Smiths Cadd-Legacy 1 PCA Plus 6500 User manual

H C E
T
N
I
C
A
A
M
L
N
U
A
L
|
|
|
|
|
|
|
|
|
|
|
|
|
|
™ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
y |
|
S |
|
|
|
|
|
|
|
|
|
|
|
|
|
c |
|
|
P |
|
|
|
|
|
|
|
|
|
|
|
|
a |
|
|
M |
||
|
|
|
|
|
|
|
|
|
|
g |
|
|
|
P |
U |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
N |
|
||
|
|
|
|
|
|
|
|
|
e |
|
I |
O |
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
L |
|
S |
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
U |
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
F |
|
|
|
|
|
|
||
|
|
|
|
|
D |
IN |
|
|
|
|
|
|
||||
C |
A |
D |
|
|
|
Y |
|
|
|
|
|
|
|
|
||
|
|
|
|
|
R |
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
T |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
A |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
L |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
U |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
B |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
M |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
A |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CADD-Legacy™ 1 Pump
CADD-Legacy™ PCA Pump
CADD-Legacy™ PLUS Pump
Deltec
44

For detailed instructions, specifications, warnings, warranties, and additional information on operating CADD® pumps, please refer to the Operator’s Manual supplied with the product. If you have additional comments or questions concerning the operation of CADD® pumps, please call this number: 800-426-2448. Our staff is available to help you twenty-four hours a day with the programming and operation of CADD® pump infusion systems.
The issue date of this Technical Manual is included on the back cover for the user’s information. In the event one year has elapsed between the issue date and product use, the user should contact SIMS Deltec, Inc. to see if a later revision of this manual is available.
Issue Date: January 2000
41

Table of Contents
1. Introduction ............................................. |
1 |
Limited Warranty .......................................... |
1 |
Exposing CADD® Pumps to Radiation, Ultrasound, or MRI or use near ECG equipment ... 1
2. CADD-Legacy™ Pump |
|
|
|
Delivery Modes .............................................. |
2 |
|
Specifications (Nominal) ................................ |
5 |
|
Compatible Medication Cassette™ Reservoirs |
|
|
and CADD® Administration Sets ................... |
7 |
|
Remote Dose Cord ......................................... |
7 |
3. |
Batteries ................................................... |
8 |
|
Battery Compatibility ..................................... |
8 |
|
DURACELL® Alkaline Battery Life ............... |
8 |
4. |
Construction .......................................... |
11 |
5. |
Theory of Operation .............................. |
12 |
|
Keyboard Circuitry ...................................... |
12 |
|
Data Memory in Real Time Clock RAM ...... |
12 |
|
EEPROM .................................................... |
12 |
|
Battery Backed RAM ................................... |
12 |
|
Time Base Circuitry ..................................... |
12 |
|
LCD Circuitry .............................................. |
12 |
|
LED Indicator ............................................. |
13 |
|
Flash PROM Technology ............................. |
13 |
|
Audible Alarm Circuitry ............................... |
13 |
|
Watchdog Timer Circuit .............................. |
13 |
|
Motor Drive/Motor Watchdog Circuit ......... |
13 |
|
Power Circuitry ............................................ |
14 |
|
Voltage Reference Circuit ............................. |
14 |
|
Pumping Mechanism .................................... |
14 |
|
Pumping Characteristics ............................... |
15 |
|
Air Detector ................................................. |
15 |
|
Downstream Occlusion Sensor .................... |
16 |
|
Upstream Occlusion Sensor ......................... |
16 |
|
Cassette Attachment Detection ..................... |
16 |
6. Safety Features and Fault Detection ....... |
17 |
Hardware Safety Features ............................ |
17 |
Software Safety Features .............................. |
19 |
7. Hardware and Software Fault Detection 20 |
|
Overview ..................................................... |
20 |
Order of Error Code Events ......................... |
20 |
8. Cleaning and Inspection Procedures ....... |
21 |
Inspection Recommendation ........................ |
21 |
Cleaning ....................................................... |
21 |
Battery Contact Cleaning ............................. |
21 |
Visual Inspection .......................................... |
22 |
Mechanical Inspection .................................. |
22 |
9. Testing Procedures ................................. |
23 |
Testing Recommendation ............................. |
23 |
Changing to Lock Level 0 (LL0) .................. |
23 |
CADD-Legacy™ PCA pump ................... |
23 |
Air Detector Test ...................................... |
25 |
Upstream Occlusion Sensor Test ............... |
25 |
Occlusion Pressure Range Tests ................ |
25 |
Accuracy Testing ....................................... |
27 |
CADD-Legacy™ 1 pump ........................ |
29 |
Air Detector Test ...................................... |
30 |
Upstream Occlusion Sensor Test ............... |
30 |
Occlusion Pressure Range Tests ................ |
31 |
Accuracy Testing ....................................... |
32 |
CADD-Legacy™ PLUS pump .................. |
34 |
Air Detector Test ...................................... |
35 |
Upstream Occlusion Sensor Test ............... |
36 |
Occlusion Pressure Range Tests ................ |
36 |
Accuracy Testing ....................................... |
37 |
Cleaning and Functional Testing |
|
Checklist ................................................ |
40 |
42

1 Introduction
This Technical Manual is intended to provide an understanding of the mechanical and electrical operation of the CADD-Legacy™ PCA, CADD-Legacy™ 1, and CADD-Legacy™ PLUS Computerized Ambulatory Drug Delivery pumps to persons familiar with these devices. The CADD-Legacy™ PCA, CADD-Legacy™ 1, and CADD-Legacy™ PLUS pump Operator’s Manuals should be used in conjunction with this publication for complete information.
This manual also outlines cleaning and functional testing procedures that can be performed on the CADD-Legacy™ PCA, CADD-Legacy™ 1, and CADD-Legacy™ PLUS pumps.
WARNING:
This Technical Manual must be used by Biomedical Technicians only. Do not permit patients to have access to this manual. Do not disclose to the patient the pump’s security codes or any other infomation that would allow the patient complete access to all programming and operating functions. Improper programming could result in death or serious injury to the patient.
IMPORTANT NOTICE:
CADD-Legacy™ PCA, CADD-Legacy™ 1, and CADD-Legacy™ PLUS pump operations and safety features are based on a microcomputer design. Inadequate servicing or tampering with the safety features of the pumps may seriously affect performance and safety.
For that reason, ALL SERVICING AND REPAIR OF THE CADD-Legacy™ PUMPS
MUST BE PERFORMED BY DELTEC OR ITS AUTHORIZED AGENTS.
The manufacturer’s warranty agreement shall become null and void if the pump is not used in accordance with the Operator’s Manual and Instructions for Use for the pump accessories; or, the pump is serviced by persons other than Deltec or those authorized by Deltec.
Limited Warranty
The limited warranty associated with the CADD-Legacy™ PCA, CADD-Legacy™ 1, and CADD-Legacy™ PLUS pumps can be found in the product literature supplied with the product when originally purchased, which is incorporated herein by reference. DELTEC SPECIFI-
CALLY DISCLAIMS ANY OTHER WARRANTY, WHETHER EXPRESS, IMPLIED OR STATUTORY, INCLUDING, WITHOUT LIMITATION, ANY IMPLIED WARRANTY OF MERCHANTABILITY OR FITNESS FOR USE. Deltec further disclaims responsibility for the suitability of the system for a particular medical treatment or for any medical complications resulting from the use of the system. The manufacturer shall not be responsible for any incidental damages or consequential damages to property, loss of profits, or loss of use caused by any defect or malfunction of the system.
If you wish to receive additional information about the extent of the warranty on these products, please contact your Deltec representative or call Customer Service at 1-800-426-2448.
All recommendations, information, and literature supplied by Deltec with respect to the CADD® product line are believed to be accurate and reliable, but do not constitute warranties. No agent, representative, or employee of Deltec has authority to bind Deltec to any representation or warranty, expressed or implied.
Exposure to Radiation, Ultrasound or Magnetic Resonance Imaging (MRI), or use near ECG equipment
CAUTION:
• Do not expose the pump to therapeutic levels of ionizing radiation as permanent damage to the pump’s electronic circuitry may occur. The best procedure to follow is to remove the pump from the patient during therapeutic radiation sessions. If the pump must remain in the vicinity during a therapy session, it should be shielded, and its ability to function properly should be confirmed following treatment.
• Do not expose the pump directly to ultrasound, as permanent damage to the pump’s electronic circuitry may occur.
• Do not use the pump in the vicinity of magnetic resonance imaging (MRI) equipment as magnetic fields may adversely affect the operation of the pump. Remove the pump from the patient during MRI procedures and keep it at a safe distance from magnetic energy.
• Do not use the pump near ECG equipment as the pump may interfere with the operation of the equipment. Monitor ECG equipment care-
fully when using this pump.
1
2 CADD-Legacy™ Pump
Delivery Modes
The CADD-Legacy™ ambulatory drug delivery pump provides measured drug therapy to patients in hospital or outpatient settings. The CADD-Legacy™ pump is indicated for intravenous, intra-arterial, subcutaneous, intraperitoneal, epidural space, or subarachnoid space infusion.
Epidural administration is limited to use with indwelling catheters for short term delivery of anesthetics and short or long term delivery of analgesics. Subarachnoid administration is limited to use with indwelling catheters for short-term delivery of analgesics.
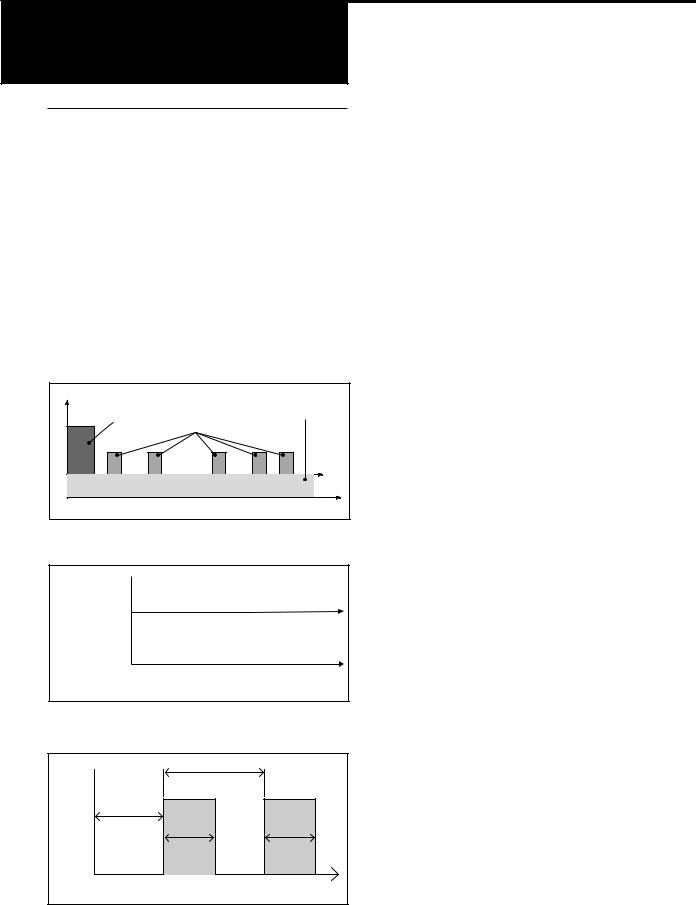
Clinician Bolus |
Continuous Rate |
(used here as a loading dose) |
Demand Doses |
Dosage |
Time
Figure 1. PCA mode delivery profile.
Delivery
Rate
CADD-Legacy™ PLUS
(ML/HR)
or
CADD-Legacy™ 1
(ML/24 HR)
Time
Continuous Delivery
Figure 2. Continuous mode delivery profile.
|
|
Dose Cycle |
|
|
Dose |
Dose |
Dose |
Dose |
Starts |
||
Volume |
in |
|
|
|
|
Duration |
Duration |
|
|
Time |
|
|
|
Intermittent Delivery |
|
Figure 3. Intermittent mode delivery profile.
The CADD-Legacy™ PCA pump may be programmed to deliver medication in one of three ways: 1) continuous rate only, 2) patientactivated dose only and 3) continuous rate and patient-activated dose. (See figure 1.)
The CADD-Legacy™ PLUS pump may be programmed to deliver in one of two modes:
(1) Continuous, (2) Intermittent . (See figures 2 and 3.)
The CADD-Legacy™ 1 pump operates in continuous mode. (See figure 2.)
Figure 4 shows a diagram of the CADDLegacy™ pump.
PCA Delivery Profile
The PCA (patient-controlled analgesia) delivery mode is used for therapies that require a continuous rate of infusion, patient-controlled demand doses, or both, such as patientcontrolled analgesia.
Continuous Mode Delivery Profile
The Continuous delivery mode allows the infusion of drug at a constant, programmed rate.
Intermittent Mode Delivery Profile
The Intermittent delivery mode allows the infusion of a specific volume of drug at regular programmed intervals.
2
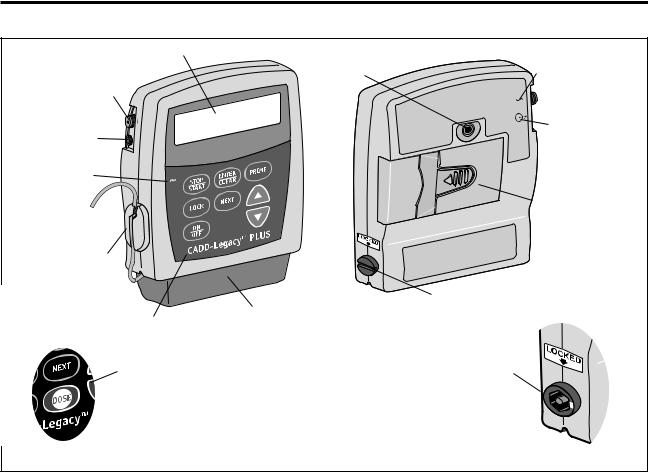
Display |
Threaded |
Power Jack |
|
Mounting |
symbol |
Power Jack |
Hole |
|
|
|
|
Accessory |
|
Accessory |
Jack |
|
Jack Symbol |
AC Indicator |
|
|
Light |
|
|
|
|
Battery |
|
|
Compartment |
Air Detector |
|
|
|
® |
|
|
Cassette Lock |
|
Keypad |
Cassette |
|
Dose Key on |
CADD-Legacy™ PCA |
|
Cassette Lock |
|
|
CADD-Legacy™ PCA |
|
|
Front View Rear View
Figure 4. Front and Rear views of the CADD-Legacy™ Pump. Features are identical on all CADD-Legacy™ pumps except as illustrated for the CADD-Legacy™ PCA pump.
3

PCA Delivery Mode Scroll Ranges
Units |
Starting |
Increment |
|
Maximum |
ML |
0.10 |
All values: |
0.10 |
50.00 |
|
10% of |
Values between 0.01 and 0.5: |
0.01 |
Concentration |
MG |
Concentration |
Values between 0.50 and 100.0: |
0.10 |
x 50 |
|
|
Values between 100.0 and 1000.0: |
1.00 |
|
|
|
Values greater than 1000.0: |
10.0 |
|
MCG |
10% of |
Values between 0.1 and 100: |
0.10 |
Concentration |
|
Concentration |
Values between 100 and 1000: |
1.00 |
x 50 |
|
|
Values greater than 1000: |
10.00 |
|
|
|
|
|
|
Table 1. PCA delivery mode: continuous rate scroll ranges.
|
|
|
Milligrams |
|
|
|
|
|
|
|
|
Micrograms |
|
|
|
||
Concentration |
|
Demand Dose |
Clinician Bolus |
|
|
|
Concentration |
|
Demand Dose |
Clinician Bolus |
|||||||
mg/ml |
increment |
max. |
increment |
max. |
|
|
|
mcg/ml |
increment max. |
increment |
max. |
||||||
0.1 |
0.01 |
0.99 |
0.01 |
2 |
|
|
|
1 |
0.05 |
9.9 |
0.05 |
20 |
|
|
|||
0.2 |
0.02 |
1.98 |
0.02 |
4 |
|
|
2 |
0.10 |
19.8 |
0.10 |
40 |
|
|
||||
0.3 |
0.03 |
2.97 |
0.03 |
6 |
|
|
3 |
0.15 |
29.7 |
0.15 |
60 |
|
|
||||
0.4 |
0.04 |
3.96 |
0.04 |
8 |
|
|
4 |
0.20 |
39.6 |
0.20 |
80 |
|
|
||||
0.5 |
0.05 |
4.95 |
0.05 |
10 |
|
|
5 |
0.25 |
49.5 |
0.25 |
100 |
|
|
||||
1 |
0.05 |
9.9 |
0.05 |
20 |
|
|
|
10 |
0.50 |
99.0 |
0.50 |
200 |
|
|
|||
2 |
0.10 |
19.8 |
0.10 |
40 |
|
|
15 |
0.75 |
148.5 |
0.75 |
300 |
|
|
||||
3 |
0.15 |
29.7 |
0.15 |
60 |
|
|
20 |
1.00 |
198.0 |
1.00 |
400 |
|
|
||||
4 |
0.20 |
39.6 |
0.20 |
80 |
|
|
|
25 |
1.25 |
247.5 |
1.25 |
500 |
|
|
|||
5 |
0.25 |
49.5 |
0.25 |
100 |
|
|
30 |
1.50 |
297.0 |
1.50 |
600 |
|
|
||||
10 |
0.50 |
99.0 |
0.50 |
200 |
|
|
35 |
1.75 |
346.5 |
1.75 |
700 |
|
|
||||
|
|
|
|
|
|
|
|
|
40 |
2.00 |
396.0 |
2.00 |
800 |
|
|
||
15 |
0.75 |
148.5 |
0.75 |
300 |
|
|
|
|
|
||||||||
20 |
1.00 |
198.0 |
1.00 |
400 |
|
|
45 |
2.25 |
445.5 |
2.25 |
900 |
|
|
||||
25 |
1.25 |
247.5 |
1.25 |
500 |
|
|
50 |
2.50 |
495.0 |
2.50 |
1000 |
|
|
||||
30 |
1.50 |
297.0 |
1.50 |
600 |
|
|
55 |
2.75 |
544.5 |
2.75 |
1100 |
|
|
||||
35 |
1.75 |
346.5 |
1.75 |
700 |
|
|
60 |
3.00 |
594.0 |
3.00 |
1200 |
|
|
||||
40 |
2.00 |
396.0 |
2.00 |
800 |
|
|
65 |
3.25 |
643.5 |
3.25 |
1300 |
|
|
||||
45 |
2.25 |
445.5 |
2.25 |
900 |
|
|
70 |
3.50 |
693.0 |
3.50 |
1400 |
|
|
||||
50 |
2.50 |
495.0 |
2.50 |
1000 |
|
|
75 |
3.75 |
742.5 |
3.75 |
1500 |
|
|
||||
55 |
2.75 |
544.5 |
2.75 |
1100 |
|
|
80 |
4.00 |
792.0 |
4.00 |
1600 |
|
|
||||
60 |
3.00 |
594.0 |
3.00 |
1200 |
|
|
85 |
4.25 |
841.5 |
4.25 |
1700 |
|
|
||||
65 |
3.25 |
643.5 |
3.25 |
1300 |
|
|
90 |
4.50 |
891.0 |
4.50 |
1800 |
|
|
||||
70 |
3.50 |
693.0 |
3.50 |
1400 |
|
|
95 |
4.75 |
940.5 |
4.75 |
1900 |
|
|
||||
75 |
3.75 |
742.5 |
3.75 |
1500 |
|
|
100 |
5.00 |
990.0 |
5.00 |
2000 |
|
|
||||
80 |
4.00 |
792.0 |
4.00 |
1600 |
|
|
200 |
10.00 |
1980.0 |
10.00 |
4000 |
|
|
||||
85 |
4.25 |
841.5 |
4.25 |
1700 |
|
|
300 |
15.00 |
2970.0 |
15.00 |
6000 |
|
|
||||
90 |
4.50 |
891.0 |
4.50 |
1800 |
|
|
|
400 |
20.00 |
3960.0 |
20.00 |
8000 |
|
|
|||
95 |
4.75 |
940.5 |
4.75 |
1900 |
|
|
500 |
25.00 |
4950.0 |
25.00 |
10000 |
|
|
||||
100 |
5.00 |
990.0 |
5.00 |
2000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table 3. Demand dose, clinician bolus scroll ranges, |
||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|||||||||||
Table 2. Demand dose, clinician bolus scroll ranges, |
|
|
micrograms |
|
|
|
|
|
|
|
|||||||
milligrams |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Milliliters |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Demand Dose |
|
Clinician Bolus |
|
||||
|
|
|
|
|
|
|
|
|
|
increment |
|
max. |
increment |
max. |
|||
|
|
|
|
|
|
|
|
|
0.05 |
9.9 |
|
0.05 |
20 |
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4 |
Table 4. Demand dose, clinician bolus scroll ranges, |
milliliters |

Specifications (Nominal)
General Pump Specifications
Resolution
Medication Cassette™ Reservoir or CADD® Administration Set, 0.050 ml/pump stroke nominal
Size
4.1 cm x 9.5 cm x 11.2 cm (1.6 in. x
3.8 in. x 4.4 in.) excluding cassette or other accessories
Weight
391 g (13.8 oz.) including 2 AA batteries, empty 100-ml Medication Cassette™ Reservoir, and air detector, excluding other accessories
Pump Alarms
Low battery power; depleted battery power; battery dislodged; pump stopped; pump fault; low reservoir volume; high delivery pressure; air in line; disposable not attached when run attempted; motor locked; upstream occlusion; reservoir volume empty; program incomplete; remote dose cord removed; key stuck; disposable detached, power removed, value not saved.
Bolus Volume at Occlusion Alarm Pressure
0.050 ml resolution sets/reservoirs: < 0.15 ml
Power Sources
Two AA alkaline batteries such as DURACELL® or EVEREADY Energizer®; AC adapter.
An internal battery powers the clock. When it is depleted, it cannot reliably maintain the clock time. This battery must be replaced by SIMS Deltec, Inc. The internal battery has an expected life of 5 years.
System Operating Temperature*
+2°C to 40°C (35°F to 104°F)
System Storage Temperature*
-20°C to 60°C (-4°F to 140°F)
System Delivery Accuracy*
± 6% (nominal)
High Pressure Alarm
26 (±14) psi, 1.79 (± 0.97) bar
Air Detector Alarm
Single bubble
Low sensitivity = greater than 0.250 ml High sensitivity = greater than 0.100 ml
Multi-bubble = 1.0 ml nominal
Delivery Mode Specifications
CADD-Legacy™ PCA pump
Reservoir Volume
1 to 9999 or Not In Use; programmable in
1 ml increments, displayed in 0.1 ml increments
Default: 1 ml
Units
Milliliters (ml), milligrams (mg), micrograms (mcg)
Default: milligrams
Concentration
Mg/ml: 0.1, 0.2, 0.3, 0.4, 0.5, 1, 2, 3, 4, 5, 10, 15, ... 95, 100 (Default: 100 mg/ml) Mcg/ml: 1, 2, 3, 4, 5, 10, 15, ...95, 100, 200, 300, 400, 500 (Default: 500 mcg/ml)
Continuous Rate
0 to 50 ml/hr (or the mg or mcg equivalent) (See Table 1 for scroll ranges)
Demand Dose
0 to 9.9 ml in 0.05 ml increments (or the mg or mcg equivalent)
(See Tables 2 and 3 for scroll ranges) Delivery rate (Continuous Rate + Demand Dose): 125 ml/hr nominal
Dose Lockout
5 minutes to 24 hours in the following increments:
1 minute for values between 5 and 20 minutes
5 minutes between 20 minutes and 24 hours
Default: 24 hours
Doses Per Hour
1 to 12 doses in 1 dose increments (will also be limited by the Demand Dose Lockout value)
Default: 1 dose/hr
*System is defined as a CADD-Legacy™ pump with an attached Medication Cassette™ Reservoir and CADD® Extension Set with |
5 |
integral anti-siphon valve, or an attached CADD® Administration Set with integral or add-on anti-siphon valve. |
|

Doses Given
0 to 999
Doses Attempted
0 to 999
Given
0 to 99999.95 in 0.05 unit increments or
0 to 99999.99 in 0.01 unit increments
(increments converted to current units based on concentration)
Clinician Bolus
0.05 ml to 20.00 ml (or mg or mcg equivalent) (See Tables 1, 2 and 3 for scroll ranges)
Delivery rate (Continuous Rate + Clinician Bolus): 125 ml/hr nominal
CADD-Legacy™ 1 pump
Continuous Delivery Mode Specifications
Reservoir Volume
1 to 9999 or Not In Use; programmable in
1 ml increments, displayed in 0.1 ml increments
Default: 1 ml
Continuous Rate
1 to 3000 ml/24 hr in increments of
1 ml/24hr Default: 0 ml/24hr
Given
0 to 99999.95 in 0.05 ml increments
CADD-Legacy™ PLUS pump
Intermittent Delivery Mode Specifications
Reservoir Volume
1 to 9999 or Not In Use; programmable in
1 ml increments, displayed in 0.1 ml increments
Default: 1 ml
Dose Volume
0.1 to 1000.0 ml in increments of 0.1 Default: 0.0 ml
Dose Duration
1 minute to 24 hours in the following increments:
1 minute from 00:01 to 00:10
5 minutes from 00:10 to 24:00 Default: 30 minutes
Dose Cycle
10 minutes to 96 hours in 5 minute increments
Default: 4 hours
KVO Rate
0 to 125.0 ml/hr in increments of 0.1 ml/hr Default: 0 ml/hr
Dose Starts in
Immediate or 1 minute to 96 hours in the following increments:
00:01 from 00:00 to 00:10 00:05 from 00:10 to 96:00 Default: Immediate
Continuous Delivery Mode Specifications
Reservoir Volume
1 to 9999 or Not In Use; programmable in
1 ml increments, displayed in 0.1 ml increments
Default: 1 ml
Continuous Rate
0.1 ml/hr to 125.0 ml/hr in increments of
0.1 ml/hr Default: 0.0 ml/hr
Given
0 to 99999.95 in 0.05 ml increments
Biomed Functions Specifications
Air Detector Status:
Off
Onlow
Onhigh
Default: On-high
Upstream Occlusion Status:
Off
On
Default: On
Delivery Mode (CADD-Legacy™ PLUS only):
Continuous
Intermittent Default: Intermittent
6

Compatible Medication Cassette™
Reservoirs and CADD® Administration
Sets
•50-ml or 100-ml Medication Cassette™ Reservoir, used with the CADD® Extension Set with Anti-siphon Valve.
•CADD® Administration Set with integral anti-siphon valve, with or without bag spike (allows use of flexible plastic bag or sterile vial with injector)
•CADD® Administration Set with add-on anti-siphon valve and bag spike (allows for gravity priming before attaching the add on anti-siphon valve)
Remote Dose Cord
Deltec provides a Remote Dose Cord for the CADD-Legacy™ PCA pump which is an extension of the Íkey. The push button is a Single Pole Double Throw (SPDT) switch which operates in the same manner as the Í key. When the Remote Dose Cord is attached to the pump, the patient may press either the Remote Dose button or the Íkey to receive a Demand Dose. The clinician may also use either the Remote Dose button or the Íkey to deliver a clinician-activated bolus. For easy access, the Remote Dose Cord may be fastened to the patient’s clothing or bedsheet with the attached clip.
There is an alarm/function present in the CADD-Legacy™ PCA pump. If the Remote Dose Cord is removed, the display shows a message “Remote Dose Removed”. The pump sounds an audible alarm until the „key is pressed to acknowledge the alarm.
NOTE:
To detach the Remote Dose Cord from the pump, grasp the Remote Dose Cord connector and pull back using a straight, steady motion.
7

3 Batteries
Battery Compatibility
Recommended Batteries
Two AA alkaline batteries are recommended for use in the CADD-Legacy™ pumps. Carbonzinc, mercury, nickel-cadmium, nickel-metal- hydride, or zinc-air AA batteries should not be used.
Battery Life
The CADD-Legacy™ pumps have been designed to provide optimal battery life. The expected battery life in the CADD-Legacy™ pumps depends on the following factors:
•Programmed delivery rate
•Operating temperatures
•Battery type and brand
•Battery age
DURACELL® Alkaline Battery Life
Recommended storage conditions are 10°C to 25°C (50°F to 77°F) with no more than 65% relative humidity noncondensing.
The following table may be used to predict typical alkaline battery life at different delivery rates when alkaline batteries are used in the CADD-Legacy™ pump. As expected, battery life decreases as the delivery rate increases. This table is based on laboratory tests using fresh DURACELL® alkaline batteries in CADD-Legacy™ pumps while the pumps were operating at room temperature.
Actual battery life may be significantly shorter depending on the operating temperature and the storage conditions of the battery.
Battery life is shortened significantly at very low operating temperatures. For example, at 0°C (32°F), an alkaline battery will yield approximately 30% of its normal capacity.
Alkaline batteries do not need to be stored in a refrigerator. After four years of storage at 21°C (70°F), an alkaline battery retains approximately 86% of its original capacity. Battery life will be shorter if the battery is stored above room temperature. An alkaline battery stored at 43°C (110°F) will be down to approximately 80% of its capacity within one year.
Continuous Delivery Battery Life with Alkaline Batteries
Rate |
Life |
Volume |
0.4 ml/hr |
338 hrs |
135 ml |
4 ml/hr |
178 hrs |
712 ml |
10 ml/hr |
112 hrs |
1120 ml |
15 ml/hr |
96 hrs |
1440 ml |
30 ml/hr |
53 hrs |
1590 ml |
75 ml/hr |
18 hrs |
1350 ml |
125 ml/hr |
15 hrs |
1875 ml |
Table 5. Two AA Alkaline-type batteries used with the CADD-Legacy™ pumps.
8

Intermittent Delivery Battery Life with Alkaline Batteries
Dose Volume |
Duration |
Dose Cycle |
KVO |
Life |
Volume |
23.5 ml |
1:00 hr |
5:00 hr |
0.2 ml/hr |
193 hr |
915 ml |
61 ml |
1:00 hr |
6:00 hr |
0.2 ml/hr |
120 hr |
1224 ml |
125 ml |
1:00 hr |
6:00 hr |
0.2 ml/hr |
65 hr |
1356 ml |
|
|
|
|
|
|
Table 6. Two AA Alkaline-type batteries used with the CADD-Legacy™ pumps.
|
135 |
|
|
|
|
|
|
|
|
|
|
120 |
|
|
|
|
|
|
|
|
|
|
105 |
|
|
|
|
|
|
|
|
|
(ml/hr) |
90 |
|
|
|
|
|
|
|
|
|
75 |
|
|
|
|
|
|
|
|
|
|
60 |
|
|
|
|
|
|
|
|
|
|
Rate |
|
|
|
|
|
|
|
|
|
|
45 |
|
|
|
|
|
|
|
|
|
|
30 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
15 |
|
|
|
|
|
|
|
|
|
|
0 |
20 |
30 |
40 |
50 |
60 |
70 |
80 |
90 |
100 |
|
10 |
|||||||||
|
|
|
|
|
Hours |
|
|
|
|
|
Figure 5. Operating time to low battery alarm using alkaline batteries.
|
18 |
|
|
|
|
|
|
|
16 |
|
|
|
|
|
|
|
14 |
|
|
|
|
|
|
(ml/hr) |
12 |
|
|
|
|
|
|
10 |
|
|
|
|
|
|
|
8 |
|
|
|
|
|
|
|
Rate |
|
|
|
|
|
|
|
6 |
|
|
|
|
|
|
|
4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 |
|
|
|
|
|
|
|
0 |
|
|
200 |
|
|
|
|
50 |
100 |
150 |
250 |
300 |
350 |
|
|
|
|
|
Hours |
|
|
|
Figure 6. Operating time to low battery alarm using alkaline batteries.
9
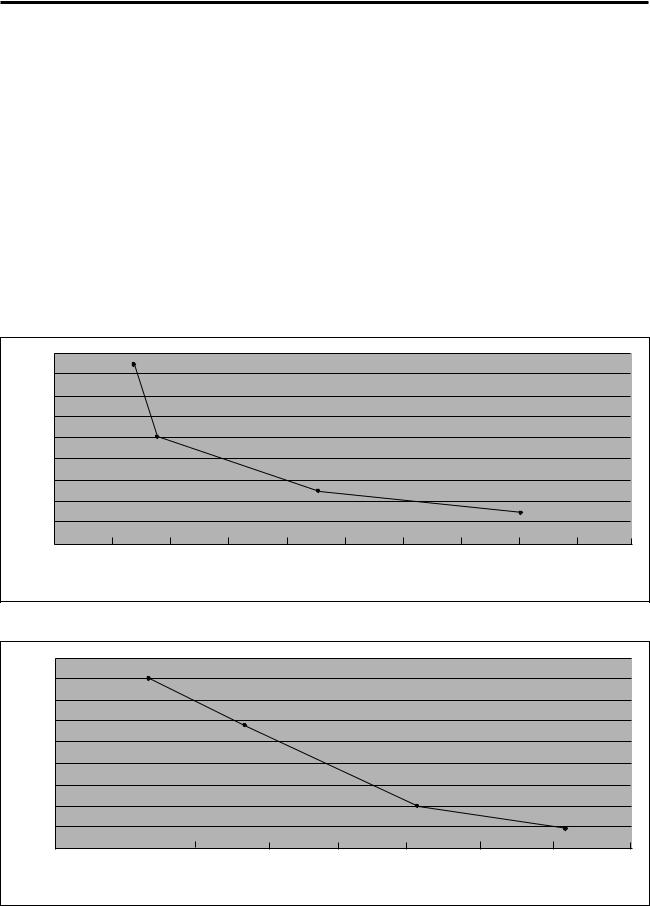
Continuous Delivery Battery Life with Lithium Batteries
Rate |
Life |
Volume |
0.4 ml/hr |
413 hrs |
165 ml |
4 ml/hr |
307 hrs |
1228 ml |
10 ml/hr |
190 hrs |
1900 ml |
15 ml/hr |
163 hrs |
2445 ml |
30 ml/hr |
90 hrs |
2700 ml |
75 ml/hr |
33 hrs |
2475 ml |
125 ml/hr |
22 hrs |
2750 ml |
|
|
|
Table 7. Two AA Lithium-type batteries used with the CADD-Legacy™ pumps.
Intermittent Delivery Battery Life with Lithium Batteries
Dose Volume |
Duration |
Dose Cycle |
KVO |
Life |
Volume |
23.5 ml |
1:00 hr |
5:00 hr |
0.2 ml/hr |
300 hrs |
1458 ml |
61 ml |
1:00 hr |
6:00 hr |
0.2 ml/hr |
185 hrs |
1911 ml |
125 ml |
1:00 hr |
6:00 hr |
0.2 ml/hr |
125 hrs |
2625 ml |
|
|
|
|
|
|
Table 8. Two AA Lithium-type batteries used with the CADD-Legacy™ pumps.
|
135 |
|
|
|
|
|
|
|
|
|
|
120 |
|
|
|
|
|
|
|
|
|
|
105 |
|
|
|
|
|
|
|
|
|
(ml/hr) |
90 |
|
|
|
|
|
|
|
|
|
75 |
|
|
|
|
|
|
|
|
|
|
60 |
|
|
|
|
|
|
|
|
|
|
Rate |
|
|
|
|
|
|
|
|
|
|
45 |
|
|
|
|
|
|
|
|
|
|
30 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
15 |
|
|
|
|
|
|
|
|
|
|
0 |
40 |
60 |
80 |
100 |
120 |
140 |
160 |
180 |
200 |
|
20 |
|||||||||
|
|
|
|
|
Hours |
|
|
|
|
|
Figure 7. Dual-stroke operating time on lithium batteries.
|
18 |
|
|
|
|
|
|
|
|
|
16 |
|
|
|
|
|
|
|
|
|
14 |
|
|
|
|
|
|
|
|
(ml/hr) |
12 |
|
|
|
|
|
|
|
|
10 |
|
|
|
|
|
|
|
|
|
8 |
|
|
|
|
|
|
|
|
|
Rate |
|
|
|
|
|
|
|
|
|
6 |
|
|
|
|
|
|
|
|
|
4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 |
|
|
|
|
|
|
|
|
|
0 |
100 |
150 |
200 |
250 |
300 |
350 |
400 |
|
|
50 |
450 |
|||||||
|
|
|
|
|
Hours |
|
|
|
|
Figure 8. Single-stroke operating time on lithium batteries.
10

4 Construction
The pump’s housing is made of a special high impact plastic. It is composed of two sections: the rear and front housing. The pump housing is sealed to ensure that the pump is water resistant. The battery compartment is not water resistant.
NOTE:
The CADD-Legacy™ ambulatory infusion pump is water resistant, but not waterproof. The pump is “Splashproof” and is characterized by the IEC code of IPX4.
The battery compartment is accessed through a removable door on the rear housing. Within the battery compartment is space for the batteries and the four battery contacts.
On CADD-Legacy™ pumps the Medication Cassette™ Reservoir or the administration set is attached to the bottom of the pump by inserting the two hooks on the cassette into the mating hinge pins on the pump. The pump and the reservoir or the administration set are then placed in an upright position on a firm, flat surface. The reservoir or the administration set must be secured in place by inserting a coin (or key if using the CADD-Legacy™ PCA pump) in the slot on the pump’s locking button, pushing the button in and turning the button onequarter turn counter-clockwise.
NOTE:
The Medication Cassette™ Reservoir and the administration set are intended for single use only.
The keyboard, located on the front housing, is composed of eight membrane switches (nine membrane switches on the CADD-Legacy™ PCA pump) and is sealed against moisture. All of the keys contain domes to provide a tactile feel when the key is pressed. The keyboard keys are sensed by the pump’s microprocessor.
The Liquid Crystal Display (LCD), also located on the front housing, shows the pump status and programmed settings. The dot matrix display consists of 16 character columns with 2 rows of characters, and is selected by the pump’s microprocessor according to status conditions and keyboard entries.
The microprocessor and other circuitry which control the pump are located on a printed circuit board. The board contains the Central Processing Unit (CPU), motor driver circuitry, and other circuitry. The circuitry is designed to reduce susceptibility to interference from electromagnetic fields and to dissipate electrostatic discharge.
The LCD controller is mounted on the LCD using chip on glass technology.
The pumping mechanism subassembly contains the motor, gear train, camshaft, valves, expulsor, sensing disk, infrared light source, infrared detector, occlusion sensors, disposable sensor, and cassette locking button. Via the motor driver circuitry, the pump’s microprocessor controls motor rotation.
Two external port connectors are utilized for remote dose and external power input. The accessory jack is used for attachment of the Remote Dose Cord (CADD-Legacy™ PCA pump only) and interface cable. The Remote Dose Cord enables the patient to use either of two options to begin a Demand Dose when using the PCA delivery mode: (1) the Remote Dose button; or (2) the Íkey.
The second port allows connection to an AC adapter.
The keyboard is connected to the printed circuit board via a flex circuit tail. Discrete wires connect the pumping mechanism, motor, and sensors to the printed circuit board.
The accessory jack in conjunction with the interface cable allows download of events using the CADD-DIPLOMAT™ software.
11
 Loading...
Loading...