spécifiées ci-dessus s’appliquent uniquement à l’acheteur initial du produit Isolibrium. Elles
démarrent à la date de livraison chez ledit acheteur initial.
Si Stryker considère, à sa seule discrétion, qu’une ou plusieurs pièces sont défaillantes dans le cadre des périodes de
garantie susmentionnées, elle peut décider de réparer ou remplacer le produit ou la pièce Isolibrium.
La garantie susmentionnée n’inclut et ne couvre pas les problèmes suivants :
• usure et déchirure anormales du produit, ou usure montrant que le produit n’a pas été correctement entretenu
(conformément aux instructions du manuel d’utilisation/de maintenance), ou produit ayant subi des contraintes
inhabituelles ;
• produit mal utilisé, modifié, rénové ou réparé sans l’accord écrit préalable de Stryker ;
• endommagement ou panne du produit dont les causes échappent au contrôle de Stryker, notamment, mauvaise
manipulation, vol, incendie, inondation, vent, foudre, gel, encrassement des pores de la surface d’appui causé par
la fumée de cigarettes, conditions atmosphériques anormales ou dégradation du matériel causée par une
exposition à l’humidité ;
• dommage considéré comme ayant été causé par l’utilisation du produit pour le transfert ou le transport de patients ;
• produit dont les numéros de série ou d’autres marques d’identification ont été enlevés ou effacés.
*« Utilisation normale » signifie toute utilisation du produit dans des conditions d’utilisation classiques ou normales
propres à un hôpital ou un établissement médical. Tout dommage causé au produit suite à une utilisation anormale, tel
qu’un dommage ayant pu être provoqué par des piqûres d’aiguille, des brûlures, des produits chimiques, une négligence,
un entretien ou un nettoyage inapproprié (tel qu’il est décrit dans le manuel d’utilisation/de maintenance) ou l’apparition
de taches résultant de telles utilisations anormales, ne sont pas couverts par la garantie suscitée.
Exclusion de garantie et limitations des dommages
La garantie expresse décrite ici est la seule garantie appliquée au produit. Toute autre garantie, qu’elle soit expresse
ou implicite, y compris toute garantie de qualité marchande ou d’adéquation à un usage particulier, est
expressément exclue par Stryker. En aucun cas, Stryker ne pourra être tenue responsable de tout dommage
accessoire ou indirect.
Pièces de rechange et service technique
Les produits Stryker bénéficient du soutien d’un réseau national de réparateurs-représentants locaux spécialisés. Ces
représentants locaux formés dans nos usines ont des stocks importants de pièces détachées qui permettent de réduire
au minimum les délais de réparation. Appeler le représentant local ou contacter le service clientèle de Stryker au +1800-327-0770.
2-28 2971-209-001 REV C www.stryker.com
Page 69

Garantie
Autorisation de retour
Le retour de produits ne peut pas être effectué sans l’accord préalable du service clientèle de Stryker. Le numéro
d’autorisation qui sera fourni doit être inscrit sur le produit retourné. Stryker se réserve le droit de facturer des frais
d’expédition et de restockage pour les articles retournés. Les articles spéciaux, modifiés ou sans suite ne peuvent pas
faire l’objet d’un retour.
Produit endommagé
La réglementation ICC (Interstate Commerce Commission) exige que les réclamations relatives à un produit
endommagé soient effectuées dans les quinze (15) jours suivant la réception du produit. Ne pas accepter de livraisons
endommagées à moins que lesdits dommages ne soient signalés sur le bordereau de livraison au moment de la
réception. Dès la réception de la notification prompte, Stryker soumettra une réclamation au transporteur approprié pour
les dommages encourus. Le montant des réclamations sera limité au coût de remplacement réel. Si cette information
n'est pas reçue par Stryker dans les quinze (15) jours suivant la livraison du produit, ou si les dommages ne sont pas
signalés sur le bordereau de livraison au moment de la réception, le client restera redevable du paiement intégral de la
facture d'origine dans un délai de trente (30) jours à compter de la réception. Les réclamations pour livraison
incomplète doivent être déposées dans les trente (30) jours suivant la date de la facture.
Clause de garantie internationale
Français
FR
La présente garantie reflète les dispositions en vigueur aux États-Unis. Hors des États-Unis, la garantie peut différer
selon le pays. Contacter le représentant Stryker Medical local pour obtenir plus d’informations.
www.stryker.com 2971-209-001 REV C 2-29
Page 70

Page 71

Page 72

Stryker Medical
3800 E. Centre Avenue
Portage, MI 49002
USA
2017/08
2971-209-001 REV C
www.stryker.com
 Loading...
Loading...