VIASYS Nicolet Elite User manual

Nicolet Elite®
User's Manual
2

Congratulations on your choice of the
Nicolet Elite®
from VIASYS Healthcare
The Elite provides the features which have made VIASYS Healthcare a world leader in Doppler technology: reliability, quality, sensitivity and value.
CAUTION: Federal law (USA) restricts this device to use by or on the order of a physician.
VIASYS Healthcare strives for continuous improvement. Please contact us if you have comments or suggestions for improvements regarding our products or services.
0344
The
 0344 Mark identifies compliance with the Medical Device Directive 93/42/EEC.
0344 Mark identifies compliance with the Medical Device Directive 93/42/EEC.
VIASYS Healthcare Inc.
P.O. Box 44994
Madison, WI 53744
Phone: 608-441-2266
Toll Free: (800) 525-2519
Fax: 608-441-2232
Email: Vascular.Info@viasyshc.com
www.viasyshealthcare.com
European Authorized Representative:
VIASYS Healthcare
Welton Road
Warwick, CV345PZ U.K.
© 2005 VIASYS Healthcare
M7-0775 Rev. I
3
Blank page.
4
TABLE OF CONTENTS
Section |
Page Number |
Limitations |
5 |
Product Description |
6 |
Safety of Ultrasound |
8 |
Contraindications |
8 |
Operation |
8 |
Care of the Instrument |
8 |
General Hints |
12 |
Care of the Instrument |
13 |
Product Compatibility |
14 |
Specifications |
14 |
Electromagnetic Compatibility (EMC) |
17 |
Troubleshooting Nicolet Elite Problems |
21 |
Service |
23 |
Accessories |
23 |
Limited Warranty |
24 |
The purpose of this manual is to guide the user in the operation and care of the Elite. Every reasonable effort has been made to present accurate information. VIASYS Healthcare hereby disclaims and makes no warranty with respect to any diagnosis, clinical condition or interpretation made based on the information presented herein. Refer to the Limited Warranty at the end of this manual for the equipment warranty.
 Caution: The 5 MHz and 8MHz vascular probes are not intended for fetal use.
Caution: The 5 MHz and 8MHz vascular probes are not intended for fetal use.
 Warning: This product is not intended for use on or around the eyes. This product is not intended for use on non-intact skin.
Warning: This product is not intended for use on or around the eyes. This product is not intended for use on non-intact skin.
LIMITATIONS
The theory of Doppler ultrasound is beyond the scope of this manual, but is covered in many recent publications. The Elite is designed to be reliable, but as with all medical instruments, should be treated with care. While the Elite augments the user’s skills, it is not a substitute for medical training and knowledge.
NOTE: TheEliteprovidesthelistenerwithinformationaboutthefetusandvascularconditions. Under poor signal conditions, the fetal heart sounds and readings may not be accurate. (Refer to the Troubleshooting section at the end of this manual for a description of sensitivity problems and solutions.) If there is any doubt about the fetal condition after using the Doppler, further investigation should be initiated immediately.
5
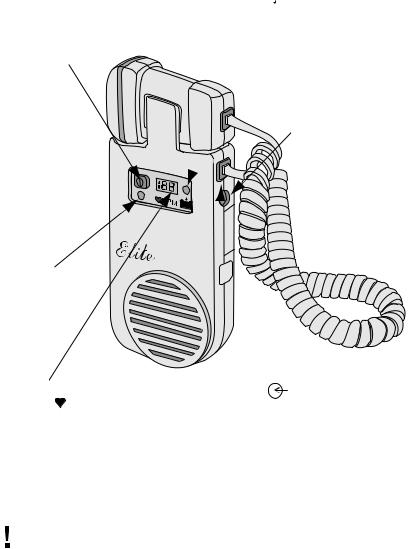
PRODUCT DESCRIPTION
The Elite is a non-invasive hand-held Doppler with a speaker. The Elite has the following special features that will enhance your product use:
On/Off: 
PressingtheOn/Off buttononthefrontpanel of the unit turns on the Elite. The Elite is turnedoffbyagainpressingtheOn/Offbutton.
On Indicator:
(Elite 100 & 100R)
Green LED “On” indicator for units without display.
HeartRate/LowBatteryIndicator (Elite
200 & 200R):
Thedigitaldisplayshowsheartrate. Thedisplay flashes when batteries are low.
Recharge Indicator:  (Elite 100R & 200R)
(Elite 100R & 200R)
The green light turns on to indicate that the unit is recharging .
Headphone Connector: 
Touseoptionalheadphones,plugconnector into the hole on the edge of the unit. Main speaker will not be heard when headphones are plugged in.
Probe Connector:
Disconnect the probe by holding the probe in one hand, grasping the connector at the bottom of the probe in the other hand, firmly depressing the clear tab on the connector, and pulling the connector from the probe.
Warning
 THE EliteIS NOT EXPLOSION PROOF. DO NOT USE THE EliteOR PROBES IN THE PRESENCE OF FLAMMABLE OR EXPLOSIVE GASES. DO NOT IMMERSE THE UNIT OR PROBES IN FLUIDS AND DO NOT AUTOCLAVE.
THE EliteIS NOT EXPLOSION PROOF. DO NOT USE THE EliteOR PROBES IN THE PRESENCE OF FLAMMABLE OR EXPLOSIVE GASES. DO NOT IMMERSE THE UNIT OR PROBES IN FLUIDS AND DO NOT AUTOCLAVE.
6

Probe Face:
Place 2 MHz, 3 MHz or 5 MHz probe flat on skin. There is no need to tip at various angles to detect fetal heartbeat or blood flow in artery or vein.
Probe Face:
8MHz probe requires an approximate 45° angle for optimal results.
Louder
Volume control:
Volume is adjusted by sliding the “Volume” control button on the side of the unit. Slide the button up to make the sound louder. Slide the button down to make the sound quieter.
CAUTION: |
Federal law |
|
|
|
|||||||||
device to |
|
|
|
restricts |
|
||||||||
|
|
|
|
use by |
or on the |
|
this |
||||||
physician. |
|
|
order of a |
||||||||||
CAUTION: |
|
|
|
|
|||||||||
No part |
|
|
|
|
|||||||||
may |
|
|
|
|
|
|
|
|
|
||||
|
|
be connected to a |
of this equipment |
||||||||||
NICOLET |
VASCULAR |
telephone |
system. |
||||||||||
a |
|
|
|
|
|
|
|
||||||
|
division of |
VIASYS |
Healthcare |
|
|
|
|
||||||
5225 Verona Rd., |
|
|
|
|
|
|
|||||||
Madison, WI |
|
Bldg. 2 |
|
|
|
|
|
||||||
1- |
800-525- |
|
53711-4495 |
|
|
|
|
|
|||||
|
|
|
2519 USA |
|
|
|
|
|
|
||||
Recharger 

 (Elite 100R & 200R):
(Elite 100R & 200R):
Transformer/recharger unit plugs into main unitforrechargingrechargeablebatteries. The unit will not operate while the recharger is plugged in.
Battery Replacement:
To replace a battery, remove the battery compartment door and remove the old battery. Carefully install the new battery with the connector end as shown on the label. Replace the battery compartment door.
Warning
USE ONLY RECHARGER AND RECHARGEABLE BATTERIES AS SPECIFIED BY VIASYS Healthcare. USE OF OTHER BATTERIES MAY CREATE A HAZARDOUS SITUATION AND VOID THE MANUFACTURERS WARRANTY.
THE DOPPLER PROBE SHOULD ONLY BE APPLIED TO A PATIENT DURING THE BATTERY OPERATION. DO NOT APPLY THE DOPPLER PROBE TO A PATIENT WHEN THE UNIT IS PLUGGED INTO THE RECHARGER (ELITE 100R/200R MODELS). THE UNIT DOES NOT OPERATE WHILE CHARGING.
7
SAFETY OF ULTRASOUND
The American Institute of Ultrasound in Medicine (AIUM) has addressed the concerns relating to the safety of ultrasound and has issued the following statement as of March 1993:
Diagnostic ultrasound has been in use since the late 1950s. Given its known benefits and recognized efficacy for medical diagnosis, including use during human pregnancy, the AIUM herein addresses the clinical safety of such use:
"No confirmed biological effects on patients or instrument operators caused by exposure at intensities typical of present diagnostic ultrasound instruments have ever been reported. Although the possibility exists that such biological effects may be identified in the future, current data indicate that the benefits to patients of the prudent use of diagnostic ultrasound outweigh the risks, if any, that may be present.”
VIASYS Healthcare's policy is to always use as low an ultrasound power as practical. The level of ultrasound power emitted by the Elite is not adjustable. Prudent use on the operator’s part would include minimizing the length of time that the patient is undergoing the ultrasound exposure.
CONTRAINDICATIONS
The Elite is not for invasive use, not for use near or on non-intact skin and not for use near the eyes. The Elite 5 MHz and 8 MHz probes are not intended for fetal use.
OPERATION
Removetheprobefromthemainunitbygraspingtheprobeandpullingitstraightupandawayfrom theunit. TurnontheunitbypressingtheOn/Offbutton.Allsectionsoftheliquidcrystaldisplay(Elite 200 and 200R) will illuminate to ensure they are working properly.
1.Make sure the probe cable is connected at both the probe and the main unit.
2.Apply gel to the face of the probe and begin the Doppler examination.
3.Adjust the volume on the main unit as needed, by sliding the slider up to increase the volume or down to decrease the volume.
4.When finished with the exam, turn the unit off by pressing the On/Off button.
5.Refer to cleaning instructions to clean the probe.
8
 Loading...
Loading...