Page 1

[ CARE AND USE MANUAL ]
XSELECT CSH130 C18, 3.5 µm AND 5 µm COLUMNS
CONTENTS
I. GETTING STARTED
a. Column Installation
b. Column Equilibration
c. Initial Column Efficiency Determination
II. COLUMN USE
a. Guard Columns
b. Sample Preparation
c. Operating pH Limits
d. Solvents
e. Pressure
f. Temperature
III. SCALING UP/DOWN ISOCRATIC METHODS
IV. TROUBLESHOOTING
V. COLUMN CLEANING, REGENERATING AND STORAGE
a. Cleaning and Regeneration
b. Storage
Thank you for choosing a Waters XSelect™ CSH™ HPLC column. XSelect
CSH130 C18, 3.5 µm and 5 µm Columns feature Waters Charged Surface
Hybrid (CSH) Technology which provides excellent peak shape, high
efficiency and loading capacity for basic compounds when using acidic,
low ionic strength mobile phases. This same particle technology is used
in the ACQUITY UPLC CSH130 C18, 1.7 µm and 2.5 µm XP family of
HPLC columns, thus enabling seamless transferability between HPLC and
UPLC® system platforms. The XSelect CSH130 C18 packing materials
were designed and are manufactured in a cGMP, ISO 9001 certified
manufacturing facility using ultra pure reagents. Each batch of XSelect
CSH130 C18 material is tested chromatographically with acidic, basic
and neutral analytes as part of qualification for use in peptide mapping.
The results are held to narrow specification ranges to assure excellent,
reproducible performance. XSelect CSH130 C18 batches are also QC tested
with a gradient separation of a tryptic digest of cytochrome c using 0.1%
formic acid containing eluents. Finally, every shipped column is individually
tested for packed bed efficiency and a Performance Chromatogram and
Certificate of Batch Analysis is available upon request.
VI. CONNECTING THE COLUMN TO THE HPLC
a. Column Connectors and System Tubing Considerations
b. Measuring System Bandspreading Volume
c. Measuring Gradient Delay Volume (or Dwell Volume)
VII. ADDITIONAL INFORMATION
a. Use of Narrow-Bore (2.1 mm i.d.)
b. Impact of Bandspreading Volume on 2.1 mm i.d. Column Performance
c. Non-Optimized vs. Optimized LC/MS/MS System: System
Modification Recommendations
XSelect CSH130 C18 Columns 1
Page 2

[ CARE AND USE MANUAL ]
I. GETTING STARTED
Each XSelect CSH130 C18 Columns comes with a Certificate of Acceptance
and a Performance Test Chromatogram. The Certificate of Acceptance
is specific to each batch of packing material contained in the Peptide
Separation Technology column and includes the batch number, analysis
of unbonded particles, analysis of bonded particles, and chromatographic
results and conditions. The Performance Test Chromatogram is specific to
each individual column and contains the information: batch number, column
serial number, USP plate count, USP tailing factor, retention factor, and
chromatographic conditions. These data should be stored for future reference.
a. Column Installation
Note: The flow rates given in the procedure below are for a typical 5 µm
packing in a 4.6 mm i.d. column. Scale the flow rate up or down accordingly
based upon the column i.d., length, particle size and backpressure of the
Peptide Separation Technology column being installed. See Scaling Up/Down
Isocratic Separations section for calculating flow rates when changing column
i.d and/or length. See Connecting the Column to the HPLC for a more detailed
discussion on HPLC connections.
1. Purge the pumping system of any buffer-containing mobile
phases and connect the inlet end of the column to the injector
outlet. An arrow on the column identification label indicates the
correct direction of solvent flow.
2. Flush column with 100% organic mobile phase (methanol or
acetonitrile) by setting the pump flow rate to 0.1 mL/min. and
increase the flow rate to 1 mL/min over 5 minutes.
b. Column Equilibration
Peptide Separation Technology columns are shipped in 100% acetonitrile.
It is important to ensure mobile phase compatibility before changing to a
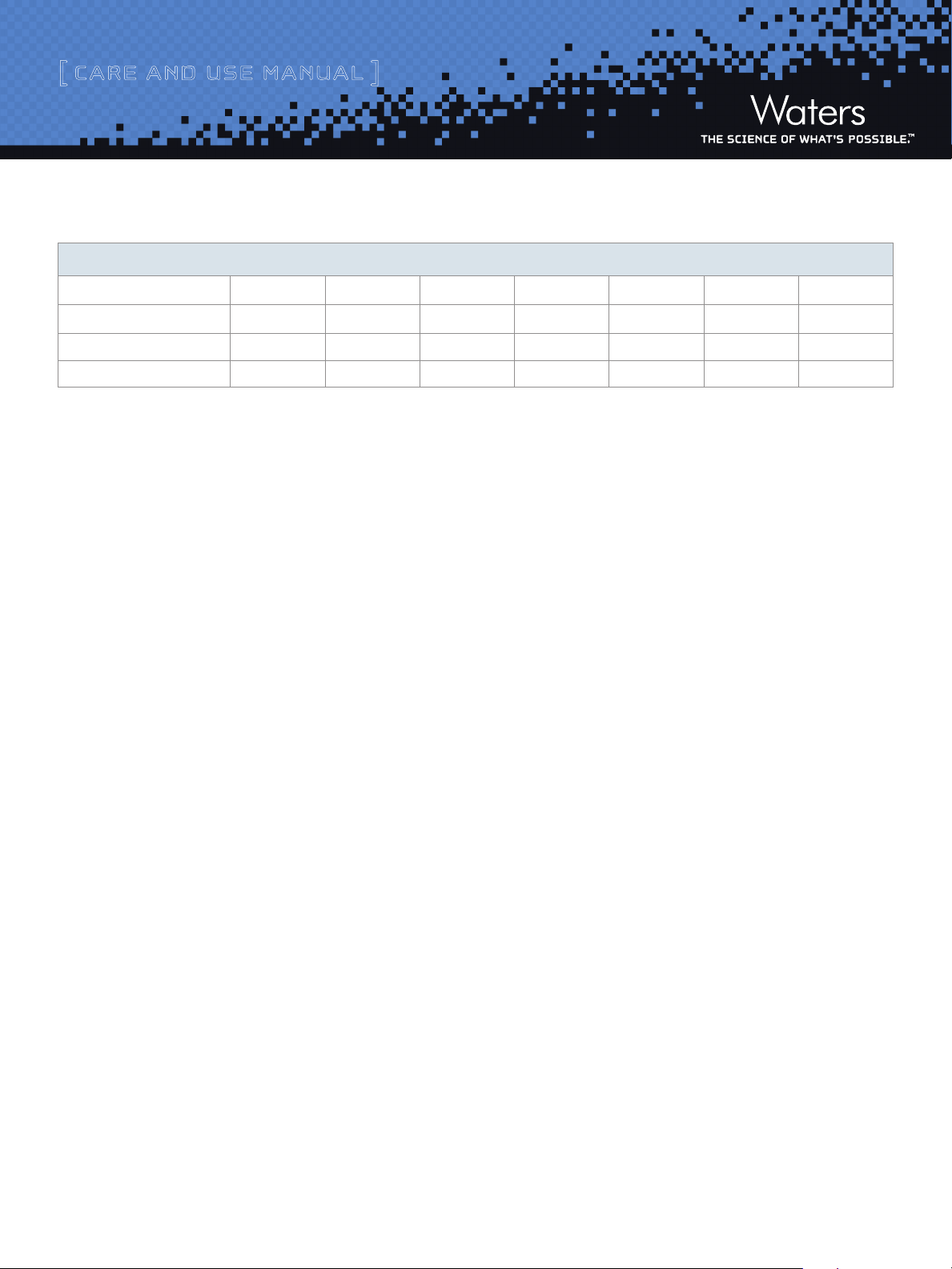
different mobile phase system.Equilibrate the column with a minimum of
10 column volumes of the mobile phase to be used (refer to Table 1 for a
listing of empty column volumes).
To avoid precipitating out mobile phase buffers on your column or in your
system, flush the column with five column volumes of a water/organic
solvent mixture, using the same or lower solvent content as in the desired
buffered mobile phase. (For example, flush the column and HPLC system with
60% methanol in water prior to introducing 60% methanol/40% buffer
mobile phase).
c. Initial Column Efficiency Determination
1. Perform an efficiency test on the column before using it in the
desired application. Waters recommends using a suitable solute
mixture, as found in the “Performance Test Chromatogram,” to
analyze the column upon receipt.
2. Determine the number of theoretical plates (N) and use this
value for periodic comparisons.
3. Repeat the test at predetermined intervals to track column
performance over time. Slight variations may be obtained on
two different HPLC systems due to the quality of the connections,
operating environment, system electronics, reagent quality,
column condition and operator technique.
3. When the mobile phase is flowing freely from the column outlet,
stop the flow and attach the column outlet to the detector. This
prevents entry of air into the detection system and gives more
rapid baseline equilibration.
4. Gradually increase the flow rate as described in step 2.
5. Once a steady backpressure and baseline have been achieved,
proceed to the next section.
Note: If mobile phase additives are present in low concentrations (e.g.,
ion-pairing reagents), 100 to 200 column volumes may be required for
complete equilibration. In addition, mobile phases that contain formate
(e.g., ammonium formate, formic acid, etc.) may also require longer initial
column equilibration times.
XSelect CSH130 C18 Columns 2
Page 3
[ CARE AND USE MANUAL ]
Table 1: Empty Column Volumes in mL (multiply by 10 for flush solvent volumes)
Column internal diameter (mm)
Column Length (mm) 1.0 2.1 4.6 10 19 30 50
50 0.04 0.17 0.83 3.9 14 35 98
100 0.08 0.35 1.7 7.8 28 70 196
150 0.12 0.52 2.5 12 42 106 294
II. COLUMN USE
To ensure the continued high performance of XSelect CSH130 C18 Columns,
follow these guidelines:
a. Guard Columns
Use a Waters guard column of matching chemistry and particle size between
the injector and main column. It is important to use a high performance
matching guard column to protect the main column while not compromising
or changing the analytical resolution.
Guard columns need to be replaced at regular intervals as determined by
sample contamination. When system backpressure steadily increases above
a set pressure limit, it is usually an indication that the guard column
should be replaced. A sudden appearance of split peaks is also indicative
of a need to replace the guard column.
b. Sample Preparation
1. Sample impurities often contribute to column contamination.
One option to avoid this is to use Waters Oasis® solid-phase
extraction cartridges/columns or Sep-Pak® cartridges of the
appropriate chemistry to clean up the sample before analysis.
4. Filter sample with 0.2 µm filters to remove particulates. If the
sample is dissolved in a solvent that contains an organic modifier
(e.g., acetonitrile, methanol, etc.) ensure that the filter material
does not dissolve in the solvent. Contact the filter manufacturer
with solvent compatibility questions. Alternatively, centrifugation
for 20 minutes at 8,000 rpm, followed by the transfer of the
supernatant liquid to an appropriate vial, could be considered.
c. Operating pH Limits
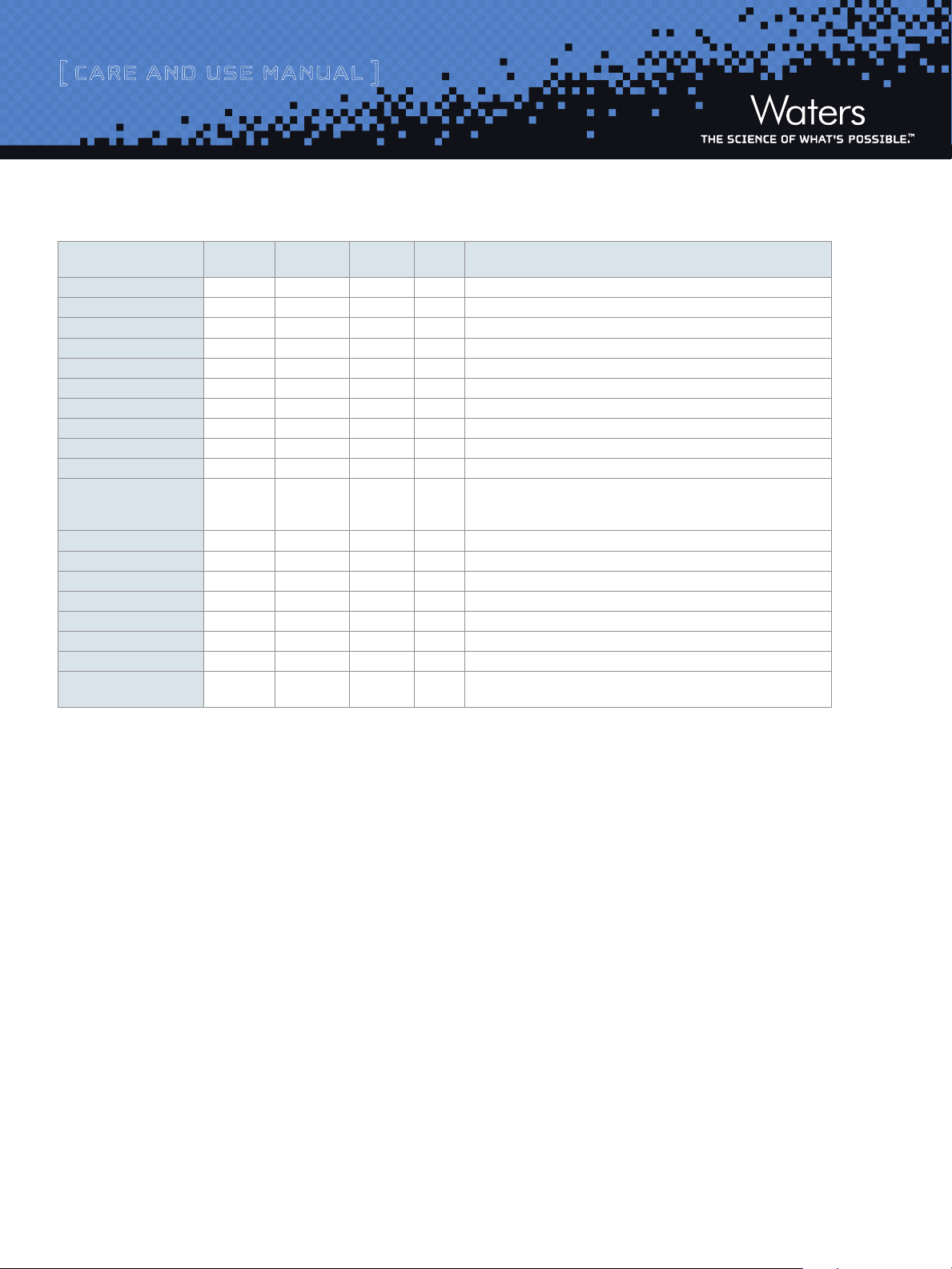
The recommended operating pH range for XSelect CSH130 C18, 3.5 µm
and 5 µm columns are 1 to 11. A listing of commonly used buffers and
additives is given in Table 2. Additionally, the column lifetime will vary
depending upon the operating temperature, the type and concentration of
buffer used.
2. It is preferable to prepare the sample in the operating mobile
phase or a mobile phase that is weaker (less organic modifier)
than the mobile phase for the best peak shape and sensitivity.
3. If the sample is not dissolved in the mobile phase, ensure that
the sample, solvent and mobile phases are miscible in order to
avoid sample and/or buffer precipitation.
XSelect CSH130 C18 Columns 3
Page 4
[ CARE AND USE MANUAL ]
Table 2: Buffer Recommendations for Using XSelect CSH130 C18 Columns from pH 1 to 11
Additive/Buffer pKa Buffer range Volatility
TFA 0.3 Volatile Yes Ion pair additive, can suppress MS signal, used in the 0.02-0.1% range.
Acetic Acid 4.76 Volatile Yes Maximum buffering obtained when used with ammonium acetate salt. Used in 0.1-1.0% range.
Formic Acid 3.75 Volatile Yes Maximum buffering obtained when used with ammonium formate salt. Used in 0.1-1.0% range.
Acetate (NH4CH2COOH) 4.76 3.76 – 5.76 Volatile Yes Used in the 1-10 mM range. Note that sodium or potassium salts are not volatile.
Formate (NH4COOH) 3.75 2.75 – 4.75 Volatile Yes Used in the 1-10 mM range. Note that sodium or potassium salts are not volatile.
Phosphate 1 2.15 1.15 – 3.15 Non-volatile No Traditional low pH buffer, good UV transparency.
Phosphate 2 7.2 6.20 – 8.20 Non-volatile No Above pH 7, reduce temperature/concentration and use a guard column to maximize lifetime.
Phosphate 3 12.3 11.3 - 13.3 Non-volatile No Above pH 7, reduce temperature/concentration and use a guard column to maximize lifetime.
4-Methylmorpholine ~8.4 7.4 – 9.4 Volatile Yes Generally used at 10 mM or less.
Ammonia (NH4OH) 9.2 8.2 – 10.2 Volatile Yes Keep concentration below 10 mM and temperatures below 30 ˚C.
Ammonium Bicarbonate 10.3 (HCO
Ammonium (Acetate) 9.2 8.2 – 10.2 Volatile Yes Used in the 1-10 mM range.
Ammonium (Formate) 9.2 8.2 – 10.2 Volatile Yes Used in the 1-10 mM range.
Borate 9.2 8.2 – 10.2 Non-Volatile No Reduce temperature/concentration and use a guard column to maximize lifetime.
CAPSO 9.7 8.7 – 10.7 Non-Volatile No Zwitterionic buffer, compatible with acetonitrile, used in the 1-10 mM range. Low odor.
Glycine 2.4, 9.8 8.8 – 10.8 Non-Volatile No Zwitterionic buffer, can give longer lifetimes than borate buffer.
1-Methylpiperidine 10.2 9.3 – 11.3 Volatile Yes Used in the 1-10 mM range.
CAPS 10.4 9.5 – 11.5 Non-Volatile No Zwitterionic buffer, compatible with acetonitrile, used in the 1-10 mM range. Low odor.
Triethylamine
(as acetate salt)
-
)
8.2 – 11.3 Volatile Yes Used in the 5-10 mM range (for MS work keep source >150 ˚C ). Adjust pH with
3
+
9.2 (NH
)
4
10.7 9.7 – 11.7 Volatile Yes Used in the 0.1-1.0% range. Volatile only when titrated with acetic acid (not hydrochloric or phosphoric).
(±1 pH unit)
Used for
Mass Spec
Comments
ammonium hydroxide or acetic acid. Good buffering capacity at pH 10
Note: use ammonium bicarbonate (NH4HCO3), not ammonium carbonate ((NH4)2CO3).
Used as ion-pair for DNA analysis at pH 7-9
d. Solvents
To maintain maximum column performance, use high quality chromatography
grade solvents. Filter all aqueous buffers prior to use. Pall Gelman Laboratory
Acrodisc® filters are recommended. Solvents containing suspended particulate
materials will generally clog the outside surface of the inlet distribution frit of
the column. This will result in higher operating pressure and poor performance.
e. Pressure
XSelect CSH130 C18 columns can tolerate pressures of up to 6,000 psi
(400 bar or 40 Mpa) although pressures greater than 4,000 – 5,000 psi
should be avoided in order to maximize column and system lifetimes.
f. Temperature
Temperatures up to 80 ˚C are recommended for operating XSelect CSH130
C18 columns in order to enhance selectivity, lower solvent viscosity and
increase mass transfer rates. However, any temperature above ambient will
have a negative effect on lifetime which will vary depending on the pH and
buffer conditions used.
XSelect CSH130 C18 Columns 4
Page 5

[ CARE AND USE MANUAL ]
III. SCALING UP/DOWN ISOCRATIC METHODS
The following formulas will allow scale up or scale down, while maintaining
the same linear velocity, and provide new sample loading values:
If column i.d. and length are altered:
F
= F1 (r2/r1)2
2
Load2 = Load1 (r2/r1)2 (L2/L1)
Injection volume2 = Injection volume1(r2/r1)2 (L2/L1)
Where: r = Radius of the column
F = Flow rate
L = Length of column
1 = Original, or reference column
2 = New column
IV. TROUBLESHOOTING
Changes in retention time, resolution, or backpressure are often due to
column contamination. See the Column Cleaning, Regeneration and Storage
section of this Care and Use Manual. Information on column troubleshooting
problems may be found in HPLC Columns Theory, Technology and Practice,
U.D. Neue, (Wiley-VCH, 1997), the Waters HPLC Troubleshooting Guide
(Literature code # 720000181EN) or visit the Waters Corporation website
for information on seminars (www.waters.com).
VI. COLUMN CLEANING, REGENERATION AND STORAGE
a. Cleaning and Regeneration
Changes in peak shape, peak splitting, shoulders on the peak, shifts in
retention, change in resolution or increasing backpressure may indicate
contamination of the column. Flushing with a neat organic solvent,
taking care not to precipitate buffers, is usually sufficient to remove the
contaminant. If the flushing procedure does not solve the problem, purge
the column using the following cleaning and regeneration procedures.
4.6 x 250 mm column) listed in Table 3. Increasing mobile phase temperature
to 35-55 ˚C increases cleaning efficiency. If the column performance is
poor after cleaning and regeneration, call your local Waters office for
additional support.
Table 3: Cleaning and Regeneration Sequence or Options
Polar Samples Proteinaceous Samples
1. water Option 1: Inject repeated 100 µL aliquots of dimethylsulfoxide
2. methanol Option 2: gradient of 10% to 90% B where:
3. isopropanol
(DMSO) using a reduced flow rate delivering 50% Eluent A
and 50% Eluent B
A = 0.1% trifluoroacetic acid (TFA) in water
B = 0.1% trifluoroacetic acid (TFA) in acetonitrile (CH
Option 3: Flush column with 7M guanidine hydrochloride, or
7M urea
CN)
3
Note: To avoid potentially damaging precipitation within your column
(e.g., if your separation eluent contains phosphate buffer), be certain to
flush column with 5 to 10 column volumes of water BEFORE using suggested
organic eluent column wash procedures.
b. Storage
For periods longer than four days at room temperature, store the column
in 100% acetonitrile. Immediately after use with elevated temperatures
and/or at pH extremes, store in 100% acetonitrile for the best column
lifetime. Do not store columns in highly aqueous (<20% organic) mobile
phases, as this may promote bacterial growth. If the mobile phase con-
tained a buffer salt, flush the column with 10 column volumes of HPLC
grade water (see Table 1 for common column volumes) and replace with
100% acetonitrile for storage. Failure to perform this intermediate step
could result in precipitation of the buffer salt in the column or system
when 100% acetonitrile is introduced. Completely seal column to avoid
evaporation and drying out of the bed.
Note: If a column has been run with a mobile phase that contains formate
(e.g., ammonium formate, formic acid, etc.) and is then flushed with 100%
acetonitrile, slightly longer equilibration times may be necessary when the
column is re-installed and run again with a formate-containing mobile phase.
Use the cleaning routine that matches the properties of the samples and/or
what you believe is contaminating the column (see Table 3 ). Flush columns
with 20 column volumes each of HPLC-grade solvents (e.g., 80 mL total for
XSelect CSH130 C18 Columns 5
Page 6

[ CARE AND USE MANUAL ]
V. CONNECTING THE COLUMN TO THE HPLC
a. Column Connectors and System Tubing Considerations
Tools needed:
• 3/8 inch wrench
• 5/16 inch wrench
Handle the column with care. Do not drop or hit the column on a hard surface
as it may disturb the bed and affect its performance.
1. Correct connection of 1/16 inch outer diameter stainless steel
tubing leading to and from the column is essential for high-
quality chromatographic results.
2. When using standard stainless steel compression screw fittings, it
is important to ensure proper fit of the 1/16 inch outer diameter
stainless steel tubing. When tightening or loosening the compres-
sion screw, place a 5/16 inch wrench on the compression screw
and a 3/8 inch wrench on the hex head of the column endfitting.
Note: If one of the wrenches is placed on the column tube flat during this
process, the endfitting will be loosened and leak.
3. If a leak occurs between the stainless steel compression screw
fitting and the column endfitting, a new compression screw
fitting, tubing and ferrule must be assembled.
Due to the absence of an industry standard, various column manufacturers
have employed different types of chromatographic column connectors. The
chromatographic separation can be negatively affected if the style of the
column endfittings does not match the existing tubing ferrule settings. This
section explains the differences between Waters style and Parker style ferrules
and endfittings (Figure 1). Each endfitting style varies in the required length
of the tubing protruding from the ferrule. The XBridge column is equipped
with Waters style endfittings that require a 0.130 inch ferrule depth. If a non-
Waters style column is presently being used, it is critical that ferrule depth be
reset for optimal performance prior to installing a XBridge column.
In a proper tubing/column connection (Figure 2), the tubing touches the bottom
of the column endfitting, with no void between them.
Figure 2: Proper Tubing/Column Connection
The presence of a void in the
flow stream reduces column
performance. This can occur if a
Parker ferrule is connected to a
Waters style endfitting (Figure 3).
Note: A void appears if tubing with a Parker ferrule is connected to a Waters
style column.
Figure 3: Parker Ferrule in a Waters Style Endfitting
4. An arrow on the column identification label indicates correct
direction of solvent flow.
Correct connection of 1/16 inch outer diameter stainless steel tubing lead-
ing to and from the column is essential for high-quality chromatographic
results. To obtain a void-free connection, the tubing must touch the bottom
of the column endfitting. It is important to realize that extra column peak
broadening due to voids can destroy an otherwise successful separation. The
choice of appropriate column connectors and system tubing is discussed in
detail below.
Figure 1: Waters and Parker Ferrule Types
Waters Ferrule Setting
.130” .090”
XSelect CSH130 C18 Columns 6
Parker Ferrule Setting
There is only one way to fix this problem: Cut the end of the tubing with the
ferrule, place a new ferrule on the tubing and make a new connection. Before
tightening the screw, make sure
that the tubing bottoms out in the
endfitting of the column.
Void
Conversely, if tubing with a Waters ferrule is connected to a column with
Parker style endfitting, the end of the tubing will bottom out before the
ferrule reaches its proper sealing position. This will leave a gap and create
a leak (Figure 4).
Note: The connection leaks if a Waters ferrule is connected to a column with
a Parker style endfitting.
Page 7

[ CARE AND USE MANUAL ]
Figure 4: Waters Ferrule in a Parker Style Endfitting
Gap
There are two ways to fix the problem:
1. Tighten the screw a bit more. The ferrule moves forward, and
reaches the sealing surface. Do not overtighten since this may
end in breaking the screw.
2. Cut the tubing, replace the ferrule and make a new connection.
Alternatively, replace the conventional compression screw fitting with
an all-in-one PEEK fitting (Waters Part Number PSL613315) that allows
resetting of the ferrule depth. Another approach is to use a Keystone, Inc.
SLIPFREE® connector to always ensure the correct fit. The fingertight SLIPFREE
connectors automatically adjust to fit all compression screw type fittings
without the use of tools (Figure 5).
Figure 5: Single and Double SLIPFREE Connectors
Table 5: Waters Part Numbers for SLIPFREE Connectors
SLIPFREE Type Tubing Internal Diameter
Tubing Length 0.005” 0.010” 0.020”
Single 6 cm PSL 618000 PSL 618006 PSL 618012
Single 10 cm PSL 618002 PSL 618008 PSL 618014
Single 20 cm PSL 618004 PSL 618010 PSL 618016
Double 6 cm PSL 618001 PSL 618007 PSL 618013
Double 10 cm PSL 618003 PSL 618009 PSL 618015
Double 20 cm PSL 618005 PSL 618001 PSL 618017
Band Spreading Minimization
Figure 6 shows the influence of tubing internal diameter on system band
spreading and peak shape. As can be seen, the larger tubing diameter causes
excessive peak broadening and lower sensitivity.
Figure 6: Effect of Connecting Tubing on System
0.005 inches
0.020 inches
0.040 inches
SLIPFREE Connector Features:
• Tubing pushed into endfitting, thereby guaranteeing
a void-free connection
• Connector(s) come(s) installed on tubing
• Various tubing IDs and lengths available
• Fingertight to 10,000 psi – Never needs wrenches
• Readjusts to all column endfittings
• Compatible with all commercially available endfittings
• Unique design separates tube-holding function
from sealing function
XSelect CSH130 C18 Columns 7
Diluted/Distorted Sample Band
b. Measuring System Bandspreading Volume and System
Variance
This test should be performed on an HPLC system with a single wavelength
UV detector (not a Photodiode Array [PDA]).
1. Disconnect column from system and replace with a zero dead
volume union.
2. Set flow rate to 1 mL/min.
3. Dilute a test mix in mobile phase to give a detector sensitivity
of 0.5 - 1.0 AUFS (system start up test mix can be used which
contains uracil, ethyl and propyl parabens; Waters Part Number
WAT034544).
4. Inject 2 to 5 µL of this solution.
Page 8

[ CARE AND USE MANUAL ]
1.0
5. Measure the peak width at 4.4% of peak height (5-sigma method):
5-sigma Bandspreading (µL) = Peak Width (min) x Flow
Rate (mL/min) x (1000 µL/1 mL)
System Variance (µL2) = (5-sigma bandspreading)2 / 25
Figure 7: Determination of System Bandspreading Volume Using
5-Sigma Method
System Volume
5
4.4 %h
In
a typical HPLC system, the Bandspreading Volume should be no greater than
100 µL ± 30 µL (or Variance of 400 µL ± 36 µL).
In a microbore (2.1mm i.d.) system, the Bandspreading Volume should be no
greater than 20 to 40 µL (or Variance no greater than 16 µL to 64 µL).
c. Measuring Gradient Delay Volume (or Dwell Volume)
Figure 8: Determination of Gradient Delay Volume
0.8
0.6
Au
0.4
0.2
0.0
1/2 Vertical
Distance
Time
t
1/2
5. Program a 0-100% B linear gradient in 10 min at 2 mL/min
(the exact conditions are not critical; just make sure the gradient
volume is at least 20 mL) with a hold at 100% B.
6. Determine the dwell time by first locating the time at the
midpoint of the formed gradient (t1/2) (half the vertical distance
between the initial and final isocratic segments as shown in
Figure 8).
7. Subtract half the gradient time (1/2 tg) (10 min/2 = 5 min in
this example) from the gradient midpoint (t1/2) to obtain the
dwell time (tD).
8. Convert the dwell time (tD) to the dwell volume (VD) by
multiplying by the flow rate (F).
For successful gradient-method transfers the gradient delay volumes should
be measured using the same method on both HPLC systems. The procedure
below describes a method for determining the gradient delay volumes.
1. Replace the column with a zero dead volume union.
2. Prepare mobile phase A (pure solvent, such as methanol) and
mobile phase B (mobile phase A with a UV absorbing sample,
such as (v/v) 0.1% acetone in methanol).
3. Equilibrate the system with mobile phase A until a stable
baseline is achieved.
4. Set the detector wavelength to the absorbance maximum of the
probe (265 nm for acetone).
XSelect CSH130 C18 Columns 8
Dwell Volume VD = (t
—1/2 tg) x F
1/2
For fast gradient methods, the gradient delay volume (or dwell volume)
should be less than 1 mL. If the gradient delay volume is greater than 1 mL,
see System Modification Recommendations section on how to reduce
system volume.
Page 9

[ CARE AND USE MANUAL ]
VII. ADDITIONAL INFORMATION
a. Use of Narrow-Bore Columns
This section describes how to minimize extra column effects and provides
guidelines on maximizing the performance of a narrow-bore column in an
HPLC system. A 2.1 mm i.d. column requires modifications to the HPLC system
in order to eliminate excessive system bandspreading volume. Without proper
system modifications, excessive system bandspreading volume causes peak
broadening and has a large impact on peak width as peak volume decreases.
b. Impact of Bandspreading Volume on 2.1 mm i.d. Column
Performance
System with 70 µL bandspreading: 10,000 plates
System with 130 µL bandspreading: 8,000 plates (same column)
Note: Flow splitters after the column will introduce additional band-
spreading.
System optimization, especially in a system that contains a flow splitter, can
have dramatic effects on sensitivity and resolution. Optimization includes
using correct ferrule depths and minimizing tubing inner diameters and
lengths. An example is given in Figure 9 where system optimization resulted
in a doubling of sensitivity and resolution of the metabolite in an
LC/MS/MS system.
c. Non-Optimized vs. Optimized LC/MS/MS System: System
Modification Recommendations
1. Use a microbore detector flow cell with 2.1 mm i.d. columns.
Note: Detector sensitivity is reduced with the shorter flow cell path length in
order to achieve lower bandspreading volume.
2. Minimize injector sample loop volume.
3. Use 0.009 inch (0.25 mm) tubing for rest of connections in
standard systems and 0.005 inch (0.12 mm) tubing for
narrowbore (2.1 mm i.d.) systems.
4. Use perfect (pre-cut) connections (with a variable depth inlet if
using columns from different suppliers).
5. Detector time constants should be shortened to less than
0.2 seconds.
Figure 9: Non-Optimized vs. Optimized LC/MS/MS System
7.00 7.50
Non-optimized LC/MS/MS System Optimized System
8.00
7.00 7.50 8.00
XSelect CSH130 C18 Columns 9
Page 10

[ CARE AND USE MANUAL ]
Figure 10: Representative Test Chromatography and Conditions for Separation of a Cytochrome c Tryptic Digest
1.20
1.10
1.00
0.90
0.80
0.70
0.60
0.50
0.40
0.30
0.20
0.10
0.00
2 345678910 11 12 Min.
Peak Identification
T1 N-AcGDVEK
T13-T14 KYIPGTK
T14 YIPGTK
T4 IFVQK
T9-T10 KTGQAPGFSYTDANK
T10 TGQAPGFSYTDANK
T8 TGPNLHGLFGR
T15 MIFAGIK
T19C EDLIAY
T19 EDLIAYLK
T12-T13 GITWGEETLMEYLENPKK
T12 GITWGEETLMEYLENPK
T5 CAQCHTVEK (heme attached)
Retention Time (min) Result
T19C 6.69
Retention Time Difference (min) Result
T4 and T14 0.312
T10 and T9-T10 0.780
T19 and T19C 0.461
T12 and T12-T13 0.290
Chromatographic Conditions
Column: 2.1 mm x 50 mm
Sample: Tryptic digest of bovine cytochrome c (P/N 186006371)
Reconstituted w/ 200µL of MP A, 5.0µL injection
Flow rate: 0.2 mL/min
Temperature: 40°C
Mobile Phase A: 0.1% formic acid in 100% waterMPA
Mobile Phase B: 0.085% formic acid in 75% acetonitrileMPB
Gradient: 1 to 21%B in 5 min; 21 to 31.4% B in 2.5 min; 31.4 to
95% B in 2.5 min; 95% B for 1.5min; 1%B for 2.5 min
Detection: UV 214 nm
XSelect CSH130 C18 Columns 10
Page 11

[ CARE AND USE MANUAL ]
Sales Offices:
Austria and European Export
(Central South Eastern Europe,
CIS and Middle East) 431 877 18 07
Australia 2 9933 1777
Belgium 32 2 726 1000
Brazil 55 11 5094 3788
Canada 800 252 4752
China 8621 6495 6999
CIS/Russia +7 495 3367000
Czech Republic 42 02 617 11384
Denmark 45 46 59 8080
Finland +358 9 5659 6288
France (33) 1 30 48 72 00
Germany 49 6196 400600
Hong Kong 852 29 64 1800
Hungary 36 1 350 5086
India and India Subcontinent
91 80 2 837 1900
Ireland 353 1 448 1500
Italy 39 02 274 211
Japan (81) 3 3471 7191
Korea (82) 2 820 2700
Mexico 5255 5200 1860
The Netherlands +31 (0)76-50 87 200
Norway 47 63 84 60 50
Poland (48) 22 833 4400
Puerto Rico 787 747 8445
Singapore 65 6273 1221
Spain 34 93 600 93 00
Sweden 46 8 555 11500
Switzerland 41 62 889 2030
Taiwan 886 2 2543 1898
United Kingdom 44 208 238 6100
©2013 Waters Corporation. Waters, The Science of W hat’s Possible, Oasis, Sep-Pak, CSH, and XSelect, are trademarks of Waters
Corporation. Acrodisc is a trademark of Pall Life Sciences.
SLI PFR EE is a trademark of Keystone, Inc.
January 2013 720004585EN Rev A IH-PDF
XSelect CSH130 C18 Columns 11
Waters Corporation
34 Maple Street
Milford, MA 01757 U.S.A.
T: 1 508 478 2000
F: 1 508 872 1990
www.waters.com
 Loading...
Loading...