Cosmed Quark PFT User Manual

Cosmed does not assume the liability for interpretation mistakes of this documentation or for casual or consequential damages in connection with the provision, representation or use of this documentation.
No parts of this manual may be reproduced or transmitted in any form without the express permission of COSMED Srl.
COSMED Software can be installed only in one device.
Excel is a registered trademark of Microsoft Corporation. DBIII is a registered trademark of Bordland International Inc.
Lotus 123 is a registered trademark of Lotus Development Corporation .
Quark PFT User manual, VII Edition
September 2003
Copyright © 1998 COSMED
Copyright © 2003 COSMED
COSMED Srl - Italy
http://www.cosmed.it
Part N. C01340-02-91

Table of contents
Getting started |
11 |
Important notices............................................................................ |
12 |
Intended use ............................................................................. |
12 |
Warnings .................................................................................. |
12 |
Contraindication ............................................................................. |
14 |
Contraindications for the Spirometer tests ............................... |
14 |
Absolute contraindications............................................. |
14 |
Relative contraindications.............................................. |
14 |
Contraindications for Bronchial provocation tests................... |
14 |
Absolute contraindications............................................. |
14 |
Relative contraindications.............................................. |
14 |
Contraindications for Exercise testing ..................................... |
14 |
Environmental condition of use ...................................................... |
15 |
Safety and conformity..................................................................... |
16 |
Safety ............................................................................. |
16 |
EMC............................................................................... |
16 |
Quality Assurance.......................................................... |
16 |
Medical Device Directive (CE mark) ............................ |
16 |
Keynotes ......................................................................................... |
17 |
Typographic keynotes .............................................................. |
17 |
Graphic keynotes...................................................................... |
17 |
Systems Overview ........................................................................... |
18 |
Tests/Products table ................................................................. |
18 |
Before starting................................................................................ |
19 |
Checking the packing contents................................................. |
19 |
Quark PFT1 standard packaging.................................... |
19 |
Quark PFT2 standard packaging.................................... |
19 |
Quark PFT3 standard packaging.................................... |
20 |
Quark PFT4 standard packaging.................................... |
20 |
Quark PFT Ergo standard packaging............................. |
21 |
Quark PFT2 ergo Standard packaging........................... |
21 |
Quark PFT4 ergo Standard packaging........................... |
22 |
Warranty registration ..................................................................... |
23 |
Register the product via software .................................. |
23 |
How to contact COSMED ............................................. |
23 |
Complain, feedback and suggestions............................. |
23 |
Options/Accessories........................................................................ |
24 |
PC configuration required .............................................................. |
25 |
Technical features........................................................................... |
26 |
Flowmeter ...................................................................... |
26 |
Oxygen Sensor (O2) ....................................................... |
26 |
Carbon Dioxide Sensor (CO2)........................................ |
26 |
2 - Quark PFT User Manual |
|

Carbon Monoxide sensor (CO)...................................... |
26 |
Methane sensor (CH4).................................................... |
26 |
Humidity absorber ......................................................... |
26 |
Power Supply................................................................. |
26 |
Environmental Sensors .................................................. |
26 |
Dimension and Weight .................................................. |
26 |
Aux inputs...................................................................... |
26 |
Measurements |
27 |
Pulmonary function tests and measured parameters .................... |
28 |
Spirometry Tests ...................................................................... |
28 |
FVC - Forced Vital Capacity......................................... |
28 |
VC/IVC - Slow Vital Capacity and Ventilatory pattern 28 |
|
MVV - Maximum Voluntary Ventilation...................... |
29 |
Bronchoprovocation Response ...................................... |
29 |
Multi-breath Nitrogen wash-out .................................... |
29 |
Closing Volume test ...................................................... |
29 |
CO diffusion Test .......................................................... |
29 |
Respiratory Drive - P0.1 Test ........................................ |
30 |
MIP/MEP....................................................................... |
30 |
Cardio pulmonary Exercise Testing (CPET) ........................... |
30 |
Breath by Breath exercise testing .................................. |
30 |
Indirect Calorimetry....................................................... |
31 |
Lactate Threshold (V-Slope) ......................................... |
31 |
O2 Kinetics .................................................................... |
31 |
Installation |
33 |
Unpacking the system..................................................................... |
34 |
Setting up the system ...................................................................... |
35 |
Installing the Calibration Gas Cylinders.................................. |
35 |
Gas pressure adjustment procedure.......................................... |
35 |
Connecting cables and tubing......................................................... |
36 |
Cables and Tubing’s installation sequence .............................. |
37 |
Assemble the breathing valve .................................................. |
37 |
Assemble the turbine ...................................................................... |
38 |
Assemble the mask and the flowmeter .......................... |
38 |
Using the "Ultimate Seal" ........................................................ |
38 |
Apply the seal to the mask............................................. |
39 |
To remove seal on mask ................................................ |
39 |
Connecting PC ......................................................................... |
39 |
Software installation ....................................................................... |
40 |
Installing the software.............................................................. |
40 |
Run the software ...................................................................... |
40 |
PC port configuration............................................................... |
40 |
Software main features................................................................... |
41 |
Display ..................................................................................... |
41 |
Index - 3

Tool bar .................................................................................... |
41 |
Show/hide the toolbar .................................................... |
41 |
Dialog windows ....................................................................... |
41 |
Use of the keyboard ....................................................... |
41 |
Use of the mouse............................................................ |
41 |
Scroll bars ................................................................................ |
41 |
On-line help.............................................................................. |
41 |
Software version....................................................................... |
41 |
Calibration |
43 |
Calibrating Sensors......................................................................... |
44 |
Running the Calibration program............................................. |
44 |
Log file..................................................................................... |
44 |
Setting reference values ........................................................... |
44 |
To set the reference values............................................. |
44 |
Calibrating analysers................................................................ |
45 |
ERGO Calibration.......................................................... |
45 |
FRC Calibration............................................................. |
45 |
DLCO Calibration.......................................................... |
45 |
Print the calibration report ....................................................... |
46 |
Edit the calibration factors ....................................................... |
46 |
O2 delay check.......................................................................... |
46 |
Turbine calibration ......................................................................... |
47 |
The calibration syringe .................................................. |
47 |
Assembling the flowmeter for calibration .................... |
47 |
Calibrating the turbine.............................................................. |
48 |
Checking the system signals........................................................... |
49 |
The control panel...................................................................... |
49 |
Using the control panel .................................................. |
49 |
Database Management |
51 |
Spirometry patient's database........................................................ |
52 |
Patient Card.................................................................... |
52 |
Visit Card....................................................................... |
52 |
Test Card........................................................................ |
53 |
Import/export a Tests card ....................................................... |
53 |
Diagnosis Database .................................................................. |
53 |
Exercise testing patient's database ................................................ |
54 |
Enter a new patient................................................................... |
54 |
Find a patient............................................................................ |
54 |
Edit patient data........................................................................ |
54 |
Delete a patient......................................................................... |
54 |
Archive maintenance ...................................................................... |
55 |
Reorganise the archive ............................................................. |
55 |
Delete the archive..................................................................... |
55 |
Delete a test.............................................................................. |
55 |
4 - Quark PFT User Manual |
|

Backup and restore................................................................... |
55 |
Backup ........................................................................... |
55 |
Restore........................................................................... |
55 |
Spirometry Settings ......................................................................... |
56 |
Graphs............................................................................ |
56 |
Serial port ...................................................................... |
56 |
Units of measurements .................................................. |
56 |
Using extra fields........................................................... |
56 |
Customise the fields....................................................... |
56 |
Spirometry |
57 |
Setting spirometry options .............................................................. |
58 |
Spirometry................................................................................ |
58 |
Automatic Interpretation................................................ |
58 |
Quality control............................................................... |
58 |
Parameters manager ................................................................. |
59 |
Predicted values manager......................................................... |
59 |
Predicteds set ................................................................. |
59 |
Set the current predicted ................................................ |
60 |
Formula definition ......................................................... |
60 |
Page set-up ............................................................................... |
61 |
Spirometry tests............................................................................... |
62 |
Forced Vital Capacity (pre).............................................................. |
63 |
Recommendations.................................................................... |
63 |
Perform a FVC (pre) test.......................................................... |
63 |
Test encouragement ................................................................. |
63 |
Perform the FVC test with the encouragement.............. |
64 |
Slow Vital Capacity.......................................................................... |
65 |
Perform a SVC test .................................................................. |
65 |
Maximum Voluntary Ventilation ..................................................... |
66 |
Perform a MVV test................................................................. |
66 |
Bronchial Provocation Test.............................................................. |
67 |
Bronchodilator test................................................................... |
67 |
Methacholine and Histamine Bronchial provocation Tests ..... |
67 |
Perform the test........................................................................ |
68 |
Bronchial Provocation protocols Database .............................. |
68 |
Enter a new Bronchial provocation protocol in the |
|
archive ........................................................................... |
68 |
Viewing results................................................................................ |
69 |
Tests of the current patient ............................................ |
69 |
Delete a test ................................................................... |
69 |
Printing results ................................................................................ |
70 |
Printing Reports ....................................................................... |
70 |
Printing the active window ...................................................... |
71 |
To print the active window............................................ |
71 |
Printing a series of reports ....................................................... |
71 |
Electronic reports (*.pdf) ......................................................... |
71 |
Index - 5

Export data ............................................................................... |
71 |
Export a test ................................................................... |
71 |
Lung Volumes |
73 |
Lung volumes tests (N2 washout) ................................................... |
74 |
Recommendations.......................................................... |
74 |
Setting N2 wash-out options .................................................... |
74 |
Perform the test ........................................................................ |
75 |
Closing Volume Test (CV) ................................................................ |
76 |
Recommendations.......................................................... |
76 |
Perform the test ........................................................................ |
76 |
P0.1 Respiratory drive |
79 |
P0,1 (Respiratory drive).................................................................. |
80 |
Aim of the test.......................................................................... |
80 |
Setting respiratory drive options .............................................. |
80 |
Recommendations.......................................................... |
80 |
Perform the test ........................................................................ |
80 |
Lung diffusing capacity |
83 |
CO Diffusing Capacity (DLCO) Test.................................................. |
84 |
Setting DLCO options.............................................................. |
84 |
Recommendations.......................................................... |
85 |
DLCO with breath-hold (standard technique).......................... |
85 |
DLCO with apnoea (standard method) .................................... |
85 |
DLCO without apnoea (“intrabreath” method)........................ |
87 |
Multiple breath DLCO test (“Steady-State” method) .............. |
87 |
MIP/MEP (option) |
89 |
MIP/MEP test................................................................................... |
90 |
How to perform the test............................................................ |
90 |
Pulse oximetry (option) |
91 |
The oximetry test............................................................................. |
92 |
How to perform the test............................................................ |
92 |
Exercise testing |
93 |
Recommendations for the exercise testing..................................... |
94 |
The evaluation of the cardiorespiratory function ..................... |
94 |
Precautions ............................................................................... |
94 |
Laboratory...................................................................... |
94 |
Ending the test ............................................................... |
94 |
Preparing the patient ................................................................ |
94 |
Before testing................................................................. |
94 |
Patient assent.................................................................. |
95 |
Ending the test ............................................................... |
95 |
6 - Quark PFT User Manual |
|

Start Testing .................................................................................... |
96 |
Start a test ...................................................................... |
96 |
Abort the test without saving data ................................. |
96 |
End the test saving data ................................................. |
96 |
View data in real-time.............................................................. |
96 |
View graphs in real-time ............................................... |
97 |
Parameters to view................................................................... |
97 |
Manual protocol ....................................................................... |
97 |
Enter Load and Phase .................................................... |
97 |
Set the markers .............................................................. |
97 |
Automatic protocol .................................................................. |
97 |
Modify the load during the test...................................... |
97 |
Set the BPM alarm................................................................... |
97 |
Enter the BPM ............................................................... |
97 |
Data management .......................................................................... |
98 |
Viewing data ............................................................................ |
98 |
View data in table form ................................................. |
98 |
Creating graphs ........................................................................ |
98 |
View data in graph form................................................ |
98 |
Customise the graphs..................................................... |
99 |
Switch from graph to data and vice versa...................... |
99 |
Viewing predicted values....................................................... |
100 |
View predicted values.................................................. |
100 |
Anaerobic (Lactate) Threshold detection............................... |
100 |
View the Lactate Threshold......................................... |
100 |
Detect the Lactate Threshold ....................................... |
100 |
Customise graphs for the LT viewing.......................... |
100 |
Fittings ................................................................................... |
101 |
Fit a graph with a linear regression.............................. |
101 |
Fit a graph with a Mono-exponential regression ......... |
101 |
Calculate the "Mean Value" ........................................ |
102 |
Oxygen Kinetic ...................................................................... |
102 |
Run the O2 Kinetic function........................................ |
102 |
Information about the Test..................................................... |
103 |
View the Information................................................... |
103 |
Modify the information................................................ |
103 |
Summary ................................................................................ |
103 |
View the summary....................................................... |
103 |
Print the data .......................................................................... |
103 |
Print the current window ............................................. |
104 |
Print the report ............................................................. |
104 |
View the report ...................................................................... |
104 |
Data Editing .................................................................................. |
105 |
Editing values and input numerical values .................. |
105 |
Data filtering................................................................ |
105 |
Using the User fields ................................................... |
106 |
Index - 7

Deleting steps............................................................... |
106 |
Advanced Editing ........................................................ |
106 |
Restore the original test ............................................... |
107 |
Overwrite the original test ........................................... |
107 |
Customise the desktop............................................................ |
107 |
Customise the display colours ..................................... |
107 |
Smart edit ............................................................................... |
107 |
Apply the graphical noise suppression ........................ |
107 |
Apply the threshold noise suppression ........................ |
107 |
Customise the parameters ...................................................... |
108 |
Create a new parameter................................................ |
108 |
Create a new predicted parameter................................. |
108 |
Exporting data ........................................................................ |
109 |
Export a test ................................................................. |
109 |
DDE with Excel........................................................... |
109 |
Creating Test Protocols ................................................................. |
110 |
Create a new protocol .................................................. |
110 |
Software configuration ................................................................. |
111 |
Data viewing .......................................................................... |
111 |
Select the parameters to view ...................................... |
111 |
Select the parameters to view during the test............... |
111 |
Sort the parameters ...................................................... |
111 |
Steady State............................................................................ |
111 |
Customise the Steady State detection criteria.............. |
111 |
Printout reports............................................................................. |
112 |
Set up the printout........................................................ |
112 |
Select parameters to be printed.................................... |
112 |
Customise the printout header ..................................... |
112 |
Electronic reports (*.pdf)............................................. |
113 |
Print the current window........................................................ |
113 |
Print the customised report..................................................... |
113 |
Events management during exercise testing ............................... |
114 |
Flow Volume loops................................................................ |
114 |
Flow Volume loop during the test ............................... |
114 |
O2, CO2 vs Time ................................................................... |
114 |
O2, CO2 vs Time during the test ................................. |
114 |
O2 Saturation (optional)......................................................... |
114 |
O2 Saturation during the test ....................................... |
115 |
Spirogram............................................................................... |
115 |
Spirogram during the test............................................. |
115 |
View the events after the test ................................................. |
115 |
Raw data................................................................................. |
115 |
Save Raw data.............................................................. |
115 |
Resting Metabolic Rate Test |
117 |
Metabolism ................................................................................... |
118 |
8 - Quark PFT User Manual |
|

Total Metabolic Rate.............................................................. |
118 |
Resting Metabolic Rate (RMR) ............................................. |
118 |
Importance to measure RMR ................................................. |
118 |
Measurement of the rest metabolic rate with indirect |
|
calorimetry ............................................................................. |
118 |
How to perform a RMR test................................................... |
118 |
Recommendations......................................................................... |
119 |
Performing a test .......................................................................... |
120 |
Calibrations............................................................................ |
120 |
How to prepare a patient ........................................................ |
120 |
Start the test............................................................................ |
120 |
Viewing the test ..................................................................... |
121 |
How to modify the average interval....................................... |
122 |
Print........................................................................................ |
122 |
Sub-maximal Exercise Testing |
125 |
Introduction................................................................................... |
126 |
Pre-test screening ................................................................... |
126 |
Sub-maximal exercise testing ....................................................... |
127 |
Considerations with sub-maximal exercise testing................ |
127 |
Staffing................................................................................... |
128 |
Test termination ..................................................................... |
128 |
Considerations for accuracy .......................................................... |
129 |
Performing the test ....................................................................... |
130 |
An example of testing protocol.............................................. |
130 |
System maintenance |
131 |
System maintenance ..................................................................... |
132 |
Cleaning and disinfection....................................................... |
132 |
Cleaning the turbine flowmeter ................................... |
133 |
Precautions during the cleaning of the turbine ............ |
133 |
Suggested disinfection solutions.................................. |
133 |
Cleaning the Breathing valve................................................. |
133 |
Masks cleaning and disinfection............................................ |
135 |
Disassembling the different parts of the mask............. |
135 |
Cleaning the mask........................................................ |
135 |
Disinfecting the mask .................................................. |
135 |
Permapure maintenance ......................................................... |
135 |
Inspections ............................................................................. |
135 |
Replace the fuses.................................................................... |
135 |
Select the proper power supply voltage ................................. |
136 |
Appendix |
137 |
Service - Warranty ........................................................................ |
138 |
Warranty and limitation of liability ....................................... |
138 |
Return goods policy for warranty or non warranty repair...... |
138 |
Repair Service Policy............................................................. |
139 |
Index - 9

Privacy Information ...................................................................... |
140 |
Personal data treatment and purposes .................................... |
140 |
How your personal data are treated........................................ |
140 |
The consent is optional, but… ............................................... |
140 |
Holder of the treatment .......................................................... |
140 |
Customer rights ...................................................................... |
140 |
Heart Rate – TTL input .................................................................. |
141 |
Converting factors configuration .................................................. |
142 |
Calculations references................................................................. |
143 |
VO2 and VCO2 ....................................................................... |
143 |
Anaerobic threshold (modified V-Slope)............................... |
143 |
References.................................................................... |
143 |
O2 kinetics .............................................................................. |
143 |
ATS 94 recommendations ............................................................. |
144 |
ATS recommendations........................................................... |
144 |
Predicted values ........................................................................... |
145 |
ERS93 .................................................................................... |
145 |
Reference Adult:.......................................................... |
145 |
Reference Paediatric: ................................................... |
145 |
KNUDSON 83 ....................................................................... |
145 |
Reference Adult/ Paediatric:........................................ |
145 |
ITS (White race)..................................................................... |
145 |
Reference Adult/ Paediatric:........................................ |
145 |
ITS (Black race) ..................................................................... |
145 |
Reference Adult/ Paediatric:........................................ |
145 |
LAM....................................................................................... |
145 |
Reference Adult/ Paediatric:........................................ |
145 |
Multicéntrico de Barcelona .................................................... |
145 |
Reference Adult/ Paediatric:........................................ |
145 |
NHANES III........................................................................... |
145 |
Reference Adult/ Paediatric:........................................ |
145 |
Automatic diagnosis (algorithm)............................................ |
146 |
Quality Control Messages ...................................................... |
146 |
References..................................................................................... |
147 |
Spirometry ................................................................... |
147 |
Lung Volumes.............................................................. |
147 |
Single-Breath with Apnea............................................ |
147 |
Single-Breath without Apnea....................................... |
147 |
Gas Exchange References............................................ |
147 |
Indirect calorimetry...................................................... |
147 |
Sub-maximal testing .................................................... |
147 |
10 - Quark PFT User Manual

Getting started

Important notices
Intended use
Quark PFT is an electrical medical device designed to perform pulmonary function tests. It is to be used by physicians or by trained personnel on a physician responsibility.
Caution: Federal law restricts this device to sale by or on the order of a physician.
This equipment has been conceived with the aim of providing an auxiliary instrument allowing:
•the formulation of lung pathology diagnosis;
•important studies concerning human physiology;
•the collection of important information in sport medicine.
No responsibility attaches COSMED Srl for any accident happened after a wrong use of the device, such as:
•use by non qualified people;
•non respect of the device intended use;
•non respect of the hereunder reported precautions and instructions.
Warnings
The device, the program algorithms and the presentation of measured data have been developed according to the specifications of ATS (American Thoracic Society) and ERS (European Respiratory Society). Other international references have been followed when these were not available. All bibliography references are reported in Appendix.
The present handbook has been developed with respect of the European Medical Device Directive requirements which sort Quark PFT within Class II a.
It is recommended to read carefully the following precautions before putting the device into operation.
The precautions reported below are of fundamental importance to assure the safety of all COSMED equipment users.
1.This user manual is to be considered as a part of the medical device and should always be kept on hand.
2.Safety, measure accuracy and precision can be assured only:
•using the accessories described in the manual or given with the device. Actually non recommended accessories can affect safety unfavourable. Before using non recommended accessories it is necessary to get in touch with the manufacturer;
•ordinary equipment maintenance, inspections, disinfection and cleaning are performed in the way and with the frequency described;
•any modification or fixing is carried out by qualified personnel;
•the environmental conditions and the electrical plants where the device operates are in compliance with the specifications of the manual and the present regulations concerning electrical plants. In particular grounding reliability and leakage current suppression can only be assured when the device three – wire receptacle is connected to a yellow - green return connected to earth ground. Attempting to defeat the proper connection of the ground wire is dangerous for users and equipment.
3.Before powering the system, check the power cables and the plugs. Damaged electrical parts must be replaced immediately by authorised personnel.
4.Large gas cylinders, which may be given by the manufacturer or purchased by the customer, should be secured with cylinder safety chains or safety stands.
5.When removing the protective cap, inspect the cylinder valve for damaged threads, dirt, oil or grease. Remove any dust or dirt with a clean cloth. If oil or grease is present on the valve of a cylinder which contains oxygen, do not attempt to use. Such combustible substances in contact with oxygen are explosive.
6.Be certain that the materials of the pressure regulators are chemically compatible with the intended gas service before installation. Inspect the regulator for the proper
12 - Quark PFT User Manual
connection and note the ranges of the pressure gauges. Also examine the physical condition of the regulator including threads and fittings. Remove any dust or dirt from the regulator or cylinder valve with a clean cloth. Do not install a regulator on a cylinder valve containing oxygen if grease or oil is present on either. Such substances in contact with oxygen are explosive.
7.Cleaning residue, particulates, and other contaminates (including pieces of torn or broken components) in the breathing circuit pose a safety risk to the patient during testing procedures. Aspiration of contaminates can potentially be life-threatening. You must follow all the cleaning procedures in System Maintenance, and you must thoroughly inspect the components after cleaning and before each patient test.
8.This device is not suitable for use in presence of flammable anaesthetics. It is not an AP nor an APG device (according to the EN 60 601-1 definitions).
9.Keep the device away from heat and flame source, flammable or inflammable liquids or gases and explosive atmospheres.
10.In accordance with their intended use Quark PFT is not to be handled together with other medical devices unless it is clearly declared by the manufacturer itself.
11.It is recommended to use a computer with electromagnetic compatibility CE marking and with low radiation emission displays.
12.It is necessary to make the PC, connected to the Quark PFT, compliant with EN 60601-1 by means of an isolation transformer.
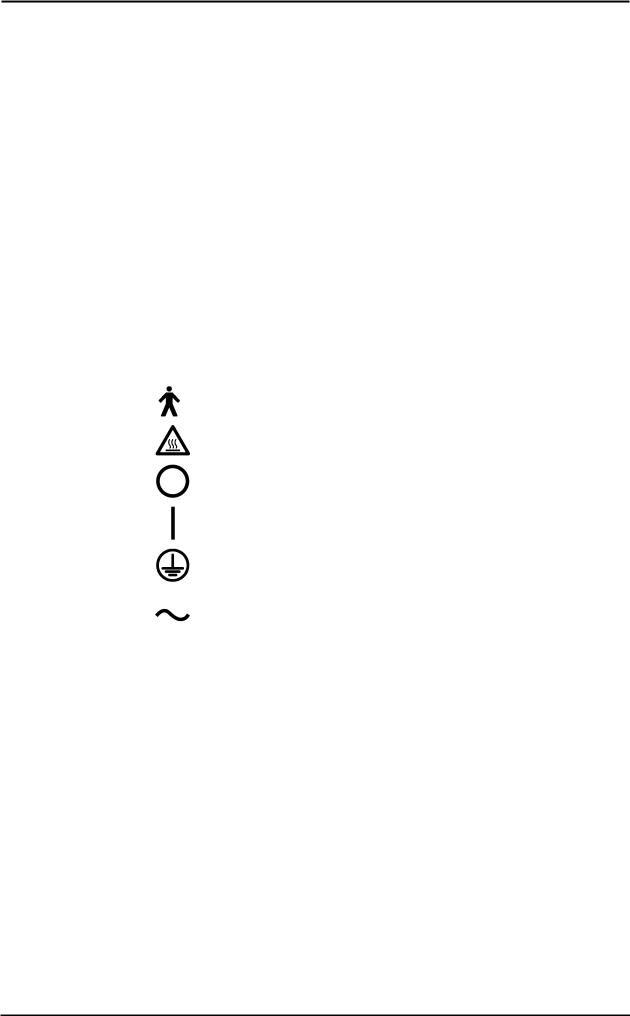
13.Graphical symbols used in accordance to present specifications are described here below:
Equipment type B (EN60601-1)
Danger: high temperature
OFF
ON
Protective earth ground
Alternating current
Chapter 1 - Getting started - 13

Contraindication
The physical strain to execute the respiratory manoeuvre is contraindicated in case of some symptoms or pathology. The following list is not complete and must be considered as a piece of mere information.
Contraindications for the Spirometer tests
Absolute contraindications
For FVC, VC and MVV tests:
•Post-operating state from thoracic surgery For FVC tests:
•Severe instability of the airways (such as a destructive bronchial emphysema)
•Bronchial non-specific marked hypersensitivity
•Serious problems for the gas exchange (total or partial respiratory insufficiency)
Relative contraindications
For FVC tests:
•spontaneous post-pneumothorax state
•arterial-venous aneurysm
•strong arterial hypertension
•pregnancy with complications at the 3rd month. For MVV test:
•hyperventilation syndrome
Contraindications for Bronchial provocation tests
The bronchial provocation tests must be executed according to the doctor’s discretion. There are not data that reveal specific contraindication for the bronchial provocation test through inhalation.
The modern standard processes have been revealing secure in several clinical studies. However it is recommendable to respect the following contraindications:
Absolute contraindications
•Serious bronchial obstruction (FEV1 in adults)
•Recent myocardium infarct
•Recent vascular-cerebral accident
•Known arterial aneurysm
•Incapacity for understanding the provocation test procedures and its implications.
Relative contraindications
•Bronchial obstruction caused by the respiratory manoeuvre.
•Moderate or serious bronchial obstruction. For ex. Predicted value FEV1 less than 1.51 in men and predicted value FEV1 in women less than 1.21.
•Recent infection in the superior air tracts
•During the asthmatic re-acuting
•Hypertension
•Pregnancy
•A pharmacology treatment epilepsy
Contraindications for Exercise testing
Read carefully the exercise testing chapter.
14 - Quark PFT User Manual

Environmental condition of use
COSMED units have been conceived for operating in medically utilised rooms without potential explosion hazards.
The units should not be installed in vicinity of x-ray equipment, motors or transformers with high installed power rating since electric or magnetic interferences may falsify the result of measurements or make them impossible. Due to this the vicinity of power lines is to be avoided as well.
Cosmed equipment are not AP not APG devices (according to EN 60601-1): they are not suitable for use in presence of flammable anaesthetic mixtures with air, oxygen or nitrogen protoxide.
If not otherwise stated in the shipping documents, Cosmed equipment have been conceived for operating under normal environmental temperatures and conditions [IEC 601-1(1988)/EN 60 601-1 (1990)].
•Temperature range 10°C (50°F) and 40°C (104°F).
•Relative humidity range 20% to 80%
•Atmospheric Pressure range 700 to 1060 mBar
•Avoid to use it in presence of noxious fumes or dusty environment and near heat sources.
•Do not place near heat sources.
•Cardiopulmonary resuscitation emergency equipment accessible.
•Adequate floor space to assure access to the patient during exercise testing.
•Adequate ventilation in the room.
Chapter 1 - Getting started - 15

Safety and conformity
Safety
IEC 601-1 (1988) /EN 60 601-1 (1990);
Find reported below the complete classification of the device:
•Class I type B device
•Protection against water penetration: IP00, ordinary equipment unprotected against water penetration
•Non sterile device
•Device not suitable in the presence of flammable anaesthetics;
•Continuous functioning equipment;
EMC
The system meets the EMC Directive 89/336
EN 60601-1-2
EN 55011 Class B (emission), IEC 1000-4-2, IEC 1000-4-3, IEC 1000-4-4
Quality Assurance
UNI ISO 9001 (Registration n° 387 Cermet)
Medical Device Directive (CE mark)
MDD 93/42/EEC (Notified Body 0476).
Class IIa
16 - Quark PFT User Manual

Keynotes
Here are the keynotes used to make the manual easier to read.
Typographic keynotes
These are the typographic keynotes used in the manual.
Style |
Description |
|
|
Bold |
indicates a control or a key to be pressed. |
“Italic” |
indicates a messages shown by the firmware. |
Graphic keynotes
These are the graphic keynotes used in the manual.
Illustration Description
shows the button to click in the software to activate the related feature.
Chapter 1 - Getting started - 17

Systems Overview
Quark PFT is a stationary and modular system designed for Pulmonary Function Testing. This system has been designed to meet the most accredited criteria and recommendations of the latest scientific publications.
The system incorporates “plug and play” circuitry, allowing the customisation of features and instant upgrades.
Quark PFT comes in the following configurations:
•PFT1; Spirometry, (FVC, SVC, MMV and bronchial-challenge tests).
•PFT2; adds Functional Residual Capacity testing via Nitrogen Washout and Closing Volume techniques.
•PFT3; combines Spirometry plus Lung Diffusing Capacity testing by adding CO and CH4 analysers.
•PFT4; integrates PFT2 and PFT3 features to become the Complete Pulmonary Function Testing instrument.
•ERGO Option: The PFT 2 and PFT 4 can be enhanced with the Pulmonary Gas Exchange module, allowing “breath by breath” analysis, indirect calorimetry and RS232 ergometer control, PFT3 can be upgraded to PFT4 or PFT4 ERGO.
Tests/Products table
Test |
PFT 1 |
PFT 2 |
PFT 3 |
PFT 4 |
PFTErgoPFT2ergoPFT4ergo |
||
Forced Vital Capacity |
• |
• |
• |
• |
option |
• |
• |
Slow Vital Capacity |
• |
• |
• |
• |
option |
• |
• |
Respiratory Pattern |
• |
• |
• |
• |
option |
• |
• |
Maximum Voluntary Ventilation |
• |
• |
• |
• |
option |
• |
• |
Bronchial Challenge Tests |
• |
• |
• |
• |
option |
• |
• |
Nitrogen Wash-out |
|
• |
|
• |
|
• |
• |
Closing Volume |
|
• |
|
• |
|
• |
• |
Lung Diffusing Capacity |
|
|
• |
• |
|
|
• |
P0.1 |
|
option |
|
option |
|
option |
option |
Exercise Testing |
|
|
|
|
• |
• |
• |
Indirect Calorimetry |
|
|
|
|
• |
• |
• |
Oxymetry |
option |
option |
option |
option |
option |
option |
option |
Ergometer Control |
|
|
|
|
• |
• |
• |
Integration with Quark C12/T12 ECG |
|
option |
option |
option |
option |
option |
option |
MIP-MEP |
option |
option |
option |
option |
|
option |
option |
18 - Quark PFT User Manual

Before starting
Before operating the Quark PFT system we strongly recommend to check the equipment and register you as a customer.
Checking the packing contents
Make sure that the package contains the items listed below. In case of missing or damaged parts, please contact Cosmed technical assistance.
Quark PFT1 standard packaging
Code |
Qty |
Description |
C00931-01-04 |
1 |
Quark PFT 1 unit |
C02120-01-05 |
1 |
Turbine Ø 28mm |
C02171-01-11 |
1 |
Kit optoelectronic reader Quark PFT1 |
A 662 100 001 |
2 |
Nose clips |
C01739-02-35 |
1 |
PC Software |
C00137-01-20 |
50 |
Paediatric paper mouthpieces |
C00136-01-20 |
50 |
Adult paper mouthpieces |
A 362 060 001 |
1 |
Power cord Schuko L 2m |
A 362 300 001 |
1 |
Serial cable RS 232 DB9 M/F |
A 680 013 630 |
2 |
Time Lag Fuses 5x20 250V T630 mA |
A 680 024 125 |
2 |
Time Lag Fuses 5x20 250V T 1,25A |
C00067-02-94 |
1 |
Registration card |
C01340-02-91 |
1 |
User manual Quark PFT |
C01999-02-DC |
1 |
Conformity declaration |
Quark PFT2 standard packaging |
||
Code |
Qty |
Description |
|
|
|
C00932-01-04 |
1 |
Quark PFT 2 unit |
C02120-01-05 |
1 |
Turbine Ø 28mm |
C02171-02-11 |
1 |
Kit optoelectronic reader Quark PFT2/3/4 |
A 182 310 001 |
5 |
Antimoisture filter |
C01590-01-05 |
1 |
Breathing valve PFT2 (1 way) |
C00243-01-06 |
1 |
Calibration syringe 3 litres |
C02210-02-08 |
1 |
Permapure L 2m |
C00269-01-20 |
20 |
PTE soft mouthpieces |
A 830 300 001 |
1 |
Air valve membrane |
A 182 300 004 |
10 |
Antibacterial filter |
A 108 106 002 |
2 |
Tank tubing 6x8 98 shore |
A 662 100 001 |
2 |
Nose clips |
C00137-01-20 |
50 |
Paediatric paper mouthpieces |
C00136-01-20 |
50 |
Adult paper mouthpieces |
C01739-02-35 |
1 |
PC software |
A 362 060 001 |
1 |
Power cord Schuko L 2 mt |
A 362 300 001 |
1 |
Serial cable RS 232 DB9 M/F |
A 680 013 630 |
2 |
Time Lag Fuses 5x20 250V T630 mA |
A 680 024 125 |
2 |
Time Lag Fuses 5x20 250V T1,25A |
C00067-02-94 |
1 |
Registration card |
C01340-02-91 |
1 |
User manual Quark PFT |
C01999-02-DC |
1 |
Conformity declaration |
Chapter 1 - Getting started - 19

Quark PFT3 standard packaging
Code |
Qty |
Description |
C00933-01-04 |
1 |
Quark PFT 3 unit |
C02120-01-05 |
1 |
Turbine Ø 28mm |
C02171-02-11 |
1 |
Kit optoelectronic reader Quark PFT2/3/4 |
C01730-01-05 |
1 |
Breathing valve PFT3/4 (3 ways) |
C00243-01-06 |
1 |
Calibration syringe 3 litres |
C02210-02-08 |
2 |
Permapure L 2 m |
A 182 300 004 |
10 |
Antibacterial filter |
A 830 300 001 |
1 |
Air valve membrane |
A 662 100 001 |
2 |
Nose clips |
C00137-01-20 |
50 |
Paediatric paper mouthpieces |
C00136-01-20 |
50 |
Adult paper mouthpieces |
A 182 310 001 |
5 |
Antimoisture filter |
C00269-01-20 |
20 |
PTE soft mouthpieces |
C01739-02-35 |
1 |
PC software |
A 108 106 002 |
1 |
Tank tubing 6x8 98 shore |
A 362 060 001 |
1 |
Power cord Schuko L 2 mt |
A 362 300 001 |
1 |
Serial cable RS 232 DB9 M/F |
A 680 013 630 |
2 |
Time Lag Fuses 5x20 250V T630 mA |
A 680 024 125 |
2 |
Time Lag Fuses 5x20 250V T630 mA |
C00067-01-94 |
1 |
Registration card |
C01340-02-91 |
1 |
User manual Quark PFT |
C01999-02-DC |
1 |
Conformity declaration |
Quark PFT4 standard packaging |
||
Code |
Qty |
Description |
|
|
|
C00934-01-04 |
1 |
Quark PFT4 unit |
C02120-01-05 |
1 |
Turbine Ø 28mm |
C02171-02-11 |
1 |
Kit optoelectronic reader Quark PFT 2/3/4 |
C01730-01-05 |
1 |
Breathing valve PFT3/4 (3 ways) |
C00243-01-06 |
1 |
Calibration syringe 3 litres |
C02210-02-08 |
2 |
Permapure L 2 m |
A 182 300 004 |
10 |
Antibacterial filter |
A 662 100 001 |
2 |
Nose clips |
C00137-01-20 |
50 |
Paediatric paper mouthpieces |
C00136-01-20 |
50 |
Adult paper mouthpieces |
A 182 310 001 |
5 |
Antimoisture filter |
C01739-02-35 |
1 |
PC software |
A 362 060 001 |
1 |
Power cord Schuko 2 mt |
A 362 300 001 |
1 |
Serial cable RS 232 DB9 M/F |
A 108 106 002 |
3 |
Tank Tubing 6x8 shore |
A 830 300 001 |
1 |
Air valve membrane |
A 680 013 630 |
2 |
Time Lag Fuses 5x20 250V T630 mA |
A 680 024 125 |
2 |
Time Lag Fuses 5x20 250V T1,25A |
C00067-02-94 |
1 |
Registration card |
C00269-01-20 |
20 |
PTE soft mouthpieces |
C01340-02-91 |
1 |
User manual Quark PFT |
C01999-02-DC |
1 |
Conformity declaration |
20 - Quark PFT User Manual

Quark PFT Ergo standard packaging
Code |
Qty |
Description |
C00935-01-04 |
1 |
Quark PFT Ergo unit |
C02120-01-05 |
1 |
Turbine Ø 28mm |
C02170-01-11 |
1 |
Kit optoelectronic reader Ergo |
C00243-01-06 |
1 |
Calibration syringe 3 litres |
C02210-02-08 |
2 |
Permapure L 2 m |
C01399-02-12 |
1 |
Cable in HR TTL |
A 800 900 001 |
2 |
Head cap for the adult masks |
C02125-01-10 |
1 |
Mask mouth/nose breath adult S |
C02135-01-10 |
1 |
Mask mouth/nose breath adult M |
C02145-01-10 |
1 |
Mask mouth/nose breath adult L |
A 182 310 001 |
5 |
Anti moisture filter |
C01739-02-35 |
1 |
PC software Spiro |
C01740-02-35 |
1 |
PC software Ergo |
A 362 060 001 |
1 |
Power cord Schuko L 2m |
A 362 300 001 |
1 |
Serial cable RS 232 DB9 M/F |
A 108 106 002 |
1 |
Tank tubing 6x8 98 Shore |
C00861-01-06 |
1 |
HR probe |
A 661 200 001 |
1 |
Polar belt |
A 661 200 002 |
1 |
HR transmitter watertight |
A 680 013 630 |
2 |
Time Lag Fuses 5x20 250V T630 mA |
A 680 024 125 |
2 |
Time Lag Fuses 5x20 250V T1,25A |
C00067-02-94 |
1 |
Registration card |
C01340-02-91 |
1 |
User manual Quark PFT |
C01999-02-DC |
1 |
Conformity declaration |
Quark PFT2 ergo Standard packaging |
||
Code |
Qty |
Description |
|
|
|
C00936-01-04 |
1 |
Quark PFT2 ergo unit |
C02120-01-05 |
1 |
Turbine Ø 28mm |
C02171-02-11 |
1 |
Kit optoelectronic reader Quark PFT 2/3/4 |
C01590-01-05 |
1 |
Breathing valve PFT2 |
A 800 900 001 |
2 |
Head cap for the adult masks |
C00243-01-06 |
1 |
Calibration Syringe 3L |
A 108 106 002 |
2 |
Tank tubing 6x8 98 shore |
C02210-02-08 |
2 |
Permapure L2m |
C01399-02-12 |
1 |
Cable in HR TTL |
C02125-01-10 |
1 |
Mask mouth/nose breath adult S |
C02135-01-10 |
1 |
Mask mouth/nose breath adult M |
C02145-01-10 |
1 |
Mask mouth/nose breath adult L |
C00861-01-06 |
1 |
HR probe |
A 661 200 001 |
1 |
HR elastic belt |
A 661 200 002 |
1 |
HR polar transmitter |
C00269-01-20 |
20 |
PTE soft mouthpiece |
C00137-01-20 |
50 |
Paediatric paper mouthpieces |
C00136-01-20 |
50 |
Adult paper mouthpieces |
A 182 300 005 |
10 |
Antibacterial filter |
A 182 310 001 |
5 |
Anti moisture filter |
A 362 060 001 |
1 |
Power cord Schuko 2m |
Chapter 1 - Getting started - 21

A 362 300 001 |
1 |
RS232 cable DB9 M/F |
A 662 100 001 |
2 |
Nose clips |
A 680 013 630 |
2 |
Time lag fuses 5x20 250V T630mA |
A 680 024 125 |
2 |
Time lag fuses 5x20 250V T1,25A |
A 830 300 001 |
1 |
Air valve membrane |
C01739-02-35 |
1 |
PC software Spiro |
C01740-02-35 |
1 |
PC software Ergo |
C01999-01-DC |
1 |
Conformity declaration |
C00067-02-94 |
1 |
Registration card |
C01340-02-91 |
1 |
Quark PFT User Manual |
Quark PFT4 ergo Standard packaging |
||
Code |
Qty |
Description |
C00937-01-04 |
1 |
Quark PFT4 ergo unit |
C02120-01-05 |
1 |
Turbine Ø 28mm |
C02171-02-11 |
1 |
Kit optoelectronic reader Quark PFT 2/3/4 |
A 800 900 001 |
2 |
Head cap for the adult masks |
C00243-01-06 |
1 |
Calibration Syringe 3L |
A 108 106 002 |
3 |
Tank tubing 6x8 98 shore |
C02210-02-08 |
1 |
Permapure L2m |
C01399-02-12 |
1 |
Cable in HR TTL |
C02125-01-10 |
1 |
Mask mouth/nose breath adult S |
C02135-01-10 |
1 |
Mask mouth/nose breath adult M |
C02145-01-10 |
1 |
Mask mouth/nose breath adult L |
C00861-01-06 |
1 |
HR probe |
A 661 200 001 |
1 |
HR elastic belt |
A 661 200 002 |
1 |
HR polar transmitter |
C00269-01-20 |
20 |
PTE soft mouthpiece |
C00137-01-20 |
50 |
Paediatric paper mouthpieces |
C00136-01-20 |
50 |
Adult paper mouthpieces |
A 182 300 005 |
10 |
Antibacterial filter |
A 182 310 001 |
5 |
Anti moisture filter |
A 362 060 001 |
1 |
Power cord Schuko 2m |
A 362 300 001 |
1 |
RS232 cable DB9 M/F |
A 662 100 001 |
2 |
Nose clips |
A 680 013 630 |
2 |
Time lag fuses 5x20 250V T630mA |
A 680 024 125 |
2 |
Time lag fuses 5x20 250V T1,25A |
A 830 300 001 |
1 |
Air valve membrane |
C01739-02-35 |
1 |
PC software Spiro |
C01740-02-35 |
1 |
PC software Ergo |
C01999-01-DC |
1 |
Conformity declaration |
C00067-02-94 |
1 |
Registration card |
C01340-02-91 |
1 |
Quark PFT User Manual |
22 - Quark PFT User Manual

Warranty registration
Before using the system, please take a moment to fill in the registration form and the warranty and return them to COSMED, by doing this you are eligible to the customers assistance service.
For further information, please refer to the enclosed registration and warranty form. If the form is not enclosed in the packaging, please contact directly COSMED.
Register the product via software
Together with the PC software, a registration software is supplied. With this software it is possible to fill in an electronic form with the customer information.
1.To run the software, double click on the icon Registration or select Registration… from ? menu.
2.Type the requested information and click Send… to send the form via e-mail to COSMED.
How to contact COSMED
For any information you may need, please contact the manufacturer directly at the following address:
COSMED S.r.l.
Via dei Piani di Monte Savello, 37 P.O. Box n. 3
00040 - Pavona di Albano Rome - ITALY
Voice: +39 (06) 931.5492
Fax: +39 (06) 931.4580
email: customersupport@cosmed.it Internet: http: //www.cosmed.it
Complain, feedback and suggestions
If you have any complain, feedback information or suggestion, please inform us at complain@cosmed.it.
Chapter 1 - Getting started - 23

Options/Accessories
The following options are available to enhance or to complete the Quark PFT system:
Code |
Quantity |
Description |
|
|
|
A 860 000 004 |
1 |
Calibration cylinder (5% CO2, 16% O2, balance N2) |
A 860 000 005 |
1 |
DLCO cylinder (0.3% CO, 0.3& CH4, 21% O2, balance N2) |
A 860 000 006 |
1 |
DLCO st. state cylinder (0.3% CO, 0.3% CH4, 21% O2, balance N2) |
A 860 000 007 |
1 |
O2 cylinder |
C01500-01-04 |
1 |
Trolley for Quark PFT |
C01700-01-04 |
1 |
Trolley with arm for Quark PFT |
A 870 150 001 |
1 |
Pressure regulator for cal./DLCO cylinder |
A 870 150 002 |
1 |
Pressure regulator for O2 cylinder |
C09001-02-99 |
1 |
Spirovis system for MIP and MEP measurement |
C00689-01-30 |
1 |
Mask mouth/nose breath ID28 adult XL |
C01278-01-30 |
1 |
Mask mouth/nose breath ID28 paediatric L |
C01277-01-30 |
1 |
Mask mouth/nose breath ID28 paediatric S |
A 800 900 004 |
1 |
Paediatric Headcap |
24 - Quark PFT User Manual

PC configuration required
•Pentium 133 MHz.
•Windows 95, 98, XP.
•16 Mb RAM .
•3.5 drive.
•VGA, SVGA monitor.
•Serial Port RS 232 available (2 serial ports in case of Ergometer control). An USB port can replace one RS232 serial port, if using the USB-RS232 adaptor (Cosmed code A 388 410 001).
•Any Mouse and Printer compatible with the MS Windows™ operative system.
•PC conform to European Directive 89/336 EMC
Chapter 1 - Getting started - 25

Technical features
Flowmeter |
|
|
Type: |
Bidirectional digital turbine Ø 28 mm |
|
Flow Range: |
0,03-20 L/sec |
|
Accuracy: |
± 2% |
|
Resistance: |
<0.7 cmH2O s/L @ 12 L/s |
|
Ventilation Range: |
0-300 litres x min |
|
Oxygen Sensor (O2) |
|
|
Type |
Galvanic Fuel Cell |
Paramagnetic (option) |
Response time: |
<120 ms |
<120 ms |
Range: |
0-100% O2 |
0-100% O2 |
Accuracy: |
±0.05% O2 |
±0.03% O2 |
Carbon Dioxide Sensor (CO2) |
|
|
Type: |
NDIR |
|
Response time: |
<120 ms |
|
Range: |
0-15% |
|
Accuracy: |
±0.03% |
|
Carbon Monoxide sensor (CO) |
|
|
Type: |
NDIR |
|
Response time: |
<200 ms |
|
Range: |
0-0.3% |
|
Accuracy: |
±0,003% |
|
Methane sensor (CH4) |
|
|
Type: |
NDIR |
|
Response time: |
<200 ms |
|
Range: |
0-0.3% |
|
Accuracy: |
±0,003% |
|
Humidity absorber |
|
|
Capillary of Nafion (Permapure ®) |
|
|
Power Supply |
|
|
Voltage: |
100V-240V ±10%; 50/60Hz |
|
Power consumption |
100W |
|
Environmental Sensors |
|
|
Temperature: |
0-50°C |
|
Barometer: |
400-800 mmHg |
|
Humidity: |
0-100% |
|
Dimension and Weight |
|
|
Dimensions: |
17x30x45 cm |
|
Weight: |
8 Kg |
|
Aux inputs
With the auxiliary RS 232 port and the digital (TTL) input for external ECG signal, Quark PFT offers the possibility to integrate data coming from many equipment such as ECGs and ergometers.
26 - Quark PFT User Manual

Measurements

Pulmonary function tests and measured parameters
Spirometry Tests
FVC - Forced Vital Capacity
Symbol |
UM |
Parameter |
FVC |
l |
Forced Expiratory Vital Capacity |
FEV1 |
l |
Forced Expiratory Volume in 1 sec |
FEV1/FVC% |
% |
FEV1 as a percentage of FVC |
PEF |
l/sec |
Peak Expiratory Flow |
FEV0.5 |
l |
Forced Expiratory Volume in 0.5 sec |
FEV6 |
l |
Forced Expiratory Volume in 6 sec |
FEV1/FEV6 |
% |
FEV1 as a percentage of FEV6 |
FEV6/FVC% |
% |
FEV6 as a percentage of FVC |
Best FVC |
l |
Best Forced Expiratory Vital Capacity |
Best FEV1 |
l |
Best Forced Expiratory Volume in 1 sec |
Best PEF |
l/sec |
Best Peak Expiratory Flow |
Vmax25% |
l/sec |
Expiratory Flow when 75% of the FVC remains to be exhaled |
Vmax50% |
l/sec |
Expiratory Flow when 50% of the FVC remains to be exhaled |
Vmax75% |
l/sec |
Expiratory Flow when 25% of the FVC remains to be exhaled |
FEF25-75% |
l/sec |
Mid-exp flow between 25-75%FVC |
FET100% |
sec |
Forced expiratory time |
FEV2 |
l |
Forced Expiratory Volume in 2 sec |
FEV3 |
l |
Forced Expiratory Volume in 3 sec |
FEV2/FVC% |
% |
FEV2 as a percentage of FVC |
FEV3/FVC% |
% |
FEV3 as a percentage of FVC |
FEV1/VC% |
% |
Tiffenau index |
FEF50-75% |
l/sec |
Mid-exp flow between 50-75%FVC |
FEF75-85% |
l/sec |
Mid-exp flow between 75-85%FVC |
FEF0.2-1.2% |
l/sec |
Mid-exp flow between 0.2 l - 1.2 l |
FiVC |
L |
Inspiratory Forced Vital Capacity |
FiF25-75% |
l/sec |
Forced mid-inspiratory flow |
FiV1 |
l/sec |
Forced Inspiratory Volume in 1 sec |
PIF |
l/sec |
Peak Inspiratory Flow |
VEXT |
ml |
Extrapolated Volume (back extrapolation) |
PEFT |
msec |
Time to PEF (10% - 90%) |
VC/IVC - Slow Vital Capacity and Ventilatory pattern |
||
Symbol |
UM |
Parameter |
EVC |
l |
Expiratory Vital Capacity |
IVC |
l |
Inspiratory Vital Capacity |
ERV |
l |
Expiratory Reserve Volume |
IRV |
l |
Inspiratory Reserve Volume |
IC |
l |
Inspiratory Capacity |
VE |
l/min |
Expiratory Minute Ventilation |
Vt |
l |
Tidal Volume |
Rf |
1/min |
Respiratory Frequency |
Ti |
sec |
Duration of Inspiration |
Te |
sec |
Duration of Expiration |
28 - Quark PFT User Manual

Ttot |
sec |
Duration of Total breathing cycle |
|
Ti/Ttot |
—- |
Ti/Ttot ratio |
|
Vt/ti |
l/sec |
Vt/ti ratio |
|
MVV - Maximum Voluntary Ventilation |
|||
Symbol |
UM |
Parameter |
|
MVV |
l/min |
Maximum Voluntary Ventilation |
|
MVt |
l |
Tidal Volume (during MVV) |
|
MRf |
1/min |
Maximum Respiratory frequency |
|
MVVt |
sec |
MVV duration time |
|
Bronchoprovocation Response |
|
||
Symbol |
UM |
Parameter |
|
FallFEV1 |
% |
Fall in FEV1 from baseline or post diluent |
|
FallVmax50% |
% |
Fall in Vmax50% from baseline or post diluent |
|
P10 |
—- |
Provocative dose causing FEV1 to fall 10% from baseline |
|
P15 |
—- |
Provocative dose causing FEV1 to fall 15% from baseline |
|
P20 |
—- |
Provocative dose causing FEV1 to fall 20% from baseline |
|
Multi-breath Nitrogen wash-out |
|||
Symbol |
UM |
Parameter |
|
|
|
|
|
FRC |
l |
Functional Residual Capacity |
|
RV |
l |
Residual Volume |
|
TLC |
l |
Total Lung Capacity |
|
Vtot |
l |
Total volume during the test |
|
FetN2 |
% |
Final Nitrogen Concentration |
|
Wash-out time |
m |
Duration of wash-out |
|
VDA |
L |
Anatomic dead space |
|
LCI |
mm:ss Lung Clearance Index |
||
AMDN |
mm:ss Mean Dilution Number |
||
FRC/TLC |
% |
FRC/TLC ratio |
|
CEV |
l |
Cumulated Volume of inspired Oxygen |
|
RV/TLC |
% |
RV/TLC ratio (Motley index) |
|
Closing Volume test |
|
|
|
Symbol |
UM |
Parameter |
|
CV |
l |
Closing Volume |
|
VC(c.v.) |
l |
Expired volume during wash-out |
|
V_I |
l |
Related Volume at the end of phase I |
|
V_II |
l |
Related Volume at the end of phase II |
|
V_III |
l |
Related Volume at the end of phase III |
|
m, q, R^2 |
l |
Parameters of the linear fitting on the alveolar plateau |
|
CV/VC% |
% |
CV as a percentage of VC |
|
VDf |
ml |
Dead volume (Fowler) |
|
CO diffusion Test |
|
|
|
Symbol |
UM |
|
Parameter |
DLco |
ml/min/mmHg |
CO Diffusion Capacity |
|
DLco 3eq |
ml/min/mmHg |
DLco computed with the Three Equations method. |
|
Chapter 2 - Measurements - 29

DLco corr |
ml/min/mmHg |
CO Diffusion Capacity corrected for Hb, COHb, PB. |
DLco/VA |
ml/min/mmHg |
Krogh factor |
Dm |
ml/min/mmHg |
Membrane Diffusion capacity |
Vc |
ml |
Capillary Volume |
FaCO |
% |
Alveolar concentration of CO |
FACH4 |
% |
Alveolar concentration of CH4 |
ta |
sec |
Apnoea time |
FiCO |
% |
Inspiratory concentration of CO |
FiCH4 |
% |
Expiratory concentration of CH4 |
wash-out vol. |
% |
Rejection Volume |
Vsample vol. |
% |
Sampling volume |
IVC (DLCO) |
l |
IVC calculated during test DLCO |
TLC (DLCO) |
l |
Total Lung Capacity |
Respiratory Drive - P0.1 Test |
|
|
Symbol |
UM |
Parameter |
|
|
|
P0.1 |
cmH2O |
Respiratory drive |
FiCO2 |
% |
Inspiratory CO2 (PFT2 and PFT4 only) |
RF |
1/min |
Respiratory Frequency |
Vt |
l |
Tidal Volume |
MIP/MEP
Symbol |
UM |
Parameter |
MIP |
cm H2O Maximum inspiratory pressure |
|
MEP |
cm H2O Maximum expiratory pressure |
|
Cardio pulmonary Exercise Testing (CPET)
The Ergo option makes the Quark PFT a complete Cardiopulmonary Exercise Testing system able to measure the following parameters:
Breath by Breath exercise testing
|
Symbol |
UM |
Parameter |
|
|
VO2 |
ml/min |
Oxygen Uptake |
|
|
VCO2 |
ml/min |
Carbon Dioxide production |
|
|
Vt |
l |
Tidal Volume |
|
|
FetO2 |
% |
End Tidal O2 |
|
|
FetCO2 |
% |
End Tidal CO2 |
|
|
R |
--- |
Respiratory Quotient |
|
|
VE |
l/min |
Ventilation |
|
|
HR |
1/min |
Heart Rate |
|
|
Qt |
l |
Cardiac output |
|
|
AT |
--- |
Anaerobic Threshold |
|
|
VE |
l/min |
Ventilation |
|
|
SV |
l/min |
Stroke volume |
|
|
RF |
1/min |
Respiratory Frequency |
|
|
FeO2, FeCO2 |
% |
Averaged expiratory concentration of O2 e CO |
|
|
VE/VO2 |
--- |
ventilatory equivalent for O2 |
|
|
VE/VCO2 |
--- |
ventilatory equivalent for CO2 |
|
|
VO2/HR |
ml/beat |
Oxygen pulse |
|
|
VO2/Kg |
ml/min/Kg |
VO2 per Kg |
|
30 - Quark PFT User Manual |
|
|
|
|
|
|
|
|
|
 Loading...
Loading...