Physio-Control LIFEPAK 15 User Manual

LIFEPAK® 15 MONITOR / DEFIBRILLATOR
OPERATING INSTRUCTIONS

LIFEPAK® 15 MONITOR/DEFIBRILLATOR
OPERATING INSTRUCTIONS

Important Information
!USA 
 Rx Only
Rx Only
!USA Device Tracking
The U.S. Food and Drug Administration requires defibrillator manufacturers and distributors to track the location of their defibrillators. If the device is located somewhere other than the shipping address or the device has been sold, donated, lost, stolen, exported, destroyed, permanently retired from use, or if the device was not obtained directly from Physio-Control, please do one of the following: register the device at http://www.physio-control.com, call the device registration phone line at 1.800.426.4448, or use one of the postage-paid address change cards located in the back of this manual to update this vital tracking information.
Text Conventions
Throughout these operating instructions, special text characters (for example, CAPITAL LETTERS such as CHECK PATIENT and SPEED DIAL) are used to indicate labels, screen messages, and voice prompts.
Version History
These operating instructions describe LIFEPAK 15 monitor/defibrillator devices with software revision 3306808-001 or later.
LIFEPAK, LIFENET, QUIK-LOOK, and QUIK-COMBO are registered trademarks of Physio-Control, Inc. CODE SUMMARY, Shock Advisory System, CODE-STAT, DT EXPRESS, LIFEPAK CR, REDI-PAK, LIFEPAK EXPRESS, SunVue, and cprMAX are trademarks of Physio-Contro Inc. Bluetooth is a registered trademark of Bluetooth SIG, Inc. CADEX is a registered trademark of Cadex Electronics Inc. Microstream, CapnoLine, and FilterLine are registered trademarks of Oridion Systems Ltd. The Oridion medical capnography in this product is covered one or more of the following US patents: 6,428,483; 6,997,880; 5,300,859; 6,437,316 and their foreign equivalents. Additional paten applications pending. PC Card is a trademark of the Personal Computer Memory Card International Association. Masimo, the Radical logo Rainbow, and SET are registered trademarks of Masimo Corporation. EDGE System Technology is a trademark of Ludlow Technical Products. Formula 409 is a registered trademark of The Clorox Company. Specifications are subject to change without notice.
©2007-2013 Physio-Control, Inc. All rights reserved. |
MIN 3306222-005 |
Publication Date: 04/2013 |

|
|
CONTENTS |
|
|
|
1 |
Preface |
|
Introduction ................................................................................................................... |
1-3 |
|
Intended Use.................................................................................................................. |
1-4 |
|
Modes of Operation ......................................................................................................... |
1-5 |
|
2 |
Safety Information |
|
Terms ............................................................................................................................ |
2-3 |
|
General Dangers and Warnings ......................................................................................... |
2-3 |
|
3 |
Basic Orientation |
|
Front View ...................................................................................................................... |
3-3 |
|
Back View .................................................................................................................... |
3-13 |
|
Batteries ...................................................................................................................... |
3-15 |
|
Home Screen................................................................................................................ |
3-17 |
|
Alarms ......................................................................................................................... |
3-21 |
|
Options ........................................................................................................................ |
3-23 |
|
Events.......................................................................................................................... |
3-25 |
|
4 |
Monitoring |
|
Monitoring the ECG ......................................................................................................... |
4-3 |
|
Acquiring a 12-Lead ECG .............................................................................................. |
4-15 |
|
Monitoring SpO2, SpCO, and SpMet ............................................................................... |
4-25 |
|
Monitoring Noninvasive Blood Pressure ........................................................................... |
4-37 |
|
Monitoring ETCO2......................................................................................................... |
4-45 |
|
Monitoring Invasive Pressure .......................................................................................... |
4-54 |
|
Monitoring Continuous Temperature................................................................................ |
4-61 |
|
Vital Sign and ST Segment Trends .................................................................................. |
4-66 |
|
5 |
Therapy |
|
General Therapy Warnings and Cautions ............................................................................ |
5-3 |
|
Therapy Electrode and Standard Paddle Placement ............................................................ |
5-4 |
|
Automated External Defibrillation (AED) ............................................................................ |
5-7 |
|
Manual Defibrillation..................................................................................................... |
5-22 |
|
Synchronized Cardioversion Procedure ............................................................................ |
5-27 |
|
Noninvasive Pacing ....................................................................................................... |
5-32 |
|
Pediatric ECG Monitoring and Manual Mode Therapy Procedures ....................................... |
5-39 |
|
6 |
Paddle Accessory Options |
|
QUIK-COMBO Therapy Electrodes .................................................................................... |
6-3 |
|
Standard Paddles............................................................................................................ |
6-6 |
|
Sterilizable Internal Defibrillation Paddles ....................................................................... |
6-10 |
|
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions iii |
|
|
7 Data Management |
|
Patient Records and Reports ............................................................................................ |
7-3 |
Memory Capacity ............................................................................................................ |
7-9 |
Managing Current Patient Records .................................................................................. |
7-10 |
Managing Archived Patient Records ................................................................................ |
7-11 |
8 Data Transmission |
|
About Transmitting Patient Records and Reports |
................................................................8-3 |
Preparing the Monitor for Transmission ............................................................................. |
8-4 |
Using Bluetooth Wireless Communication.......................................................................... |
8-5 |
Using a Direct Connection.............................................................................................. |
8-10 |
Transmitting Reports ..................................................................................................... |
8-12 |
Considerations When Transmitting Data .......................................................................... |
8-14 |
Troubleshooting Tips .................................................................................................... |
8-15 |
9 Power Adapter
Basic Orientation ............................................................................................................ |
9-3 |
Using the Power Adapter.................................................................................................. |
9-5 |
General Maintenance....................................................................................................... |
9-8 |
Warranty ...................................................................................................................... |
9-10 |
10 Maintaining the Equipment
General Maintenance and Testing |
...................................................................................10-3 |
Battery Maintenance ................................................................................................... |
10-12 |
Cleaning the Device..................................................................................................... |
10-15 |
Storing the Device....................................................................................................... |
10-16 |
Loading Paper ............................................................................................................ |
10-17 |
General Troubleshooting Tips ....................................................................................... |
10-18 |
Service and Repair ..................................................................................................... |
10-22 |
Product Recycling Information ..................................................................................... |
10-23 |
Warranty .................................................................................................................... |
10-23 |
Accessories ................................................................................................................ |
10-24 |
Appendix A: Specifications and Performance Characteristics
Appendix B: Screen Messages
Appendix C: Shock Advisory System
Appendix D: Electromagnetic Compatibility Guidance
Appendix E: Symbols
iv
LIFEPAK 15 Monitor/Defibrillator Operating Instructions

 1
1
PREFACE
This chapter provides a brief introduction to the LIFEPAK® 15 monitor/defibrillator and describes the product’s intended use.
Introduction. . . . . . . . . . . . . . . . . . . . . . . . . . . . . page 1-3 Intended Use. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-4 Modes of Operation . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-5
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
1-1 |

PREFACE  1
1
Introduction
The LIFEPAK 15 monitor/defibrillator is a complete acute cardiac care response system designed for basic life support (BLS) and advanced life support (ALS) patient management protocols.
These operating instructions include information and procedures related to all features of the LIFEPAK 15 monitor/defibrillator. Your LIFEPAK 15 monitor/defibrillator may not have all of these features.
These operating instructions describe the operation of the LIFEPAK 15 monitor/defibrillator when the factory default settings are used. The factory default settings for all setup options are identified in Table A-5 on page A-16. Your device may be set up with different default settings, based on your protocols. For information about changing default settings, see the LIFEPAK 15 Monitor/ Defibrillator Setup Options provided with your device.
IMPORTANT! Some LIFEPAK 15 monitor/defibrillator accessories are not interchangeable with accessories that are used with other LIFEPAK monitor/defibrillators. Specific accessory incompatibilities are noted in the related sections.
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
1-3 |

Intended Use
Intended Use
The LIFEPAK 15 monitor/defibrillator is intended for use by trained medical personnel in out-of- doors and indoor emergency care settings within the environmental conditions specified on page A-12. The LIFEPAK 15 monitor/defibrillator is designed to be used during ground transportation except when specified otherwise.
Monitoring and therapy functions may only be used on one patient at a time. Manual mode monitoring and therapy functions are intended for use on adult and pediatric patients. Automated external defibrillation mode is intended for use on patients eight years of age and older.
For additional intended use information, and information about the indications and contraindications of the monitoring and therapy functions, see the individual sections identified below.
• |
ECG Monitoring |
See page 4-3 |
Standard feature |
• |
12-Lead Electrocardiography |
See page 4-15 |
Optional |
• |
SpO2, SpCO, and SpMet Monitoring |
See page 4-25 |
Optional |
• Noninvasive Blood Pressure Monitoring |
See page 4-37 |
Optional |
|
• |
End-Tidal CO2 Monitoring |
See page 4-45 |
Optional |
• |
Invasive Pressure Monitoring |
See page 4-54 |
Optional |
• |
Temperature Monitoring |
See page 4-61 |
Optional |
• Vital Sign and ST Segment Trends |
See page 4-66 |
Optional |
|
• |
Automated External Defibrillation |
See page 5-7 |
Standard feature |
• |
Manual Defibrillation |
See page 5-22 |
Standard feature |
• |
Noninvasive Pacing |
See page 5-32 |
Standard feature |
1-4 |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |

PREFACE  1
1
Modes of Operation
The LIFEPAK 15 monitor/defibrillator has the following modes of operation:
•AED mode—for automated ECG analysis and a prompted treatment protocol for patients in cardiac arrest.
•Manual mode—for performing manual defibrillation, synchronized cardioversion, noninvasive pacing, and ECG and vital sign monitoring.
•Archive mode—for accessing stored patient information.
•Setup mode—for changing default settings of the operating functions. For more information, see the LIFEPAK 15 Monitor/Defibrillator Setup Options provided with your device.
•Demo mode—for simulated waveforms and trend graphs for demonstration purposes.
•Service mode—for authorized personnel to perform diagnostic tests and calibrations. For more information, see the LIFEPAK 15 Monitor/Defibrillator Service Manual.
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
1-5 |

 2
2
SAFETY INFORMATION
This chapter provides important information to help you operate the LIFEPAK 15 monitor/ defibrillator. Familiarize yourself with all of these terms and warnings.
Terms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . page 2-3 General Dangers and Warnings . . . . . . . . . . . . . . . . . . . 2-3
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
2-1 |

SAFETY INFORMATION  2
2
Terms
The following terms are used either in these operating instructions or on the LIFEPAK 15 monitor/ defibrillator:
Danger: Immediate hazards that will result in serious personal injury or death.
Warning: Hazards or unsafe practices that may result in serious personal injury or death.
Caution: Hazards or unsafe practices that may result in minor personal injury, product damage, or property damage.
General Dangers and Warnings
The following are general danger and warning statements. Other specific warnings and cautions are provided as needed in other sections of these operating instructions.
DANGER!
EXPLOSION HAZARD
Do not use this defibrillator in the presence of flammable gases or anesthetics.
WARNINGS
SHOCK OR FIRE HAZARDS
SHOCK HAZARD
The defibrillator delivers up to 360 joules of electrical energy. Unless properly used as described in these operating instructions, this electrical energy may cause serious injury or death. Do not attempt to operate this device unless thoroughly familiar with these operating instructions and the function of all controls, indicators, connectors, and accessories.
SHOCK HAZARD
Do not disassemble the defibrillator. It contains no operator serviceable components and dangerous high voltages may be present. Contact authorized service personnel for repair.
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 2-3 |

General Dangers and Warnings
WARNINGS (CONTINUED)
SHOCK OR FIRE HAZARD
Do not immerse any portion of this defibrillator in water or other fluids. Avoid spilling any fluids on defibrillator or accessories. Spilled liquids may cause the defibrillator and accessories to perform inaccurately or fail. Do not clean with ketones or other flammable agents. Do not autoclave or sterilize this defibrillator or accessories unless otherwise specified.
POSSIBLE FIRE
Use care when operating this device close to oxygen sources (such as bag-valve-mask devices or ventilator tubing). Turn off gas source or move source away from patient during defibrillation.
ELECTRICAL INTERFERENCE HAZARDS
POSSIBLE ELECTRICAL INTERFERENCE WITH DEVICE PERFORMANCE
Equipment operating in close proximity may emit strong electromagnetic or radio frequency interference (RFI), which could affect the performance of this device. If use of equipment in close proximity is necessary, observe the device to verify normal operation in the configuration in which the device will be used. RFI may result in distorted ECG, incorrect ECG lead status, failure to detect a shockable rhythm, cessation of pacing, or incorrect vital sign measurements. Avoid operating the device near cauterizers, diathermy equipment, or other portable and mobile RF communications equipment. Do not rapidly key EMS radios on and off. Refer to Appendix D for recommended distances of equipment. Contact Physio-Control Technical Support if assistance is required.
POSSIBLE ELECTRICAL INTERFERENCE
Using cables, electrodes, or accessories not specified for use with this defibrillator may result in increased emissions or immunity from electromagnetic or radio frequency interference (RFI) which could affect the performance of this defibrillator or of equipment in close proximity. Use only parts and accessories specified in these operating instructions.
POSSIBLE ELECTRICAL INTERFERENCE
This defibrillator may cause electromagnetic interference (EMI) especially during charge and energy transfers. EMI may affect the performance of equipment operating in close proximity. Verify the effects of defibrillator discharge on other equipment prior to using the defibrillator in an emergency situation, if possible.
IMPROPER DEVICE PERFORMANCE HAZARDS
POSSIBLE IMPROPER DEVICE PERFORMANCE
Using other manufacturers’ cables, electrodes, power adapters, or batteries may cause the device to perform improperly and may invalidate the safety agency certifications. Use only the accessories that are specified in these operating instructions.
2-4 |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |

SAFETY INFORMATION  2
2
WARNINGS (CONTINUED)
POSSIBLE IMPROPER DEVICE PERFORMANCE
Changing factory default settings will change the behavior of the device. Changes to the default settings must only be made by authorized personnel.
POSSIBLE DEVICE SHUTDOWN
Always have immediate access to a spare, fully charged, properly maintained battery. Replace the battery when the device displays a low battery warning.
SAFETY RISK AND POSSIBLE EQUIPMENT DAMAGE
POSSIBLE INJURY OR SKIN BURNS
Monitors, defibrillators, and their accessories (including electrodes and cables) contain ferromagnetic materials. As with all ferromagnetic equipment, these products must not be used in the presence of the high magnetic field created by a Magnetic Resonance Imaging (MRI) device. The high magnetic field created by an MRI device will attract the equipment with a force sufficient to cause death or serious personal injury to persons between the equipment and the MRI device. This magnetic attraction may also damage and affect the performance of the equipment. Skin burns will also occur due to heating of electrically conductive materials such as patient leads and pulse oximeter sensors. Consult the MRI manufacturer for more information.
POSSIBLE SKIN BURNS
A defect in the neutral electrode connection on HF surgical equipment could cause burns at the lead or sensor site and damage to the monitor/defibrillator. Do not apply patient leads or sensors when using high frequency (HF) surgical (electrocautery) equipment.
Note: The features of the LIFEPAK 15 monitor/defibrillator which could come in either direct or casual contact with the patient or caregiver during normal use are not manufactured with materials that contain latex as an intentionally added component or expected impurity.
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 2-5 |

 3
3
BASIC ORIENTATION
This chapter provides a basic orientation to the LIFEPAK 15 monitor/defibrillator device and its controls, indicators, and connectors.
Front View . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . page 3-3 Back View . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-13 Home Screen. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-17 Alarms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-21 Options . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-23 Events. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-25
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 3-1 |

BASIC ORIENTATION  3
3
Front View
Figure 3-1 shows the front of the LIFEPAK 15 monitor/defibrillator. The front of the device is described in the following sections.
 CO2
CO2
SpO2 

NIBP 

ECG
LIFEPAK 15 MONITOR/DEFIBRILLATOR
MONITOR/DEFBRILLATOR
Self Test in progress...
©Physio-Control, Inc. 2008 All Rights Reserved 3207410-001
Recommended
Adult VF Dose: 200-300-360J
P1
P2
DANGER Explosion hazard. Do not use in the presence of flammable gases.
WARNING Hazardous electrical output. For use only by qualified personnel.
Figure 3-1 Front View
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 3-3 |

Front View
Area 1
Recommended
Adult VF Dose: 200-300-360J
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
ON |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CPR |
ENERGY |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
SELECT |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
ANALYZE |
CHARGE |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
LEAD |
SIZE |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
SYNC
Figure 3-2 Area 1 Controls
3-4 |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
BASIC ORIENTATION  3
3
Table 3-1 Area 1 Controls
CONTROL |
DESCRIPTION |
FOR MORE INFORMATION |
|
|
|
Physio-Control recommended energy dose for adult
See Biphasic Clinical
VF dose label Summaries at Ventricular Fibrillation (VF).
www.physio-control.com
1 |
ON |
Turns device ON or OFF. LED illuminated when ON. |
|
Press and hold to turn device off. |
|
||
|
|
|
|
|
|
|
|
2 |
ENERGY |
Increases or decreases energy level in Manual |
See page 5-22 |
SELECT |
mode. |
|
|
|
|
||
|
|
|
|
3 |
CHARGE |
Charges the defibrillator in Manual mode. |
See page 5-22 |
|
|
|
|
|
|
Shock button. Initiates discharge of defibrillator |
See page 5-22 |
|
|
energy to patient. LED flashes when charging is |
|
|
|
complete. |
|
|
|
|
|
|
|
Auxiliary power indicator. LED illuminated when |
See page 9-5 |
|
|
defibrillator is connected to auxiliary AC or DC |
|
|
|
power source, whether defibrillator is turned on or |
|
|
|
off. |
|
|
|
|
|
|
|
Battery charging indicator. LED illuminated when |
See page 9-5 |
|
|
installed batteries are fully charged. LED flashes |
|
|
|
when either battery is charging. LED is not |
|
|
|
illuminated when no batteries are installed or a |
|
|
|
battery is unable to be charged. |
|
|
|
|
|
|
|
Illuminated Service LED indicates a condition |
See page 10-21 |
|
|
exists that prevents or could prevent normal |
|
|
|
defibrillator operation. |
|
|
|
|
|
|
CPR |
Controls CPR metronome. LED illuminated when |
See page 5-25 |
|
metronome function is active. |
|
|
|
|
|
|
|
|
|
|
|
|
Activates Shock Advisory System™ (AED mode). |
See page 5-7 |
|
ANALYZE |
LED illuminated when AED is analyzing the ECG, |
|
|
and flashes when user is prompted to push |
|
|
|
|
|
|
|
|
ANALYZE. |
|
|
|
|
|
|
LEAD |
Changes ECG lead. |
See page 4-4 |
|
|
|
|
|
SIZE |
Changes ECG size. |
See page 4-5 |
|
|
|
|
|
|
Activates Synchronized mode. LED illuminated |
See page 5-27 |
|
SYNC |
when Sync mode is active and flashes with |
|
|
|
detection of each QRS. |
|
|
|
|
|
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 3-5 |
Front View
Area 2
PACER
RATE
 CURRENT
CURRENT 
PAUSE
Figure 3-3 |
Area 2 Controls |
|
|
Table 3-2 |
Area 2 Controls |
|
|
|
|
|
|
CONTROL |
DESCRIPTION |
FOR MORE INFORMATION |
|
|
|
|
|
PACER |
|
Activates pacer function. LED illuminated when function |
See page 5-32 |
|
|
is activated and flashes with each current pulse. |
|
|
|
|
|
RATE |
|
Increases or decreases pacing rate. |
See page 5-32 |
|
|
|
|
CURRENT |
Increases or decreases pacing current. |
See page 5-32 |
|
|
|
|
|
PAUSE |
|
Temporarily slows pacing rate. |
See page 5-32 |
|
|
|
|
3-6 |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |

BASIC ORIENTATION  3
3
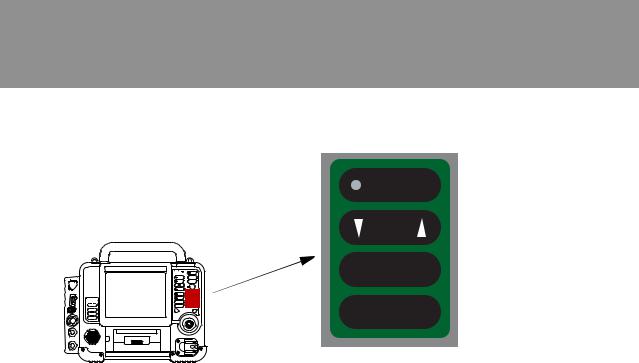
Area 3
NIBP
ALARMS
OPTIONS
EVENT
HOME
SCREEN
Speed dial
Figure 3-4 |
Area 3 Controls |
|
|
Table 3-3 |
Area 3 Controls |
|
|
|
|
|
|
CONTROL |
DESCRIPTION |
FOR MORE INFORMATION |
|
|
|
|
|
NIBP |
|
Initiates blood pressure measurement. LED |
See page 4-37 |
|
|
illuminated when BP measurement is being |
|
|
|
obtained. |
|
|
|
|
|
ALARMS |
|
Activates and silences alarms. LED illuminated |
See page 3-21 |
|
|
when alarms are enabled and flashes when an alarm |
|
|
|
condition occurs. |
|
|
|
|
|
OPTIONS |
Accesses optional functions. |
See page 3-23 |
|
|
|
|
|
EVENT |
|
Accesses user-defined events. |
See page 3-25 |
|
|
|
|
HOME SCREEN |
Returns to Home Screen display. |
See page 3-17 |
|
|
|
|
|
SPEED DIAL |
Scrolls through and selects screen or menu items. |
See page 3-19 |
|
|
|
|
|
|
|
Display mode button switches between color display |
|
|
|
and high contrast SunVue™ display. |
|
|
|
|
|
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 3-7 |

Front View
Area 4
12-LEAD
TRANSMIT
CODE
SUMMARY
Figure 3-5 |
Area 4 Controls |
|
|
Table 3-4 |
Area 4 Controls |
|
|
|
|
|
|
CONTROL |
DESCRIPTION |
FOR MORE INFORMATION |
|
|
|
|
|
12-LEAD |
Initiates acquisition of 12-lead ECG. |
See page 4-15 |
|
|
|
|
|
TRANSMIT |
Initiates transmission of patient data. |
See page 8-12 |
|
|
|
|
|
CODE SUMMARY |
Prints CODE SUMMARY™ critical event record. |
See page 7-4 |
|
|
|
|
|
|
Starts and stops printer. |
See page 7-10 |
|
|
|
|
|
3-8 |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
BASIC ORIENTATION  3
3
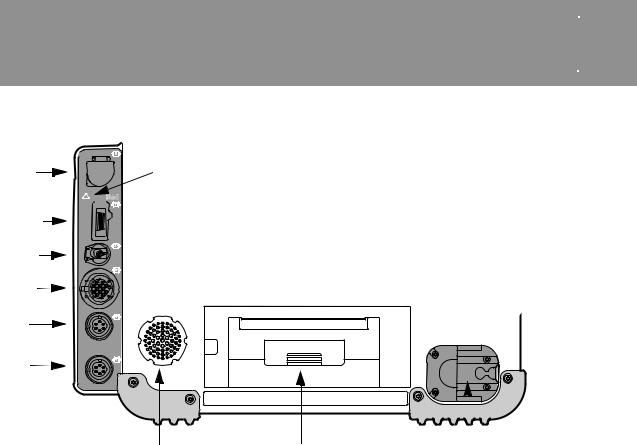
Area 5
CO2
SpO2/
SpCO/
SpMet
NIBP
ECG
P1
P2
 CO2
CO2
See warnings page 2-4
SpO2

NIBP
ECG
P1
P2
Speaker |
Printer |
Therapy cable receptacle |
Figure 3-6 |
Area 5 Connectors, Speaker, and Printer |
|
|
Table 3-5 |
Area 5 Connectors, Speaker, and Printer |
|
|
|
|
|
|
LABEL |
|
DESCRIPTION |
FOR MORE INFORMATION |
|
|
|
|
CO2 |
|
FilterLine® set port |
See page 4-45 |
SpO2/SpCO/SpMet |
Sensor cable port |
See page 4-25 |
|
|
|
|
|
NIBP |
|
Pneumatic tubing port |
See page 4-37 |
|
|
|
|
ECG |
|
Green electrically isolated ECG cable port |
See page 4-3 |
|
|
|
|
P1 |
|
Invasive pressure cable port |
See page 4-54 |
|
|
|
|
P2 |
|
Invasive pressure cable port |
See page 4-54 |
|
|
|
|
Speaker |
|
Projects device tones and voice prompts |
|
|
|
|
|
Printer |
|
Door for 100 mm printer paper |
See page 10-17 |
|
|
|
|
Therapy cable |
QUIK-COMBO® therapy cable and standard (hard) |
See page 3-12 |
|
receptacle |
paddles cable receptacle |
|
|
|
|
|
|
Note: If your LIFEPAK 15 monitor/defibrillator is configured for temperature monitoring, P1 and P2 are replaced by a single port labeled TEMP. For more information about temperature monitoring, see page 4-61.
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 3-9 |

Front View
Connectors
 CO2
CO2
SpO2

NIBP 
ECG
P1
P2
CONNECTOR |
ACTION |
|
|
|
|
|
Connect: Open CO2 port door, insert FilterLine connector, and turn |
|
CO2 |
clockwise until connector is firmly seated. |
|
Disconnect: Rotate FilterLine connector counterclockwise and pull |
||
|
||
|
connector out. |
|
|
|
|
SpO2/ |
Connect: Align cable connector with SpO2 port and push in until |
|
connector clicks into place. |
||
SpCO/ |
||
Disconnect: Press the gray buttons on each side of the cable |
||
SpMet |
||
connector simultaneously and pull connector out. |
||
|
||
|
|
|
|
Connect: Insert NIBP tubing connector into the NIBP port. |
|
NIBP |
Disconnect: Press the latch on the left side of the port and pull |
|
|
tubing connector out. |
|
|
|
|
|
Connect: Align the green ECG connector with the ECG port; |
|
ECG |
position the white line on the cable facing left. Insert the cable |
|
connector into the port until the connector is firmly seated. |
||
|
||
|
Disconnect: Pull the ECG connector straight out. |
|
|
|
|
|
Connect: Align the IP (invasive pressure) cable connector with the |
|
|
P1 or P2 port; position the gap on the connector facing up. Insert |
|
P1/P2 |
the cable connector into the port until the connector is firmly |
|
|
seated. |
|
|
Disconnect: Grip the connector and pull straight out. |
|
|
|
Figure 3-7 Connectors for IP Monitoring Configuration
Note: If your LIFEPAK 15 monitor/defibrillator is configured for temperature monitoring, P1 and P2 are replaced by a single port labeled TEMP. For more information, see Figure 3-8 on page 3-11.
3-10 |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
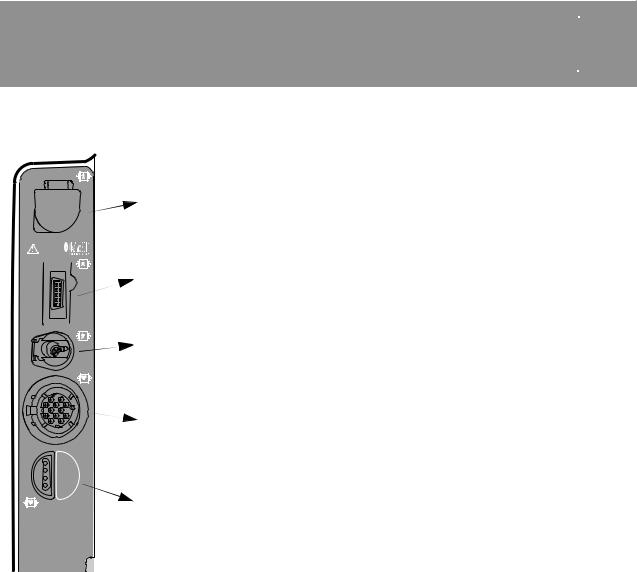
BASIC ORIENTATION  3
3
 CO2
CO2
SpO2

NIBP 

ECG
TEMP
CONNECTOR |
ACTION |
|
|
|
|
|
Connect: Open CO2 port door, insert FilterLine connector, and |
|
CO2 |
turn clockwise until connector is firmly seated. |
|
Disconnect: Rotate FilterLine connector counterclockwise and |
||
|
||
|
pull connector out. |
|
|
|
|
SpO2/ |
Connect: Align cable connector with SpO2 port and push in |
|
until connector clicks into place. |
||
SpCO/ |
||
Disconnect: Press the gray buttons on each side of the cable |
||
SpMet |
||
connector simultaneously and pull connector out. |
||
|
||
|
|
|
|
Connect: Insert NIBP tubing connector into the NIBP port. |
|
NIBP |
Disconnect: Press the latch on the left side of the port and pull |
|
|
tubing connector out. |
|
|
|
|
|
Connect: Align the green ECG connector with the ECG port; |
|
|
position the white line on the cable facing left. Insert the |
|
ECG |
cable connector into the port until the connector is firmly |
|
|
seated. |
|
|
Disconnect: Pull the ECG connector straight out. |
|
|
|
|
|
Connect: Align the temperature adapter cable connector with |
|
TEMP |
the TEMP port. Insert the cable connector into the port until |
|
the connector is firmly seated. |
||
|
||
|
Disconnect: Grip the connector and pull straight out. |
|
|
|
Figure 3-8 Connectors for Temperature Monitoring Configuration
©2007-2013 Physio-Control, Inc. |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions 3-11 |

Front View
Connecting and Disconnecting the Therapy Cable
WARNING
POSSIBLE EQUIPMENT DAMAGE AND INABILITY TO DELIVER THERAPY
To help protect the therapy cable connector from damage or contamination, keep therapy cable connected to the defibrillator at all times. Inspect and test the therapy cable daily according to the Operator’s Checklist in the back of this manual. PhysioControl recommends replacement of therapy cables every three years to reduce the possibility of failure during patient use.
IMPORTANT! The LIFEPAK 15 monitor/defibrillator QUIK-COMBO therapy cable and standard (hard) paddles have the same type of connector and connect to the defibrillator at the same location. These therapy cables are not compatible with other LIFEPAK defibrillator/monitors.
To connect a therapy cable to the defibrillator:
1. Align the therapy cable connector with the receptacle.
2. Slide the therapy cable until you feel the connector lock in place. You will also hear a “click.”
Figure 3-9 Connect Therapy Cable
To disconnect the therapy cable from the defibrillator:
1. Press the release button on the therapy cable connector.
2. Slide the therapy cable connector out.
Figure 3-10 Disconnect Therapy Cable
3-12 |
LIFEPAK 15 Monitor/Defibrillator Operating Instructions |
 Loading...
Loading...