Page 1

Bio-Plex Pro
™
Diabetes Assays
Instruction Manual
For technical support, call your local Bio-Rad office, or in the U.S., call 1-800-424-6723.
For research use only. Not for diagnostic procedures.
Page 2

Table of Contents
Introduction 1
Principle 2
Kit Contents and Storage 4
Recommended Materials 5
Assay Workflow 6
Important Considerations 7
Detailed Instructions
1. Plan Plate Layout 8
2. Prepare Instrument 9
3. Prepare Wash Method 10
4. Prepare Standards 11
5. Prepare Samples 16
6. Prepare Coupled Beads 20
7. Run Assay 22
8. Read Plate 28
Troubleshooting Guide 35
Appendix: Protease Inhibitors 40
Plate Layout Template 41
Calculation Worksheet 42
Safety Considerations 46
Legal Notices 46
Ordering Information 47
Page 3

Introduction
Bio-Plex Pro™ diabetes assays are magnetic bead–based multiplex assays
designed to measure multiple diabetes-related biomarkers in a minimal
volume of matrix such as serum, plasma, tissue culture supernatant,
or other biological fluids. The biomarkers chosen for these assays are
circulating proteins involved in the regulation of glucose metabolism.
These multiplex assays are configured for the detection of various
metabolic markers in human, mouse, and rat samples. The multiplex
assays for human matrices were tested and found to be cross-reactive to
at least four common non-human primate (NHP) species, including rhesus,
cynomolgus, baboon, and marmoset. The degree of cross-reactivity was
profiled according to the ability of each assay to detect these metabolic
markers in the sera and mitogen-stimulated peripheral blood mononuclear
cell (PBMC) culture supernatant of these animals.
The high level of gene homology across the major NHP species may
allow the NHP diabetes assays to be used in other species, such as
chimpanzees, African green monkey, and pigtail macaques. However,
assay performance has not been specifically evaluated in these animals.
Cross-Reactivity with Non-Human Primates
African green Pigtail
Rhesus Cynomolgus Baboon Marmoset Chimpanzee monkey macaque
Tested
Not tested • • •
• • • •
Bio-Plex Pro assays enable researchers to quantify multiple protein
biomarkers in a single well of a 96-well plate in 3–4 hours. These robust
immunoassays require as little as 12.5 μl serum or plasma or 50 μl cell
culture supernatant or other biological fluid. The use of magnetic (MagPlex)
beads allows researchers to automate wash steps on a Bio-Plex Pro (or
similar) wash station. Magnetic separation offers greater convenience,
productivity, and reproducibility compared to vacuum filtration.
For more information please visit www.bio-rad.com/bio-plex.
1
Page 4

Principle
Technology
The Bio-Plex® multiplex system is built upon the three core elements of
xMAP technology:
n
Fluorescently dyed microspheres (also called beads), each with a distinct
color code or spectral address to permit discrimination of individual
tests within a multiplex suspension. This allows simultaneous detection
of up to 500 different types of molecules in a single well of the 96-well
microplate on the Bio-Plex
molecules on the Bio-Plex
of molecules on the Bio-Plex
n
On the Bio-Plex 200 and Bio-Plex 3D systems, a dedicated flow
cytometer with two lasers and associated optics to measure the
different molecules bound to the surface of the beads. In the
Bio-Plex MAGPIX, the entire sample load volume is injected into a
chamber where the beads are imaged using LED and CCD technology
n
A high-speed digital signal processor that efficiently manages the
fluorescence data
Assay Format
Bio-Plex Pro™ assays are essentially immunoassays formatted on
magnetic beads. The assay principle is similar to that of a sandwich
ELISA (Figure 1). Capture antibodies directed against the desired
biomarker are covalently coupled to the beads. Coupled beads react
with the sample containing the biomarker of interest. After a series of
washes to remove unbound protein, a biotinylated detection antibody
is added to create a sandwich complex. The final detection complex is
formed with the addition of streptavidin-phycoerythrin (SA-PE) conjugate.
Phycoerythrin serves as a fluorescent indicator or reporter.
®
3D system, up to 100 different types of
®
200 system, and up to 50 different types
®
MAGPIX™ system
2
Page 5
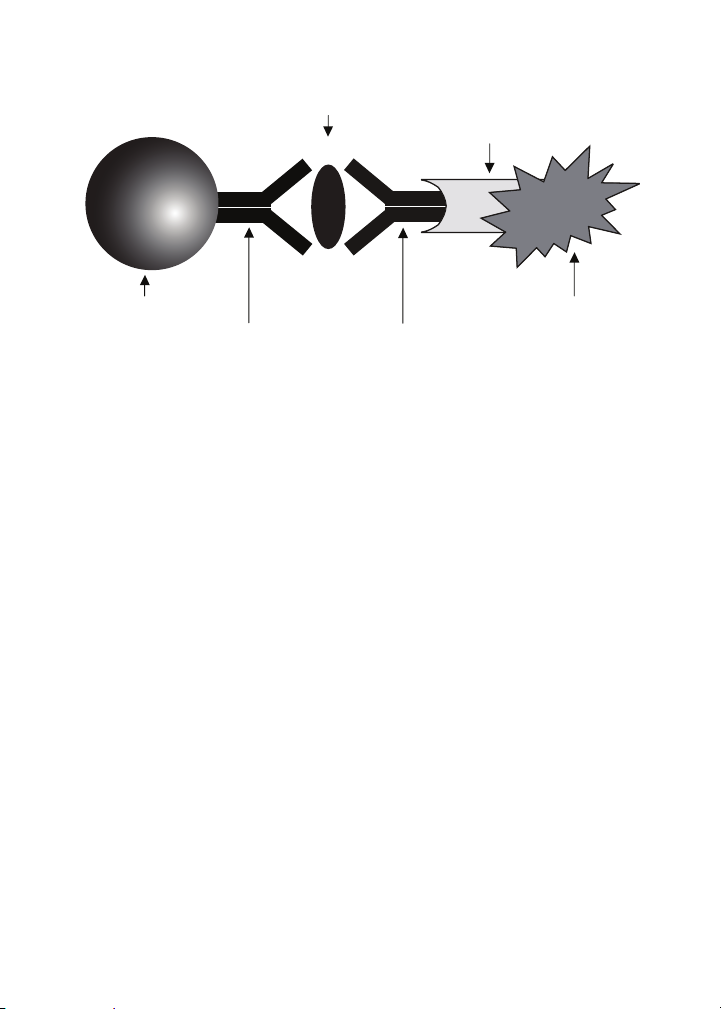
Biomarker
of Interest
Streptavidin
Magnetic Bead
Capture
Antibody
Fig. 1. Bio-Plex sandwich immunoassay.
Biotinylated
Detection
Antibody
Phycoerythrin
Fluorescent
Reporter
Data Acquisition and Analysis
Data from the reactions are acquired using a Bio-Plex system or similar
Luminex-based reader. When a multiplex assay suspension is drawn into
the Bio-Plex 200 reader, for example, a red (635 nm) laser illuminates the
fluorescent dyes within each bead to provide bead classification and thus
assay identification. At the same time, a green (532 nm) laser excites PE
to generate a reporter signal, which is detected by a photomultiplier tube
(PMT). A high-speed digital processor manages data output, and
Bio-Plex Manager
™
software presents data as median fluorescence
intensity (MFI) as well as concentration (pg/ml). The concentration of
analyte bound to each bead is proportional to the MFI of reporter signal.
Using Bio-Plex Data Pro
™
software, data from multiple instrument runs
can be combined into a single project for easy data management, quick
visualization of results, and simple statistical analysis.
3
Page 6

Kit Contents and Storage
Reagents Supplied
Bio-Plex Pro™ diabetes assays are offered in a convenient kit format that
includes assay, reagent, and diluent components in a single box (Table 1).
Table 1. Contents of Bio-Plex Pro diabetes assays.*
Component
Standard diluent (not included in adiponectin/adipsin assays)
Sample diluent (not included in adiponectin/adipsin assays)
Serum-based diluent (adiponectin/adipsin assays only)
Assay buffer
Wash buffer
Detection antibody diluent
Streptavidin-PE (100x)
Filter and/or flat bottom plate (96-well)
Sealing tape
Assay Quick Guide 1 booklet
Coupled magnetic beads (20x)
Detection antibodies (20x)
Standard
* Volumes shown are approximate. Quantities in Bio-Plex
1 x 96-Well
Format
10 ml
40 ml
70 ml
50 ml
200 ml
5 ml
1 tube
1 plate
1 pack of 4
1 tube
1 tube
1 vial
®
Express assays will vary.
Storage and Stability
Kit contents should be stored at 4°C and never frozen. Coupled magnetic
beads and streptavidin-PE should be stored in the dark. All components
are guaranteed for a minimum of six months from the date of purchase
when stored as specified.
10 x 96-Well
Format
100 ml
80 ml
N/A
500 ml
1.5 L
50 ml
1 tube
10 plates
10 packs of 4
1 tube
1 tube
10 vials
4
Page 7

Table 2. Recommended materials.
Item
Bio-Plex Pro Assays Quick Guide 2
Ordering Information
Bulletin #10024973 (download
at www.bio-rad.com/bio-plex)
Bio-Plex 200 system or Luminex system with HTF
Bio-Plex validation kit
Bio-Rad catalog #171-000205
Bio-Rad catalog #171-203001
Run the validation kit monthly to ensure optimal performance
of fluidics and optics systems
Bio-Plex calibration kit
Bio-Rad catalog #171-203060
Run the calibration kit daily to standardize fluorescence signal
Bio-Plex Pro wash station
Bio-Rad catalog #300-34376
For use with magnetic bead–based assays only
Bio-Plex Pro II wash station
Bio-Rad catalog #300-34377
For use with both polystyrene (nonmagnetic) and magnetic
bead–based assays
Bio-Plex handheld magnetic washer
Bio-Rad catalog #170-20100
For use with magnetic bead–based assays only
Bio-Plex Pro flat bottom plates, 40 x 96-well
Bio-Rad catalog #171-025001
For magnetic separation on the Bio-Plex Pro wash station
Microtiter plate shaker
IKA MTS 2/4 shaker for 2 or 4 microplates
IKA catalog #320-8000
or
Barnstead/Lab-Line Model 4625 plate
VWR catalog #57019-600
shaker (or equivalent capable of 300–1,100 rpm)
®
Aurum™ vacuum manifold
Bio-Rad
For vacuum filtration
BR-2000 vortexer
Reagent reservoirs, 25 ml
For capture beads and detection antibodies
Reagent reservoir, 50 ml (for reagents and buffers)
Pall Life Science Acrodisc, 25 mm PF syringe filter
(0.8/0.2 µm Supor membrane)
Filter plate, 1 x 96-well, clear plastic lid and tray
®
Titertube
Micro test tubes
For preparing replicate standards, samples, and controls
Bio-Rad catalog #732-6470
Bio-Rad catalog #166-0610
VistaLab catalog #3054-1002
or
VistaLab catalog #3054-1004
VistaLab catalog #3054-1006
Pall Life Sciences
catalog #4187
Bio-Rad catalog #171-304502
Bio-Rad catalog #223-9390
prior to loading the plate
Other: 15 ml polypropylene tubes for reagent dilutions, calibrated pipets, pipet tips, sterile
distilled water, aluminum foil, absorbent paper towels, 1.5 or 2 ml microcentrifuge tubes, and
standard flat bottom microplate (for calibrating vacuum manifold).
5
Page 8

Assay Workflow
Prewet wells
(for lter plate only)
Add 50 μl 1x beads to wells
Wash 2 x 100 μl
Add 50 μl standards, blank, samples
incubate 1 hr at RT with shaking at 850 rpm
Wash 3 x 100 μl
Add 25 μl 1x detection antibody, incubate
30 min at RT with shaking at 850 rpm
Wash 3 x 100 μl
Add 50 μl 1x streptavidin-PE, incubate
10 min at RT with shaking at 850 rpm
Wash 3 x 100 μl
Resuspend in 125 μl assay buffer,
shake at 850 rpm for 30 sec
Read plate on Bio-Plex system
6
Page 9

Important Considerations
Instruments and Software
The diabetes assays described in this manual are compatible with all
currently available Luminex-based life science research instruments.
Assays can be read and analyzed with either Bio-Plex Manager
or Luminex xPONENT software.
Assay Procedures
Pay close attention to vortexing, shaking, and incubation times and to
Bio-Plex
®
reader PMT (RP1) setting, as these have been optimized
specifically for each assay panel.
Assay Quick Guide
Each assay kit includes a printed Bio-Plex Pro Assay Quick Guide (bulletin
#10024973), which can be used to prepare and run a full 1 x 96-well assay
plate. Users can also download a copy at www.bio-rad.com/bio-plex.
Bead Regions
Bead regions for all analytes are listed in the Read Plate section.
Multiplexing Compatibility
For human and mouse, the maximum number of singleplex diabetes
and cytokine analytes that may be mixed is limited by the 10x cytokine
antibody stock concentrations as shown in the table below.
Table 3. Maximum number of singleplex diabetes and cytokine analytes that may
be multiplexed.
Human, mouse, and rat diabetes
Mouse cytokine (group III) analytes (20x)
0 2 4 6 8 10
™
software
Human and mouse cytokine (groups I, II)
analytes (10x)
10 9 8 7 6 5
7
Page 10

1. Plan Plate Layout
Prior to running the assay, determine the total number of wells in the
experiment using the Plate Layout Template on page 41 or the Plate
Formatting tab in Bio-Plex Manager
shown in Figure 2, with all conditions in duplicate.
1. Assign standards to columns 1 and 2, with the highest
concentration in row A and the lowest concentration in row H.
2. Assign the blank to wells A3 and A4. The blank should consist of your
chosen standard diluent and be processed in the same manner as
sample and standard wells. Note that Bio-Plex Manager automatically
subtracts the blank (B) MFI value from all other assay wells.
3. User-defined controls are assigned to wells in columns 3 and 4.
4. The remainder of the plate is available for samples.
5. Once the total number of wells is known, calculate the required
volumes of beads, detection antibody, and streptavidin-PE needed.
Use Tables 9–11, 14–16, and 17, respectively, or the Calculation
Worksheet on page 43.
Legend
S Standard
™
software. A suggested plate layout is
B Blank
X Samples
C Controls
Fig. 2. Suggested plate layout. For detailed instructions on
plate formatting in Bio-Plex Manager, see Section 8.
8
Page 11

2. Prepare Instrument
Start up and calibrate the Bio-Plex® 100/200 or similar system with
Bio-Plex Manager
kit should be run daily or before each use of the instrument to standardize
the fluorescent signal. To prepare either a Bio-Plex 3D or Bio-Plex
MAGPIX
The validation kit should be run monthly to ensure performance of fluidics
and optics systems. Refer to either the software manual or online Help for
directions on how to conduct validation.
™
reader, consult its respective user manual.
™
software prior to setting up the assay. The calibration
®
Start Up System (Bio-Plex 100, 200, or Similar)
1. Empty the waste bottle and fill the sheath fluid bottle before starting
if high throughput fluidics (HTF) are not present. This will prevent
fluidic system backup and potential data loss.
2. Turn on the reader, XY platform, and HTF (if included). Allow the
system to warm up for 30 min (if not already done).
3. Select Start up
for 4 hr without acquiring data, the lasers will automatically turn off.
To reset the 4-hr countdown, select Warm up
lasers/optics to reach operational temperature.
and follow the instructions. If the system is idle
and wait for the
Calibrate System
1. Select Calibrate and confirm that the default values for CAL1
and CAL2 are the same as the values printed on the bottle of
Bio-Plex calibration beads. Use the Bio-Plex system low RP1
target value even if assays will be run at high RP1.
2. Select OK and follow the software prompts for step-by-step
instructions for CAL1 and CAL2 calibration.
Note: In Bio-Plex Manager version 6.1 and higher, startup, warm up,
and calibration can be performed together by selecting the “Start up and
calibrate”
icon.
9
Page 12

3. Prepare Wash Method
Bio-Plex Pro™ assays are compatible with both magnetic separation and
vacuum filtration methods. However, for best results, we recommend
performing the assays in a flat bottom plate with magnetic separation.
Table 4. Summary of compatible wash stations and plate types.
Wash Method Wash Station Assay Plate
Magnetic separation Bio-Plex Pro Flat bottom plate
Bio-Plex Pro II (use MAG programs)
Bio-Plex
Vacuum filtration Bio-Plex Pro II (use VAC programs) Filter plate
Vacuum manifold (manual)
Setting up the Bio-Plex Pro or Bio-Plex Pro II
Wash Station
The wash station does not require calibration; however, it should be primed
before use. For more information, refer to the Bio-Plex Pro and Pro II wash
station quick guide (bulletin #5826).
1. Install the appropriate plate carrier on the wash station.
2. Use the prime procedure to prime channel 1 with wash buffer.
Setting Up the Bio-Plex Handheld Magnetic Washer
Place an empty flat bottom plate on the magnetic washer by sliding
it under the retaining clips. Push the clips inward to secure the plate.
Make sure the plate is held securely. If needed, the clips can be adjusted
for height and tension. For detailed instructions, refer to the user guide
(bulletin #10023087).
®
handheld magnetic washer
Setting up a Vacuum Manifold
Calibrate the vacuum manifold by placing a standard 96-well flat bottom
plate on the unit and adjusting the pressure to –1 to –3" Hg. In general,
100 µl liquid should take 3–4 sec to clear the well. For more detailed
instructions, refer to bulletin #10005042.
10
Page 13

4. Prepare Standards
General Instructions
n
It is essential to prepare standards exactly as described in this section.
Incorrect preparation may lead to low signal or variable measurements
from plate to plate
n
The peel-off sticker provided with the standards lists the most
concentrated point on the standard curve (S1). Enter this information
into Bio-Plex Manager
n
For users who wish to mix assays from different panels, such as
diabetes assays with group I cytokines, guidance is provided here for
mixing 2 different lyophilized standards. Bead regions were chosen to
avoid overlap whenever possible. However, performance of multiplexes
containing assays from different groups have not been extensively
validated. Therefore, users must confirm that the assay performance is
still fit for their purpose
Selecting a Diluent for Standards
Refer to Table 5 for recommended diluents based on different sample types.
As a general rule, reconstitute and dilute standards in a diluent similar to
the final sample type or sample matrix.
Table 5. Summary of recommended diluents for standards.
Sample Type Diluent for Standards Add BSA
Serum and plasma Standard diluent None
Culture media, with serum Culture media None
Culture media, serum-free Culture media To 0.5% final
For Adiponectin and/or Adipsin Assays
Serum and plasma Serum-based diluent None
™
software as instructed in section 8
11
Page 14

RP1 (PMT) Setting for Standard Curves
The Bio-Plex 200 and 3D systems have two RP1 (PMT or photomultiplier
tube) setting options, while the Bio-Plex
®
MAGPIX™ has no PMT and
therefore no PMT setting options. Instead, MAGPIX uses default instrument
settings similar to low PMT on the Bio-Plex 200 (Table 6).
Table 6. Overview of PMT setting options on Bio-Plex systems.
Instrument RP1 (PMT)
Bio-Plex 100, 200* Low, high
Bio-Plex 3D* Standard, enhanced
Bio-Plex MAGPIX* N/A, use default instrument settings
* Or similar Luminex-based system.
The Bio-Plex Pro diabetes assays were developed on the high PMT
setting using the Bio-Plex 200 system. Protocols using alternative standard
dilution series or low PMT settings should be validated by the end user, for
example when mixing diabetes assays with cytokine assays (Table 7).
Table 7. Settings for optimal sensitivity on the Bio-Plex 200 system*.
Assay Low RP1 (PMT) High RP1 (PMT)
Human/NHP* diabetes
Mouse diabetes User validation required*
Rat diabetes
Low RP1 (PMT), broad High RP1 (PMT), narrow
Cross-panel mixing
Human diabetes + cytokines
Mouse diabetes + cytokines User validation required*
Rat diabetes + cytokines
* Contact Bio-Rad technical support for the most up-to-date recommendations on PMT
settings and cross-panel multiplexing compatibility.
range cytokine curve range cytokine curve
•
•
•
•
•
•
Reconstitute a Single Vial of Diabetes Standards
This procedure prepares enough material to run each dilution in duplicate.
1. Gently tap the vial containing the lyophilized standard.
2. Add 500 μl of the appropriate standard diluent. Do not use assay
buffer or sample diluent to reconstitute the standards.
12
Page 15

3. Gently vortex the reconstituted standard for 5 sec then incubate on
ice for 30 min. Be consistent with the incubation time in every assay
to ensure best results.
4. During the incubation period, prepare the samples as instructed in the
Prepare Samples section.
Prepare Diabetes Standard Dilution Series from a
Single Antigen Vial
The following procedure produces an eight-point standard curve with a
fourfold dilution between each point. Pipet carefully using calibrated pipets
and use new pipet tips for every volume transfer.
1. Label nine 1.5 ml polypropylene tubes S1 through S8 and Blank.
2. Add the specified volume of standard diluent to each tube
(Figures 3 and 4).
3. Vortex the reconstituted standards gently for 5 sec before removing
any volume. Add 128 µl into the S1 tube containing 72 µl of standard
diluent. Vortex at medium speed for 5 sec, then use a new pipet tip
to transfer 50 µl from S1 tube to S2 tube.
4. Continue with 1:4 (fourfold) serial dilutions from tube S2 to S8 as
shown in Figure 3. Use reconstituted and diluted standards
immediately. Do not freeze for future use.
Fig. 3. Preparing a fourfold dilution series of diabetes standards.
128 50 50 50 50 50 50 50
Transfer Volume, µl
Reconstituted
Diabetes
Standard
72 150 150 150 150 150 150 150 150
S1 S2 S3 S4 S5 S6 S7 S8 Blank
13
Diluent, µl
Page 16

Please skip this section if not mixing diabetes with cytokine assays.
Reconstituting Standards for Cross-Panel Plexing
Follow these directions when mixing human or mouse diabetes and
cytokine assays. Note that rat diabetes and cytokine standards are
premixed into one standard vial. Therefore, no extra mixing is required.
Two mixing scenarios are provided in Figures 4 and 5 for detection at high
and low PMT respectively. One results in a narrow range cytokine standard
curve for detection at high RP1 (PMT); the other gives a broad range
cytokine standard curve for detection at low RP1 (PMT) setting.
1. Gently tap both vials of lyophilized diabetes and cytokine standards.
2. For high PMT setting/narrow range cytokine standard curve, add
500 μl of the appropriate standard diluent to each vial. For low PMT
setting/broad range cytokine standard curve, add 250 µl of diluent to
each vial. Do not use assay buffer or sample diluent to reconstitute
the standards.
3. Gently vortex the reconstituted standards for 5 sec then incubate
on ice for 30 min. Be consistent with the incubation time in every
assay to ensure best results.
4. During the incubation period, prepare the samples as instructed in the
Prepare Samples step.
Preparing Serial Dilutions
Pipet carefully with calibrated pipets, and use new pipet tips for every
volume transfer.
1. Label nine 1.5 ml polypropylene tubes S1 through S8 and Blank.
2. For high PMT/narrow range cytokine standard curve, add 59.2 µl of
standard diluent to the S1 tube. For low PMT/broad range cytokine
standard curve, add 72 µl of standard diluent to S1 (Figures 4 and 5).
14
Page 17

3. For high PMT/narrow range cytokine standard curve, add 12.8 μl
of the reconstituted cytokine standard and 128 μl of the
reconstituted diabetes standard to the S1 tube for a total volume
of 200 μl (Figure 4). Vortex at medium speed for 5 sec.
4. Add 150 μl of standard diluent to the remaining tubes, as shown
in Figure 4.
Fig. 4. S1 mixture and fourfold dilution series of diabetes and cytokine standards for
detection at high PMT. Produces a narrow range cytokine standard curve.
12.8 µl 128 µl
50 50 50 50 50 50 50
Transfer
Volume, µl
500 µl
Reconstituted
Cytokine
Standard
“S1” Tube, 200 µl Total
59.2 µl
Standard Diluent
500 µl
Reconstituted
Diabetes
Standard
150 150 150 150 150 150 150 150
S2 S3 S4 S5 S6 S7 S8 Blank
Diluent, µl
5. For low PMT/broad range cytokine standard curve, add 64 µl of each
standard to the S1 tube, total volume 200 µl (Figure 5). Vortex for 5 sec.
6. Use a new pipet tip to transfer 50 μl from the S1 tube to the S2 tube
containing standard diluent.
7. Continue with 1:4 (fourfold) serial dilutions from tube S2 to S8 as
shown in Figure 5.
Fig. 5. S1 mixture and fourfold dilution series of diabetes and cytokine standards for
detection at low PMT. Produces a broad range cytokine standard curve.
Transfer
Volume, µl
Diluent, µl
250 µl
Reconstituted
Cytokine
Standard
Standard Diluent
“S1” Tube, 200 µl Total
64 µl 64 µl
72 µl
50 50 50 50 50 50 50
250 µl
Reconstituted
Diabetes
Standard
150 150 150 150 150 150 150 150
S2 S3 S4 S5 S6 S7 S8 Blank
15
Page 18

5. Prepare Samples
General guidelines on preparing samples derived from serum, plasma,
and tissue culture supernatant are provided here, including the use of
protease inhibitors with plasma samples.
n
Once thawed, keep samples on ice. Prepare dilutions just prior to the
start of the assay and equilibrate to room temperature before use
n
Prepare sample dilutions in 1.5 or 2 ml polypropylene microcentrifuge
tubes. If a multichannel pipet will be used to load the plate, then aliquot
the required volumes into Titertube
n
Do not freeze diluted samples
Table 8. Summary of recommended sample diluents and dilution factors.
Sample Type Diluent Add BSA Sample Dilution
Serum and plasma Sample diluent None Fourfold (1:4)
Culture media, with serum Culture media None User optimized
Culture media, serum-free Culture media To 0.5% final User optimized
For Adiponectin Assay
Serum and plasma Serum-based None Human (1:400)
NHP (1:1600)
For Adipsin Assay
Serum and plasma Serum-based None Human (1:400)
Note: certain sample types may require a different dilution factor.
diluent
diluent
®
micro test tubes.
Mouse (1:1600)
16
Page 19

Protease Inhibitors
In general, diabetes biomarkers are detectable in EDTA-treated plasma.
Freezing plasma immediately after preparation and keeping samples
frozen until use should provide adequate protection from degradation.
However, users may choose to add protease inhibitors as a
precautionary measure.
Note: Protease inhibitors are recommended for use with plasma samples
only, not with serum.
Either protease inhibitors may be added to samples at the time of blood
collection (see protocol in the Appendix) or blood samples may be
collected directly into a BD P800 collection tube (Becton-Dickinson,
catalog #366420 or #366421).
Note: BD P800 tubes are not designed for use with mouse samples;
therefore protease inhibitors, if required, should be added at the point of
blood collection.
Plasma
K2EDTA-treated plasma is acceptable as long as the sample is immediately
frozen upon collection. Avoid using heparin-treated plasma as it may absorb
certain soluble proteins. Avoid using hemolyzed samples as this may lead to
false positive results.
1. Draw whole blood into collection tubes containing anticoagulant.
2. If desired, add protease inhibitors (see protocol in the Appendix).
3. Invert tubes several times to mix with either the anticoagulant or the
protease inhibitors.
4. Perform centrifugation at 1,000 x g for 15 min at 4°C and transfer the
plasma to a clean polypropylene tube.
5. To completely remove platelets and precipitates, centrifuge again at
10,000 x g for 10 min at 4°C. Alternatively, filter the samples with a
0.8/0.2 μm dual filter to prevent instrument clogging.
17
Page 20

6. For assays other than adiponectin and adipsin, dilute plasma fourfold
(1:4) by adding 1 volume of sample to 3 volumes of Bio-Plex sample
diluent (for example: 40 μl sample + 120 μl sample diluent).
7. Assay samples immediately or aliquot into single-use tubes and store
at –70°C. Avoid repeated freeze/thaw cycles.
Serum
1. To prepare serum, allow blood to clot at room temperature for
30 to 45 min.
2. Perform centrifugation at 1,000 x g for 15 min at 4°C and transfer the
serum to a clean polypropylene tube.
3. To completely remove platelets and precipitates, centrifuge again at
10,000 x g for 10 min at 4°C. Alternatively, carefully filter the samples
with a 0.8/0.2 μm dual filter to prevent instrument clogging.
4. Dilute and handle samples as described in steps 6 and 7 above.
Tissue Culture Supernatant
1. Collect supernatants and centrifuge at 1,000 x g for 15 min at 4°C.
For cell lines cultured in serum-free culture media, collect samples
and add BSA as a carrier protein to a final concentration of 0.5%.
This is done to stabilize protein analytes and to prevent adsorption
to labware.
2. Transfer to a clean polypropylene tube. If cellular debris or precipitates
are present, centrifuge again at 10,000 x g for 10 min at 4°C.
3. If high levels of analyte are expected, samples can be further diluted
in culture media. Supplement serum-free media with 0.5% BSA final.
4. Assay samples immediately or aliquot and store at –70°C.
18
Page 21

Lavage, Sputum, and Other Biological Fluid Samples
Keep all samples on ice until ready for use.
1. If dilution is required, use Bio-Plex sample diluent with
0.5% BSA final.
2. Centrifugation at 10,000 x g for 10 min at 4°C may be needed to
clarify the sample.
Sample Dilution for Adiponectin and Adipsin Assays
Note: Physiological levels of adiponectin and adipsin are typically found
at high concentrations; therefore, higher sample dilutions are required to
achieve measurable concentrations within the standard curve.
1. For human adiponectin and adipsin assays, dilute serum or plasma
1:400 in serum-based diluent with two 1:20 serial dilutions
n
First dilution: 10 µl sample + 190 µl serum-based diluent
n
Second dilution: 10 µl from the first dilution + 190 µl serum-
based diluent
2. For mouse and NHP adiponectin assays, dilute serum or plasma
1:1,600 in serum-based diluent with two 1:40 serial dilutions
n
First dilution: 10 μl sample + 390 μl serum-based diluent
n
Second dilution: 10 μl from the first dilution + 390 μl serum-
based diluent
19
Page 22

6. Prepare Coupled Beads
Instructions are provided for diluting the coupled beads to a 1x
concentration. When mixing diabetes and cytokine assays, keep in mind
the stock concentrations of coupled beads as listed in Table 9.
Table 9. Stock concentration of coupled beads.
Assay Stock Concentration of Coupled Beads
Human/NHP, mouse, and rat diabetes 20x
Human and mouse cytokines (groups I, II) 10x
Rat cytokines (group I) 20x
Mouse cytokines (group III) 20X
Note: When using 10-pack reagents, ensure that only the required
volumes of coupled beads, detection antibodies, streptavidin-PE, and
buffers have been removed from the tubes or bottles. For example, transfer
a one-time volume of assay buffer sufficient to perform all steps of the
assay procedure (that is, prewetting the filter plate, diluting coupled beads,
diluting streptavidin-PE, and resuspending the beads) into a 50 ml reservoir.
1. Use Tables 10–12 to calculate the volume of coupled beads and
assay buffer needed.
2. Add the required volume of Bio-Plex assay buffer to a 15 ml
polypropylene tube.
3. Vortex the stock coupled beads at medium speed for 30 sec.
Carefully open the cap and pipet any liquid trapped in the cap back
into the tube. This is important to ensure maximum bead recovery.
Do not centrifuge the vial; doing so will cause the beads to pellet.
4. Dilute coupled beads to 1x by pipetting the required volume into the
15 ml tube. Vortex.
Each well of the assay plate requires either 2.5 µl (20x stock) or 5.0 µl
(10x stock) adjusted to a final volume of 50 µl in assay buffer.
20
Page 23

5. Protect the beads from light with aluminum foil. Equilibrate to room
temperature prior to use.
Table 10 summarizes volumes required for preparing 1x beads from a
single 20x stock. Table 11 summarizes volumes required for preparing
1x beads from a mix of two 20x stocks; volumes required for preparing
beads of one diabetes (20x) assay and one cytokine (10x) assay (for
example, human insulin and human IL-6) are listed in Table 12.
Note: To minimize volume loss, use a 200–300 μl capacity pipet to
remove beads from the stock tube. If necessary, perform the volume
transfer in two steps. Do not use a 1,000 μl capacity pipet and/or
wide bore pipet tip.
Preparing 1x coupled beads from 20x stock (includes 20% excess volume).
Table 10. Premixed panel or one singleplex assay.
# of Wells
96
20x
Beads, µl
288
Assay
Buffer, µl
5,472
Total
Volume, µl
5,760
Table 11. Mixing two singleplex assays or a premixed panel + singleplex assay.
# of Wells
96
Table 12. Preparing 1x beads from two stocks at different concentrations. Mixing human
insulin (20x) with human IL-6 (10x) is one example*.
# of Wells
96
* Due to differences in dilution factors, it is not possible to multiplex adiponectin, adipsin
(human), VCAM-1, or ICAM-1 with other diabetes or cy tokine assays.
20x beads, µl
Singleplex #1
288
20x
Beads, µl
Diabetes
288
20x beads, µl
Singleplex #2
288
10x
Beads, µl
Cytokines
576
Assay
Buffer, µl
5,184
Assay
Buffer, µl
4,896
Total
Volume, µl
5,760
Total
Volume, µl
5,760
21
Page 24

7. Run Assay
Considerations
n
Bring all assay components and samples to room temperature before use
n
Use calibrated pipets and pipet carefully, avoiding bubbles. Use new
pipet tips for every volume transfer
n
Pay close attention to vortexing, shaking, and incubation instructions.
Deviation from the protocol may result in low assay signal and
assay variability
n
Assay incubations are carried out in the dark on a shaker at
850 ± 50 rpm. Cover the plate with sealing tape and protect from
light with aluminum foil
Table 13. Summary of wash options and protocols. After each assay step, select the
appropriate Bio-Plex Pro
as summarized below.
Bio-Plex Pro or Bio-Plex Pro II Handheld Magnet or
Pro II Wash Station Wash Station Vacuum Manifold
Assay Step Magnetic Program Vacuum Program Manual Wash Steps
Add beads to plate MAG x2 VAC x2 2 x 100 μl
Sample incubation MAG x3 VAC x3 3 x 100 μl
Detection Ab incubation MAG x3 VAC x3 3 x 100 μl
SA-PE incubation MAG x3 VAC x3 3 x 100 μl
™
wash station program or perform the appropriate manual wash step
Considerations When Using a Vacuum Manifold
n
After each incubation, place the filter plate on a calibrated vacuum
apparatus and remove the liquid by vacuum filtration
n
To wash, add 100 μl wash buffer to each well and remove the liquid as
before. Ensure that all wells are exposed to the vacuum
n
Thoroughly blot the bottom of the filter plate with a clean paper towel
between each vacuum step to prevent cross contamination
n
Place the assay plate on the plastic plate holder/tray as needed
n
Before each incubation, gently cover the plate with a new sheet of
sealing tape. Avoid pressing down over the wells to prevent leaking
from the bottom
22
Page 25

Add Coupled Beads, Standards, Blanks, Samples,
and Controls
1. Cover unused wells with sealing tape.
2. Prewet the filter plate. Skip this step if using a flat bottom plate.
a) Prewet the wells with 100 µl of assay buffer and remove the liquid
by vacuum filtration. Dry the bottom of the filter plate thoroughly
by blotting on a clean paper towel.
3. Vortex the diluted (1x) coupled beads for 30 sec at medium speed.
Pour the diluted coupled beads into a reagent reservoir and transfer
50 µl to each well of the assay plate.
Tip: A multichannel pipet is highly recommended for ease of use
and efficiency.
4. Wash the plate two times with 100 µl Bio-Plex wash buffer using
the wash method of choice.
5. Gently vortex the diluted standards, blanks, samples, and controls
(if applicable) for 5 sec. Transfer 50 µl to each well of the assay plate,
changing the pipet tip after every volume transfer
6. Cover plate with a new sheet of sealing tape and protect from light
with aluminum foil. Incubate on shaker at 850 ± 50 rpm for 1 hr at
room temperature (RT).
Note: 850 rpm provides equivalent performance to previously
recommended shaker settings (1,100 rpm for 30 sec, 300 rpm for
incubation).
Note: Be consistent with this incubation time for optimal assay
performance and reproducibility.
23
Page 26

Prepare and Add Detection Antibodies
Instructions are provided for diluting the detection antibodies to a 1x
concentration. When mixing diabetes and cytokine assays, keep in mind
the stock concentrations of detection antibodies as shown below.
Table 14. Stock concentration of detection antibodies.
Stock Concentration of
Assay Detection Antibodies
Human/NHP, mouse, and rat diabetes 20x
Human and mouse cytokines (groups I, II) 10x
Rat cytokines (group I) 20x
Mouse cytokines (group III) 20X
1. While the samples are incubating, use Tables 15–17 to calculate the
volume of detection antibodies and detection antibody diluent needed.
Detection antibodies should be prepared 10 m in before use.
2. Add the required volume of Bio-Plex detection antibody diluent to a
15 ml polypropylene tube.
3. Vortex the stock detection antibodies for 15–20 sec at medium
speed, then perform a 30 sec spin to collect the entire volume at the
bottom of the tube.
4. Dilute detection antibodies to 1x by pipetting the required volume into
the 15 ml tube.
Each well of the assay requires either 1.25 μl (20x stock) or 2.5 µl
(10x stock) adjusted to a final volume of 25 μl in detection
antibody diluent.
24
Page 27

For reference, Table 15 summarizes volumes required for preparing
1x detection antibodies from a single 20x stock.
Table 16 summarizes volumes required for preparing 1x detection
antibodies from a mix of two 20x stocks; volumes required for
preparing detection antibodies of one diabetes (20x) assay and one
cytokine (10x) assay (for example, human insulin and human IL-6)
are listed in Table 17.
Preparing 1x detection antibodies from 20x stock (includes 25% excess volume).
Table 15. Premixed panel or one singleplex assay.
# of Wells
96
Table 16. Mixing two singleplex assays or a premixed panel + singleplex assay.
# of Wells
96
Table 17. Preparing 1x detection antibodies from two stocks at different concentrations.
Mixing human insulin (20x) with human IL-6 (10x) is one example.*
# of Wells
96
* Due to differences in dilution factors, it is not possible to multiplex adiponectin, adipsin
(human), VCAM-1, or ICAM-1 with other diabetes or cytokine assays.
20x Detection
Antibodies, µl
150
20x Detection
Antibodies, µl
Singleplex #1
150
20x Detection
Antibodies, µl
Diabetes
150
Detection
Antibody
Diluent, µl
2,850
20x Detection
Antibodies, µl
Singleplex #2
150
10x Detection
Antibodies, µl
Cytokines
300
Total
Volume, µl
3,000
Detection
Antibody
Diluent, µl
2,700
Detection
Antibody
Diluent, µl
2,550
Total
Volume, µl
3,000
Total
Volume, µl
3,000
5. After incubating the beads, samples, standards, and blank, slowly
remove and discard the sealing tape.
25
Page 28

6. Wash the plate three times with 100 µl wash buffer.
7. Vortex the diluted (1x) detection antibodies gently for 5 sec.
Pour into a reagent reservoir and transfer 25 μl to each well of the
assay plate using a multichannel pipet.
8. Cover plate with sealing tape and protect from light with aluminum foil.
Incubate on shaker at 850 ± 50 rpm for 30 min at room temperature.
Prepare and Add Streptavidin-PE (SA-PE)
1. While the detection antibodies are incubating, use Table 18 to
calculate the volume of SA-PE (100x) and assay buffer needed.
Streptavidin-PE should be prepared 10 min before use.
2. Add the required volume of assay buffer to a 15 ml
polypropylene tube.
3. Vortex the 100x SA-PE for 5 sec at medium speed.
Perform a 30 sec spin to collect the entire volume at the bottom
of the vial.
4. Dilute SA-PE to 1x by pipetting the required volume into the 15 ml
tube. Vortex and protect from light until ready to use.
Each well of the assay requires 0.5 µl (100x stock) adjusted to a final
volume of 50 µl in assay buffer.
Table 18 shows an example calculation.
Table 18. Preparing 1x SA-PE from 100x stock (includes 25% excess volume).
# of Wells 100x SA-PE, µl Assay Buffer, µl Total Volume, µl
96 60 5,940 6,000
26
Page 29

5. After the detection antibody incubation, slowly remove and discard
the sealing tape.
6. Wash the plate three times with 100 µl wash buffer.
7. Vortex the diluted (1x) SA-PE at medium speed for 5 sec. Pour into a
reagent reservoir and transfer 50 µl to each well using a multichannel
pipet.
8. Cover plate with sealing tape and protect from light with aluminum foil.
Incubate on shaker at 850 ± 50 rpm for 10 min at room temperature.
9. After the streptavidin-PE incubation step, slowly remove and discard
the sealing tape.
10. Wash the plate three times with 100 µl wash buffer.
11. To resuspend beads for plate reading, add 125 µl of assay buffer to
each well. Cover the plate with a new sheet of sealing tape. Shake
the plate at room temperature at 850 ± 50 rpm for 30 sec, and
slowly remove the sealing tape. Ensure that the plate cover has been
removed before placing the plate on the reader.
12. Remove the sealing tape and read the plate using the settings below.
Note: Reading at alternative PMT settings on the Bio-Plex 100,
200, or Bio-Plex 3D (for example when mixing diabetes assays with
cytokine assays) requires validation by the end user to ensure that
results meet the user’s acceptance criteria.
Table 19. Read the plate using the appropriate instrument settings.
Instrument RP1 (PMT) DD Gates Bead Events
Bio-Plex 100, 200* High 5,000 (low), 25,000 (high) 50
Bio-Plex 3D* Standard Select MagPlex beads 50
®
Bio-Plex
MAGPIX™* N/A, use default instrument settings
* Or similar Luminex-based system.
27
Page 30

8. Read Plate
Bio-Plex Manager™ software is recommended for all Bio-Plex Pro™ assay
data acquisition and analysis. Instructions for Luminex xPONENT software
are also included. For instructions using other xMAP system software
packages, contact Bio-Rad Technical Support or your regional Bio-Rad
field applications specialist.
Prepare Protocol in Bio-Plex Manager
Software v 6.0 and Higher
The protocol should be prepared in advance so that the plate is read as
soon as the experiment is complete.
A protocol file specifies the analytes used in the reading, the plate wells
to be read, sample information, the values of standards and controls, and
instrument settings.
Bio-Plex Manager software version 6.0 contains protocols for most
Bio-Plex
protocol. To create a new protocol, select File, then New from the main
menu. Locate and follow the steps under Protocol Settings.
1. Click Describe Protocol and enter information about the
2. Click Select Analytes and create a new panel. Visually confirm the
a. Click the Add Panel button
Enter a new panel name. Select Bio-Plex Pro Assay Magnetic
from the assay pull-down menu. If using Bio-Plex Manager version
5.0 or lower, select MagPlex from the assay pull-down menu.
b. Click the Add button. Enter the bead region number and name
For reference, bead regions are shown in Table 20.
®
assays. Choose from available protocols or create a new
assay (optional).
selected analytes and proceed to step 3.
in the Select Analytes toolbar.
for the first analyte. Click Add Continue to repeat for each
analyte in the assay.
28
Page 31

c. Click the Add button when the last analyte has been added and
click OK to save the new panel.
d. Highlight analytes from the Available list (left) and move to the
Selected list (right) using the Add button. To move all analytes at
once, simply click the Add All button.
e. If some of the analytes need to be removed from the Selected
list, highlight them and select Remove. If desired, it is possible to
rename the panel by clicking on Rename Panel and entering a
new panel name.
Note: Do not use preset panels found in Bio-Plex Manager
software version 5.0 or earlier as the bead regions are not
up to date.
Table 20. Bead regions for available Bio-Plex Pro diabetes assays.
Human/NHP Diabetes Mouse Diabetes Rat Diabetes
Bead Bead Bead
Analyte Region Analyte Region Analyte Region
Adiponectin 64 Adiponectin 29 Ghrelin 64
Adipsin 35 Ghrelin 64 GLP-1 62
C-Peptide 72 GIP 46 Glucagon 63
Ghrelin 26 GLP-1 62 Leptin 65
GIP 14 Glucagon 63 PAI-1 61
GL P-1 27 Insulin 66
Glucagon 15 Leptin 65
Insulin 12 PAI-1 48
Leptin 78 Resistin 30
PAI-1 61
Resistin 65
Visfatin 22
29
Page 32

Table 21. Bead regions for compatible Bio-Plex Pro cytokine assays. Assays shown have
bead regions that are non-overlapping with diabetes assays.
Human Cytokines Mouse Cytokines Rat Cytokines
Bead
Group I
Region Region Region Region Region
IL-1b 39 IL-1a 63 IL-1a 53 IL-15 42 IL-1a 21
IL-1ra 25 IL-2R
IL-2 38 IL-3 64 IL-2 36 Basic FGF 25 IL-2 22
IL-4 52 IL-12p 40 28 IL-3 18 LIF 45 IL-4 33
IL-5 33 IL-18 42 IL-4 39 M-CSF 26 IL-5 52
IL-6 19 HGF 62 IL-5 52 MIG 44 IL-6 56
IL-7 74 IFNIL-8 54 LIF 29 IL-9 33 PDGF-BB 35 IL-10 19
IL-9 77 M-CSF 67 IL-10 56 VEGF 47 IL-12p40 76
IL-10 56 MIF 35 IL-12p40 76 IL-12p70
IL-12p70 75
IL-13 51 TNFIL-15 73 TRAIL 66 IL-17 72 IL-18 20
IL-17 76 Eotaxin 74 EPO 14
Eotaxin 43 G-CSF 54 G-CSF 54
Basic FGF 44 GM-CSF 73 GM-CSF 37
G-CSF 57 IFNGM-CSF 34 KC 57 IFNIFN-
g 21 MCP-1 (MCAF) 51 M-CSF 26
IP-10 48 MIP-1
MCP-1 53 MIP-1
MIP-1
a 55 RANTES 55 MIP-3a 36
MIP-1
b 18 TNF-a 21 RANTES 55
PDGF-BB 47 TNFRANTES 37 VEGF 47
TNF-
a 36 MCP-1 51
VEGF 45
* Mouse group III cytokines have not been tested for cross reactivity with diabetes or other panels.
Bead
Group II
a 13 IL-1b 19 IL-18 20 IL-1b 28
a2 20 IL-6 38 MIP-2 27 IL-7 38
b-NGF 46 IL-12p70 78 IL-13 15
b 30 IL-13 37 IL-17 72
Bead
Group I
g 34 GRO/KC 57
a 77 MIP-1a 77
b 75 MIP-2 27
Bead
Group II
Bead
*
Group III
IL-17F 28
IL-21 14
IL-22 15
IL-23p19 61
IL-25 67
IL-27p28 43
IL-31 29
IL-33 13
MIP-3a 12
Region
Group I
Bead
78
g 34
a 43
3. Click Format Plate and format the plate according to the plate layout
created in Section 1 (Plan Plate Layout). To modify the plate layout,
follow the steps below (see Figure 6).
a. Select the Plate Formatting tab.
b. Select the standards icon
S
and drag the cursor over all
the wells that contain standards. Repeat this process for
blanks
B
, controls C, and samples X.
30
Page 33

Fig. 6. Plate formatting.
4. Click Enter Standards Info in the Protocol Settings bar.
a. Enter the highest concentration of each analyte in the top row
(labeled S1) of the table. S1 concentration information is included
on the peel-off sticker provided with each vial of standards.
b. Enter a dilution factor of 4 and click Calculate. The concentrations
for each standard point will be populated for all analytes in the table.
c. Optional: enter the lot number of the vial of standards into the
Standard Lot box and click Save.
5. Click Enter Controls Info, and for user-defined controls, select
an analyte from the dropdown menu, then enter a description and
concentration. Repeat for each additional analyte in the assay.
6. Click Enter Sample Info and enter sample information and the
appropriate dilution factor.
31
Page 34

7. Click Run Protocol and confirm that the assay settings are correct.
a. The Bio-Plex Pro diabetes assays were developed on the high
RP1 (high PMT) setting using the Bio-Plex 200 system. Protocols
using alternative PMT settings should be validated by the end user,
for example when mixing diabetes assays with cytokine assays.
b. Confirm data acquisition is set to 50 beads per region. In
Advanced Settings, confirm that the bead map is set to 100
region, the sample size is set to 50 μl, and the DD gates are
set to 5,000 (Low) and 25,000 (High). In Bio-Plex Manager
software versions 4.0, 4.1, and 4.1.1, check Override Gates
and set the DD gate values as indicated.
c. Select Start, name and save the .rbx file, and begin data
acquisition. The Run Protocol pop-up screen will appear. Click
Eject/Retract to eject the plate carrier.
Acquire Data
1. Shake the assay plate at 850 ± 50 rpm for 30 sec and visually
inspect the plate to ensure that the assay wells are filled with buffer.
Slowly remove the sealing tape and any plate cover before placing
the plate on the plate carrier.
2. Click Run Protocol — on the pop-up screen, select Load Plate and
click OK to start acquiring data.
3. Use the Wash Between Plates
to reduce the possibility of clogging the instrument.
4. If acquiring data from more than one plate, empty the waste bottle
and refill the sheath bottle after each plate (if HTF are not present).
Select Wash Between Plates and follow the instructions. Then
repeat the Prepare Protocol and Acquire Data instructions.
5. When data acquisition is complete, select Shut Down
follow the instructions.
command after every plate run
and
32
Page 35

Reacquire Data
It is possible to acquire data from a well or plate a second time using the
Rerun/Recovery mode located below Start in the Run Protocol step.
Any previous data will be overwritten.
1. Check the wells from which data will be reacquired.
2. Remove the buffer with the wash method of choice.
3. Add 100 µl assay buffer to each well. Cover the filter plate with a
new sheet of sealing tape. Shake the plate at 850 ± 50 rpm for
30 sec. Slowly remove the sealing tape before placing the plate on
the plate reader.
4. Repeat the Acquire Data steps to reacquire data. The data acquired
should be similar to those acquired initially; however, the acquisition
time will be extended because the wells have fewer beads.
Data Analysis: Removing Outliers
Outliers are identified as standard data points that do not meet accuracy
or precision requirements and should be considered invalid when
performing curve fitting. As such, they should be removed to generate a
more realistic and accurate standard curve. This may result in an extended
assay working range and allow quantitation of samples that might
otherwise be considered out of range (OOR).
In Bio-Plex Manager software version 6.0 and higher, outliers can be
automatically removed by selecting the Optimize button in the Standard
Curve window. In Bio-Plex Manager software 6.0 and earlier versions,
outliers also can be manually selected in the Report Table. Visit online Help
to learn more about the standard curve optimizer feature and how outliers
are determined.
Previous Versions of Bio-Plex Manager Software
For instructions on using previous versions of Bio-Plex manager software,
please contact Bio-Rad Technical Support.
33
Page 36

Luminex xPONENT Software
Although guidelines are provided here, consult the xPONENT software
manual for more details. Perform a system initialization with Luminex’s
calibration and performance verification kit, as directed by Luminex. Select
Batches to set up the protocol and follow the information under Settings.
Note: The instrument settings described below apply to Luminex 100/200
and FLEXMAP 3D or Bio-Plex
MAGPIX
™
reader, use the default instrument settings.
1. Select MagPlex as the bead type for magnetic beads, which
automatically sets the DD gates.
2. Volume = 50 µl.
3. Refer to Table 19 to select the appropriate PMT setting for
your instrument.
4. Plate name: 96-well plate.
5. Analysis type: Quantitative; 5PL Curve Fit.
6. Number of standards: 8.
Select Analytes to set up the panel.
1. Enter pg/ml in the Units field.
2. Enter 50 in the Count field.
3. Select the bead region and enter the analyte name.
4. Click Apply all for Units and Count.
®
3D instruments. For the Bio-Plex®
Select Stds and Ctrls.
1. Enter standard concentrations, lot number, dilution factor, and other
information as applicable.
After the assay is complete, select Results, then select Saved Batches.
34
Page 37

Troubleshooting Guide
This troubleshooting guide addresses problems that may be encountered
with Bio-Plex Pro
™
assays. If you experience any of the problems listed
below, review the possible causes and solutions provided. Poor assay
performance may also be due to the Bio-Plex
®
suspension array reader.
To eliminate this possibility, use the validation kit to assist in determining if
the array reader is functioning properly.
Possible Causes
High Inter-Assay CV
Possible Solutions
Standards were not reconstituted
consistently between assays
Incubate the reconstituted
standards for 30 min on ice. Always
be consistent with the incubation
time and temperature.
Reconstituted standards and
diluted samples were not stored
properly
Reconstituted standards and diluted
samples should be prepared on ice
as instructed. Prior to plating, the
reconstituted standards and diluted
samples should be equilibrated to
room temperature.
Bottom of filter plate not dry
Dry the bottom of the filter plate with
absorbent paper towel (preferably
lint-free) to prevent cross-well
contamination.
35
Page 38

Possible Causes
High Intra-Assay CV
Improper pipetting technique
Possible Solutions
Pipet carefully when adding
standards, samples, detection
antibodies, and streptavidin-PE,
especially when using a multichannel
pipet. Use a calibrated pipet. Change
pipet tip after every volume transfer.
Reagents and assay components
not equilibrated to room
temperature prior to pipetting
Contamination with wash buffer
during wash steps
Slow pipetting of samples and
reagents across the plate
Bio-Plex Wash Station:
insufficient washing due to
clogged pins
All reagents and assay components
should be equilibrated to room
temperature prior to pipetting.
During the wash steps, be careful
not to splash wash buffer from one
well to another. Be sure that the
wells are filtered completely and that
no residual volume remains. Ensure
that the microplate shaker setting is
not too high. Reduce the microplate
shaker speed to minimize splashing
Sample pipetting across the entire
plate should take less than 4 min.
Reagent pipetting across the entire
plate should take less than 1 min.
Clean dispensing pins with the thicker
of the 2 cleaning needles provided
with washer. Perform regular rinses to
minimize salt build up.
36
Page 39

Possible Causes
Low Bead Count
Miscalculation of bead dilution
Possible Solutions
Check your calculations and be
careful to add the correct volumes
Beads clumped in multiplex
bead stock tube
Vacuum on for too long when
aspirating buffer from wells
Filter plate not shaken enough
before incubation steps and prior
to reading
Reader is clogged
Low Signal or Poor Sensitivity
Standards reconstituted
incorrectly
Vortex for 30 sec at medium speed
before aliquoting beads.
Do not apply vacuum to the filter
plate for longer than 10 sec after the
buffer is completely drained from
each well.
Shake the filter plate at
850 ± 50 rpm for 30 sec before
incubation steps and immediately
before reading the plate.
Refer to the troubleshooting guide
in the Bio-Plex system hardware
instruction manual (bulletin
#10005042).
Follow the standard preparation
instructions carefully.
Detection antibody or
streptavidin-PE diluted incorrectly
Check your calculations and be
careful to add the correct volumes.
37
Page 40

Possible Causes
High Background Signal
Incorrect buffer was used
(for example, assay buffer used
to dilute standards)
Possible Solutions
Use standard diluent or diluent
similar to final sample matrix to dilute
standards.
Accidentally spiked blank wells
Detection antibodies or
streptavidin-PE incubated
too long
Poor Recovery
Expired Bio-Plex reagents
were used
Incorrect amounts of components
were added
Microplate shaker set to an
incorrect speed
Do not add any antigens to the
blank wells.
Follow the procedure incubation
time precisely.
Check that reagents have not
expired. Use new or nonexpired
components.
Check your calculations and be
careful to add the correct volumes.
Check the microplate shaker speed
and use the recommended setting.
Setting the speed too high may
cause splashing and contamination.
Use the recommended plate shaker.
38
Page 41

Possible Causes
Poor Recovery
Improper pipetting
technique
Impact of Sample Matrix
Negative MFI values in
samples or standards
Possible Solutions
Pipet carefully when adding
standards, samples, detection antibodies,
and streptavidin-PE, especially when using
a multichannel pipet. Use a calibrated pipet.
Change pipet tip after every volume transfer.
If samples contain little or no analyte, negative
values observed may be due to statistical
variation. If assay drift is suspected, retest
the samples by positioning them next to the
standards. If contamination of standards
is suspected, check the standard replicate
value and be careful when adding samples to
the wells. Matrix effects could also produce
negative sample values.
Bio-Plex Manager
™
software automatically
subtracts the blank (B) FI value from all other
assay wells. While this has no impact on
observed concentrations of samples within the
assay working range, it may result in a negative
FI value if the blank’s FI value is greater than
either the standard or sample value. If this is
undesirable, then assign wells as a sample (X) or
control (C) in the protocol or results file.
Poor precision in serum
and plasma sample
measurements
Check if any interfering components such as
heparin-based anticoagulant, additives, or
gel from separators were introduced into the
samples. Avoid using hemolyzed and heavily
lipemic samples. Remove visible particulate
in samples by centrifugation. Avoid multiple
freeze/thaw cycles of samples.
39
Page 42

Appendix: Protease Inhibitors
Refer to the recommended protocol below for preparing the inhibitors.
Materials
n
DDP-IV Inhibitor (Sigma Aldrich, K4264, Molecular Weight: 370.24):
Store at 4ºC
n
Aprotinin (Sigma Aldrich, A3428, 3–8 TIU/mg or 3,900–10,400 KIU/mg):
Store at 4ºC
Preparation of Protease Inhibitors Stock Solution
1. Preparation of 10 mM DDP-IV inhibitor solution
a) Weigh 18.5 mg of DDP-IV inhibitor.
b) Dissolve completely in 5 ml of 0.9% NaCl.
c) Aliquot and store at –20ºC. Avoid repeated freeze/thaw cycles.
2. Preparation of 1.3% Aprotinin
a) Weigh 100 mg Aprotinin.
b) Dissolve completely in 7.5 ml of 0.9% NaCl.
c) Aliquot and store at –20ºC. Avoid repeated freeze/thaw cycles.
Blood Sample Preparation
1. Collect whole blood in plasma collection tubes.
2. Add 10 μl of 10 mM DDP-IV inhibitor per 1 ml of whole blood for a
final concentration of 100 μM.
3. Add 10 μl of 1.3% Aprotinin per 1 ml of whole blood for a final
amount of >500 KIU per 1 ml of whole blood.
4. Invert the tubes several times to mix the protease inhibitors with blood.
40
Page 43

Plate Layout Template
41
Page 44

Calculation Worksheet
If using either a premixed panel or one singleplex assay with 20x stocks of beads
and detection antibodies, follow these directions.
Plan the plate layout and enter the number of wells to be used in the assay:_______
1
1. Determine the volume of 1x coupled beads needed.
a. Each well requires 50 µl of coupled beads (1x): _______ x 50 µl = _______ µl
b. Include 20% excess to ensure enough volume: _______ µl x 0.20 = _______ µl
c. Total volume of 1x coupled beads: _______ µl + _______ µl = _______ µl
d. Volume of 20x coupled beads required: _______ µl /20 = _______ µl
e. Volume of assay buffer required: _______ µl – _______ µl = _______
2 3 4
4 5 6
2. Determine the volume of 1x detection antibody needed.
a. Each well requires 25 µl detection antibodies (1x): _______ x 25 µl = _______ µl
b. Include 25% excess to ensure enough volume: _______ µl x 0.25 = _______ µl
c. Total volume of 1x detection antibodies: _______ µl + _______ µl = _______ µl
d. Volume of 20x detection antibodies required: _______ µl /20 = _______ µl
e. Volume of detection antibody diluent required: _____ µl – _____ µl = _____ µl
3. Determine the volume of 1x streptavidin-PE needed.
a. Each well requires 50 µl streptavidin-PE (1x): _______ x 50 µl = _______ µl
b. Include 25% excess to ensure enough volume: _______ µl x 0.25 = _______ µl
c. Total volume of 1x streptavidin-PE: ______ µl + ______ µl = ______ µl
d. Volume of 100x streptavidin-PE required: _______ µl / 100 = _______ µl
e. Volume of assay buffer required: _______ µl – _______ µl = _______ µl
10 11 12
12 13 14
1 2
2 3
4 5
1 7
7 8
7 8 9
9 10
9 10 11
1 10
10 11
12 13
42
Page 45

If mixing singleplex assays with 20x stocks of beads and detection antibodies, follow these
directions. Enter the number of wells to be used in the assay:_______
1
1. Determine the volume of 1x coupled beads needed.
a. Each well requires 50 µl coupled beads (1x): _______ x 50 µl = _______ µl
b. Include 20% excess to ensure enough volume: _______ µl x 0.20 = _______ µl
c. Total volume of 1x coupled beads: _______ µl + _______ µl = _______ µl
d. Enter the number of singleplex sets (or analytes) tubes that will be multiplexed = _______
2 3 4
1 2
2 3
5
e. Volume of 20x coupled beads required from each stock tube:
_______ µl / 20 = _______ µl
f. Total volume of combined bead stocks: _______ x _______ µl = _______ µl
g. Volume of assay buffer required: _______ µl – _______ µl = _______ µl
4 6
5 6 7
4 7 8
2. Determine the volume of 1x detection antibody needed.
a. Each well requires 25 µl detection antibodies (1x): _______ x 25 µl = _______ µl
b. Include 25% excess to ensure enough volume: _______ µl x 0.25 = _______ µl
c. Total volume of 1x detection antibodies: _______ µl + _______ µl = _______ µl
d. Enter the number of singleplex sets (or analytes) that will be multiplexed = _______
9 10 11
1 9
9 10
5
e. Volume of 20x detection antibodies required from each stock tube:
_______ µl / 20 = _______ µl
11 12
f. Total volume of combined detection antibody stock: _____ µl x _____ = _____ µl
g. Volume of detection antibody diluent required: ____ µl – ____ µl = ____µl
12 5 13
11 13 14
3. Determine the volume of 1x streptavidin-PE needed.
a. Each well requires 50 µl streptavidin-PE (1x): _______ x 50 µl = _______ µl
b. Include 25% excess to ensure enough volume: _______ µl x 0.25 = _______ µl
c. Total volume of 1x streptavidin-PE: ______ µl + ______ µl = _______ µl
d. Volume of 100x streptavidin-PE required: _______ µl / 100 = _______ µl
e. Volume of assay buffer required: _______ µl – _______ µl = _______ µl
15 16 17
17 18 19
1 15
15 16
17 18
43
Page 46

If mixing diabetes assays (20x bead and detection antibody stocks) with cytokine assays
(10x stocks), follow these directions.
Note: Refer to Table 20 for the maximum number of diabetes and cytokine assays
that may be multiplexed. Mixing across panels is not applicable to NHP diabetes assays.
Table 20. Maximum number of singleplex diabetes and cytokine analytes that may be multiplexed.
Human, mouse, and rat diabetes
Mouse cytokine (group III) analytes (20x)
Human and mouse cytokine (groups I, II) analytes (10x) 10 9 8 7 6 5
Enter the number of wells to be used in the assay:_______
1
Enter the number of diabetes tubes (either singleplex or multiplex) that will be mixed: ______
2
Enter the number of cytokine tubes (either singleplex or multiplex) that will be mixed: _______
3
0 2 4 6 8 10
1. Determine the volume of 1x diabetes and cytokine coupled beads needed.
a) Each well requires 50 µl of coupled beads (1x): _______ x 50 µl = _______ µl
b) Include 20% excess to ensure enough volume: _______ µl x 0.20 = _______ µl
c) Total volume of 1x coupled beads: _______ µl + _______ µl = _______ µl
d) Volume of 20x beads required from each diabetes tube(s): _______ µl /20 = _____ __ µl
e) Volume of 10x beads required from each cytokines tube(s): ______ μl / 10 = ______ μl
f) Total volume of diabetes bead stock required: ______ μl x _______ = _______ μl
7 2 9
g) Total volume of cytokine bead stock required: ______ μl x _______ = _______ μl
h) Total volume of combined bead stocks required: _____ μl + ______ = _____ μl
i) Volume of assay buffer required: _______ µl - _______ µl = _______
4 5 6
6 11 12
1 4
4 5
6 7
6 8
8 3 10
9 10 11
44
Page 47

2. Determine the volume of 1x diabetes and cytokine detection antibodies needed.
a) Each well requires 25 μl of detection antibodies (1x): _______ x 25 μl = ______ μl
b) Include 25% excess to ensure enough volume: _______ μl x 0.25 = ______ μl
c) Total volume of 1x detection antibodies: ______ μl + ______ μl = ______ μl
13 14 15
1 13
13 14
d) Volume of 20x detection antibodies required from each diabetes tube(s):
______ μl / 20 = ______ μl
15 16
e) Volume of 10x detection antibodies required from each cytokines tube(s):
______ μl / 10 = ______ μl
15 17
f ) Total volume of diabetes detection antibodies stock required: ____ μl x ____ =_____ μl
g) Total volume of cytokine detection antibodies stock required: ____ μl x ____ =_____ μl
16 2 18
17 3 19
h) Total volume of combined detection antibodies required:
_____ μl + ______ = _____ μl
18 19 20
i) Volume of detection antibody diluent required: ______ μl – _____ μl = _____ μl
15 20 21
3. Determine the volume of 1x streptavidin-PE needed.
a) Each well requires 50 μl of streptavidin-PE (1x): _________ x 50 μl = _________ μl
b) Include 25% excess to ensure enough volume: _______ μl x 0.25 = ______ μl
c) Total volume of 1x streptavidin-PE: ______ μl + ______ μl = ______ μl
d) Volume of 100x streptavidin-PE required: _______ μl / 100 = ______ μl
e) Volume of assay buffer required: ______ μl – ______ μl = __________ μl
15 16 17
17 18 19
1 15
15 16
17 18
45
Page 48

Safety Considerations
Eye protection and gloves are recommended when using these products.
Consult the MSDS for additional information. The Bio-Plex Pro
contain components of animal origin. This material should be handled as
if capable of transmitting infectious agents. Use universal precautions.
These components should be handled at Biosafety Level 2 containment
(U.S. government publication: Biosafety in Microbiological and Biomedical
Laboratories (CDC, 1999).
™
assays
Legal Notices
Acrodisc, Acroprep, and Supor are trademarks of Pall Corporation.
MagPlex, xMAP, xPONENT, and Luminex are trademarks of
Luminex Corporation.
The Bio-Plex suspension array system includes fluorescently labeled
microspheres and instrumentation licensed to Bio-Rad Laboratories, Inc.
by the Luminex Corporation.
46
Page 49

Ordering Information
Premixed All-in-One Multiplex Assays
Description Catalog #
Bio-Plex Pro
Bio-Plex Pro Mouse Diabetes 8-plex Assay, 1 x 96-well 171-F7001M
Bio-Plex Pro NHP Diabetes 11-plex Assay, 1 x 96-well 171-W7001M
Bio-Plex
Fast and economical custom assay service using the Bio-Plex Assay Builder,
www.bio-rad.com/bio-plex/assaybuilder, to select analytes and plate
type of interest. Assays are supplied as individual sets of coupled beads and
detection antibodies in the all-in-one kit format, ready for you to mix.
Singleplex Sets and Individual Components
A host of singleplex sets and individual assay components are available. For
more information, refer to bulletin 5507 or go to www.bio-rad.com/bio-plex.
™
Human Diabetes 10-plex Assay, 1 x 96-well 171-A7001M
®
Express Assays (You Mix)
47
Page 50

Bio-Rad
Laboratories, Inc.
Life Science
Group
Web site ww w.bio-rad.com USA 800 424 6723 Australia 61 2 9914 2800
Austria 01 877 89 01 Belgium 09 385 55 11 Brazi l 55 11 5044 5699
Canada 905 364 3435 China 86 21 6169 8500
Czech R epubl ic 420 241 430 532 De nmark 4 4 52 10 00
Finland 09 804 22 00 France 01 47 95 69 65 Ger many 08 9 31 884 0
Greece 30 210 9532 220 Hon g Kong 852 2789 3 300
Hungary 36 1 459 6100 India 91 124 4029300 Israel 03 963 6050
Italy 39 02 216091 Japan 03 6361 7000 Korea 82 2 3473 4460
Mexico 52 5 55 488 7670 The Netherlands 03 18 540666
New Zealand 64 9 415 2280 No rway 23 38 41 30
Poland 48 22 331 99 99 Portugal 351 21 472 7700
Russia 7 495 721 14 04 Singapore 65 6415 3188
South Africa 27 861 246 723 Spain 3 4 91 590 5200
Sweden 08 555 1270 0 Switzerland 026 674 55 05
Taiwan 886 2 2578 7189 Thailand 800 88 22 88
United Kingdom 020 8328 200 0
10-0021 0113 Sig 121210010747 Rev D
 Loading...
Loading...