
IntelliVue TRx/TRx
+
Transceivers
for the Philips IntelliVue Telemetry System with
Smart-Hopping Technology
Notice
Operation of this equipment in the United States requires the prior coordindation with a frequency coordinator designated by the Federal Communications
Commission (FCC) for the Wireless Medical Telemetry Service.
Instructions for Use
Part Number: M4841-91001
Printed in the U.S.A. November 2004
First Edition

Printing History
Notice Equipment specifications are subject to alteration without notice. All changes
will be in compliance with regulations governing manufacture of medical
equipment.
Printed in the USA.
Document number: M4841-91001
© Copyright 2004 Koninklijke Philips Electronics N.V. All Rights Reserved.
OxiCliq and OxiMax are registered trademarks of Nellcor® Incorporated.
Printing History
New editions of this document will incorporate all material updated since the
previous edition. Update packages can be issued between editions and contain
replacement and additional pages to be merged by a revision date at the bottom
of the page. Note that pages which are rearranged due to changes on a previous
page are not considered revised.
The documentation printing date and part number indicate its current edition.
The printing date changes when a new edition is printed. (Minor corrections
and updates which are incorporated at reprint do not cause the date to change.)
The document part number changes when extensive technical changes are
incorporated.
First Edition ...............................................................................November 2004
Philips IntelliVue Telemetry System with Smart Hopping Technology is
compatible with:
Philips Information Center, software revision F.00
Philips TeleMon Companion Monitor, #A02/A03
ii

About this Book
This book contains operating instructions for use of the IntelliVue TRx and
TRx
Smart-hopping Technology. It also includes operational information for the
Telemetry functions of the IntelliVue Information Center. The intended
audience is the clinician who uses and/or teaches others to use the equipment
in a healthcare environment. For operating information on other functionality
of the Information Center, see IntelliVue Information Center Instructions for
Use. For preventive maintenance, repair, and test methods for verification of
device performance, refer to the Philips IntelliVue Telemetry System Service
Kit.
This book does not address the Philips M2601B Transmitter or the M2600B
Philips Telemetry System. For information on those products, refer to the
manual Philips Telemetry System Instructions for Use.
Note—Use this product in conjunction with Philips IntelliVue Information
Center Instructions for Use and Online Help, and with Philips TeleMon A02/
A03 Companion Monitor Instructions for Use. See also the Philips IntelliVue
Telemetry Training Program.
About this Book
+
Transceivers as used with the Philips IntelliVue Telemetry System with
Document
Conventions
Warnings
WarningWarning
Warnings are information you should know to avoid injuring patients and
personnel.
Cautions
Caution
Cautions are information you should know to avoid damaging your equipment
and software.
iii
Product Safety Information
Notes
Note—Notes contain additional information on use of the Philips IntelliVue
Telemetry System.
Procedures
Procedures are indicated in text by the heading “Task Summary” followed by
the following table:
Step
Bold Typeface
Objects of actions in procedures appear in
example:
Action
1
2
3
Select the
Standby button.
Product Safety Information
The following general warnings and cautions apply to use of the Philips
IntelliVue Transceivers in a Philips IntelliVue Wireless Network. Additional
warnings and cautions specific to a particular feature are provided in the
appropriate section.
bold typeface. Note the following
General
iv
WarningWarning
For continued safe use of this equipment, it is necessary that the listed
instructions are followed. Instructions in this manual in no way supersede
established medical procedures.

Product Safety Information
WarningWarning
Do not touch the patient, or table, or instruments, during defibrillation.
The battery door must be closed during defibrillation. These steps protect
the clinician from high defibrillator voltage.
WarningWarning
This device is not to be used in the vicinity of electrosurgical units because
use may interrupt or interfere with the transmission of signals from the
transceiver.
WarningWarning
This equipment is not suitable for use in the presence of a flammable
anesthetic mixture with air or with oxygen or nitrous oxide
WarningWarning
Do not use patient cables with detachable lead wires that have exposed
male pins. Electrocution could result if these pins are plugged into AC
power.
WarningWarning
Use of product accessories (e.g., ECG leadsets, SpO2 sensors) other than
those prescribed by Philips could lead to patient injury.
WarningWarning
To avoid strangulation, do not tie a pouch solely around the patient’s
neck.
v

Product Safety Information
ECG/
Arrhythmia All Patients
WarningWarning
ECG SAFETY FOR ALL PATIENTS
Always confirm Information Center observations with clinical
observation of the patient before administering interventions.
Every lead must be secured to an electrode on the patient.
Conductive parts of electrodes must not contact earth or other conductive
parts.
Philips recommends that you change the lead label only to reflect the
physical placement of electrodes. This will ensure a match between the
monitored lead and the label, and prevent any possible confusion.
When switching between EASI and standard monitoring, there is a loss of
data for 30 seconds.
vi

Product Safety Information
WarningWarning
ST/AR ARRHYTHMIA SAFETY FOR ALL PATIENTS
During complete heart block or pacemaker failure (to pace or capture),
tall P-waves (greater than 1/5 of the average R-wave height) can be
erroneously counted by the arrhythmia algorithm, resulting in missed
detection of cardiac arrest.
Learning/Relearning
- If you initiate learning during ventricular rhythm, the ectopics can be
incorrectly learned as the normal QRS complex. This can result in missed
detection of subsequent events of V-Tach and V-Fib.
- When using EASI ECG monitoring, Relearn happens automatically
when there is a LEADS OFF technical alarm. If learning takes place
during ventricular rhythm, the ectopics can be incorrectly learned as the
normal QRS complex. This can result in missed detection of subsequent
events of V-Tach and V-Fib. Be sure to check the beat labels and initiate a
relearn to correct. Therefore, when a technical alarm is generated:
1. Respond to the technical alarm [for example, reconnect the
electrode(s)].
2. Ensure that the arrhythmia algorithm is labeling beats correctly.
vii

Product Safety Information
ECG/
Arrhythmia Paced
Patients
WarningWarning
ECG SAFETY FOR PACED PATIENTS
The output power of the transceiver and other sources of radio frequency
energy, when used in the proximity of a pacemaker, can be sufficient to
interfere with pacemaker performance. Due to the shielding effects of the
body, internal pacemakers are somewhat less vulnerable than external
pacemakers. However, caution should be exercised when monitoring any
paced patient.
In order to minimize the possibility of interference, position electrodes,
electrode wires, and the transceiver as far away from the pacemaker as
possible.
Consult the pacemaker manufacturer for information on the RF
susceptibility of their products and the use of their products with the
Philips IntelliVue Telemetry System. See the IntelliVue Information Center
Instructions for Use for additional information on monitoring paced
patients.
viii

Product Safety Information
WarningWarning
ST/AR ARRHYTHMIA SAFETY FOR PACED PATIENTS
It is possible that pacemaker pulses will not be detected when the ECG
analog output of a defibrillator or telemetry unit is plugged into a bedside
monitor. This can result in the arrhythmia algorithm’s failure to detect
pacemaker non-capture or asystole.
Some pace pulses can be difficult to reject. When this happens, the pulses
are counted as a QRS complex, and could result in an incorrect HR and
failure to detect cardiac arrest or some arrhythmias. Keep pacemaker
patients under close observation.
-- During complete heart block or pacemaker failure (to pace or capture),
tall P-waves (greater than 1/5 of the average R-wave height) can be
erroneously counted by the arrhythmia algorithm, resulting in missed
detection of cardiac arrest.
-- When arrhythmia monitoring paced patients who exhibit only intrinsic
rhythm, the monitor can erroneously count pace pulses as QRS complexes
when the algorithm first encounters them, resulting in missed detection of
cardiac arrest.
For patients who exhibit intrinsic rhythm only, the risk of missing cardiac
arrest can be reduced by monitoring these patients with the low heart rate
limit at or slightly above the basic/demand pacemaker rate. A low heart
rate alarm alarms you when the patient begins pacing. Proper detection
and classification of the paced rhythm can then be determined.
-- When an external pacemaker is being used on a patient, arrhythmia
monitoring is severely compromised due to the high energy level in the
pacer pulse. This can result in the arrhythmia algorithm’s failure to
detect pacemaker non-capture or asystole.
ix

Product Safety Information
SpO
2
WarningWarning
SpO2 SAFETY
Always confirm Information Center observations with clinical
observation of the patient before administering interventions.
Prolonged, continuous monitoring can increase the risk of changes in skin
characteristics, such as irritation, reddening, blistering or pressure
necrosis, particularly on patients with impaired perfusion and varying or
immature skin morphology. Specific attention must be given to sensor site
inspection for changes in skin quality, proper optical path alignment and
attachment. Check the application site at regular intervals and change the
site if any compromise in skin quality should occur. More frequent
checking can be required due to an individual patient's condition.
Using a sensor during MR imaging can cause severe burns. To minimize
this risk, ensure that the cable is positioned so that no inductive loops are
formed. If the sensor does not appear to be operating properly, remove it
immediately from the patient.
Do not use disposable sensors on patients who exhibit allergic reactions to
the adhesive.
Injected dyes such as methylene blue or intravascular dyshemoglobins
such as methemoglobin and carboxyhemoglobin can lead to inaccurate
(over-estimated) measurements.
Interference leading to inaccurate measurements can be caused by:
- High levels of ambient light (Hint: cover application site with opaque
material)
- Electromagnetic interference
- Excessive patient movement and vibration.
x

Content s
General. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-iv
ECG/ Arrhythmia -All Patients . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-vi
ECG/Arrhythmia - Paced Patients . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-viii
SpO2. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-x
1. Basic Operation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-1
The Philips IntelliVue Telemetry System. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-2
System Features . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-3
The IntelliVue Transceiver. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-3
Transceiver Controls - Front . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-7
Transceiver Controls - Back . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-11
Turning the Transceiver On . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-15
Turning the Transceiver Off . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-16
Testing intelliVue Transceiver Functionality . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-17
Self Test . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-17
Status Check. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-18
Battery Information. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-19
Battery Safety Information. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-19
Inserting/Removing Batteries . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-20
Checking the Battery Power Level . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-22
Briefing the Patient . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-24
Pouch Use . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-24
Securing the Pouch. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-25
Showering . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-26
Transceiver Use with TeleMon A02/A03. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-27
2. Alarms. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-1
Alarm Indicators . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-2
Testing Alarm Indicators. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-2
Suspending/Pausing Alarms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-2
Resuming/ Unsuspending Alarms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-4
Standby Mode . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-5
Alarm Behavior with Own Bed Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-7
Physiologic Alarms. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-9
Technical Alarms (INOPs) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-14
Contents-1

3. ECG Monitoring . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-1
ECG Safety Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-2
Measuring ECG. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-3
EASI ECG . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-3
ECG Leadsets. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-3
ECG Leads Monitored. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-4
Positioning ECG Electrodes. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-8
Electrode Placement. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-10
Connecting the ECG Cable. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-19
Verifying Electrode Connections . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-22
Monitoring during Leads Off . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-23
ECG Fallback. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-23
Extended Monitoring . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-24
Relearning . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-24
Using EASI Leads to Troubleshoot. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-24
Optimizing ECG Measurement Performance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-25
The Telemetry Signal. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-25
Trouble- shooting Signal Disturbances . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-26
Dropouts. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .3-26
Muscle and Movement Artifact . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-27
4. ST/AR Arrhythmia & ST Segment Monitoring. . . . . . . . . . . . . . . . . . . . . . . . 4-1
ST/AR Arrhythmia Algorithm . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-2
Safety Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-2
ST/AR Arrhythmia Analysis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-3
ST/AR ST Segment Algorithm. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-6
The Measurement. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-6
Algorithm Processing. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-8
Displayed ST Data. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-8
EASI ST Analysis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-8
ST Operation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-9
ST Alarm Settings . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-11
5. SpO2 Monitoring . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-1
SpO2 Safety Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-2
SpO2 Information for the User . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-3
Pulse Oximetry Measurement. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-4
Pulse Indication . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-5
Selecting a SpO2 Sensor. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-5
Applying the Sensor. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-9
Contents-2

Sensor Application Safety Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-9
Site Selection . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-9
Sensor Application . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-10
Connecting the SpO2 Cable. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-14
Measuring SpO
2. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-15
Spot Check . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-15
Continuous. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-16
SpO
Measurement when Connected to TeleMon . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-17
2
Turning SpO2 Monitoring Off . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-18
Turning the SpO2 Parameter On/Off . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-19
SpO
Parameter Auto ON. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-19
2
Understanding SpO2 Alarms. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-20
Optimizing SpO2 Measurement Performance. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-21
Optimizing Sensor Performance. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .5-22
6. Telemetry Functions at the Information Center . . . . . . . . . . . . . . . . . . . . . . . 6-1
Telemetry Controls in the Patient Window . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-2
Locating the Transceiver (Find Device) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-3
To locate a transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-3
To silence the sound . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-3
Patient-Configurable Settings in Telemetry Setup . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-3
Unit-Configurable Settings . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-7
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .6-8
7. Maintenance & Troubleshooting . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7-1
Maintenance. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-2
Basic Monitoring . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-2
Label Assignment for Replacement Transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-2
Transceiver Cleaning, Disinfection, & Cross-Infection Prevention . . . . . . . . . . . . . . . . . . . . . . .7-4
Cleaning the Transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-5
Disinfecting the Transceiver. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-6
Cross-Infection Prevention for the Transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-7
Troubleshooting . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-15
Basic Troubleshooting . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-15
Testing Alarms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-15
Information Signals. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .7-15
8. Safety Standards & Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-1
Regulatory Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-2
Intended Use. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-2
Contents-3

Indications for Use. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-2
Rx. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-2
Patient Population . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-2
Safety Standards. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-2
Essential Performance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-3
System Classification. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-3
FCC Compliance (USA only) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-4
AC Power Source. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-5
Battery Life Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-5
Electromagnetic Compatibility . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-6
Restrictions for Use . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-8
Radio Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-8
M4841A Transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-8
WMTS Channel Frequencies. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-10
Physical Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-11
ECG-only Transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-11
ECG/SpO2 Transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-12
Environmental Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-12
M4841A Transceiver . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-12
Measurement Specifications . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-13
ECG . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-13
SpO2. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8-15
SpO2 Sensor Accuracy . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .8-16
A. Accessory List. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-1
Accessory Safety. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-2
ECG Accessories. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-2
Electrodes. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-2
Leadsets . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-2
Trunk Cables . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-3
Pouches . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-3
Skin Prep Paper . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-3
Alignment Guides . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-4
Gunk Guards . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-4
SpO2 Accessories . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-4
Reusable Sensors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-4
Disposable Sensors - Single Use . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-5
Wristband. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . A-6
B. Sales and Support Offices . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . B-1
Contents-4

1
Basic Operation
This chapter introduces the Philips IntelliVue Telemetry System with Smarthopping Technology and the IntelliVue TRx and TRx
the following sections:
• The Philips IntelliVue Telemetry System. . . . . . . . . . . . . . . . . . . . . . . 1-2
• The IntelliVue Transceiver. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-3
• Turning the Transceiver On . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-15
• Testing intelliVue Transceiver Functionality . . . . . . . . . . . . . . . . . . . 1-17
• Battery Information. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1-19
• Pouch Use. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .1-24
• Transceiver Use with TeleMon A02/A03. . . . . . . . . . . . . . . . . . . . . . 1-27
• Transceiver Use with TeleMon A02/A03. . . . . . . . . . . . . . . . . . . . . . 1-27
+
Transceivers. It includes
Introduction
Basic Operation 1-1

The Philips IntelliVue Telemetry System
The Philips IntelliVue Telemetry System
The Philips IntelliVue Telemetry System with Smart-hopping Technology
provides ambulatory and bedside monitoring of ECG and SpO
the radio frequency (RF) spectrum newly allocated for medical telemetry
applications by the Federal Communications Commission (FCC). The System
enables clinically significant data and control information for adult and pediatric
patients in healthcare facilities to be received from and sent to the transceiver, a
patient-worn device, via a bi-directional RF link over the Wireless Medical
Telemetry Service (WMTS) spectrums 1395-1400 MHz and 1427-1432 MHz.
The System uses smart-hopping technology to dynamically manage the RF
spectrum utilization per transceiver, thus allowing a virtually unlimited number
of simultaneously operating transceivers within the Philips IntelliVue Telemetry
System. The frequency-agile system changes frequency without user
involvement or awareness whenever interference occurs.
The System encompasses a number of individual units which connect together to
form a complete method of transporting ambulatory patient data to a central
repository for subsequent distribution to clinical staff. An installation typically
consists of the following components:
parameters over
2
1-2 Basic Operation
• M4841A Transceivers, bi-directional patient-worn devices
• M4842A Access Points (AP), centers for bidirectional communication
between the transceivers and the Information Center.
• IntelliVue Wireless Network (IWN) infrastructure (including M4843A
Access Point Controllers, M4844A Sync Units, M4845A Power Supply
Units)
• M3150A IntelliVue Information Center for centralized monitoring
• M3154A IntelliVue Database Server (optional) for centralized data
management
• M2636A TeleMon A02/A03 Companion Monitor (optional) for local
display, NBP measurement and local alarms.
The network interconnects Access Points to the Information Center and other
central equipment via the same network that connects IntelliVue bedsides to the
Information Center. Access points receive signals, and unlike traditional antenna
systems, can communicate bidirectionally. Access Points are powered,
controlled and managed remotely via the IWN.

The IntelliVue Transceiver
System
Features
• Full patient mobility within the areas defined by the wireless coverage
provided by multiple Access Points.
• Expanded geographic coverage area for a a given patient assigned to an
IntelliVue Clinical Network. Physiological data is transported from the
transceiver; a reverse data channel enables data to be transported to the
transceiver.
• 3-minute Alarm Pause/Suspend initiated at the transceiver.
• Standby mode when a patient is away from the unit and not being
monitored by the Philips IntelliVue Telemetry System.
• Find Device feature for locating a lost transceiver within the coverage
area.
• Access Points operating concurrently with the networked bedside wireless
capability while sharing some of the ICN infrastructure.
• Use of the radio spectrums newly allocated by the FCC specifically for
medical telemetry applications.
• Connectivity to TeleMon for display of patient measurements - including
NBP - at the bedside.
diagram to come
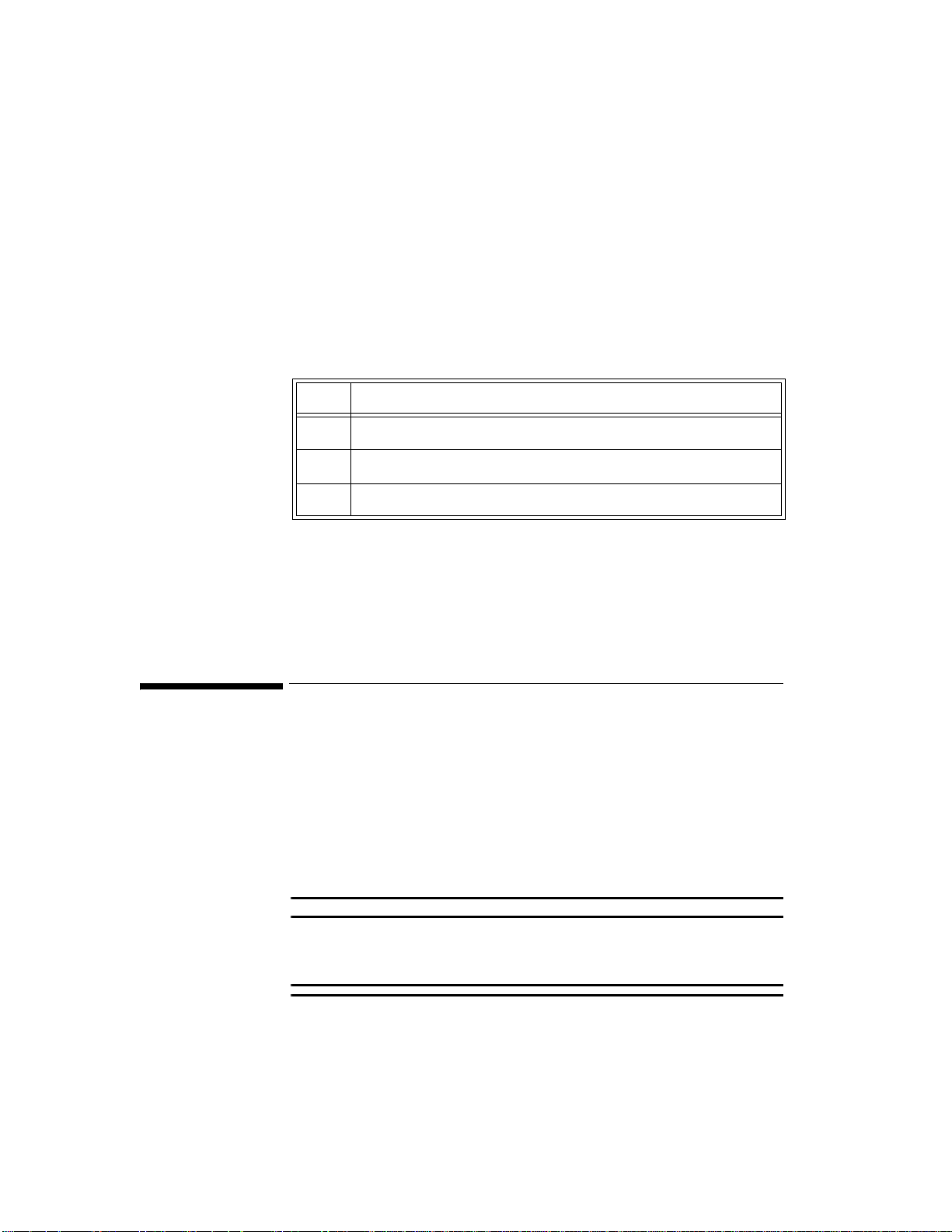
The IntelliVue Transceiver
The Philips IntelliVue transceiver is a patient-worm device for monitoring ECG
and SpO
on adult and pediatric patients in the IntelliVue Telemetry System
2
System Information Flow/Smart-hopping
Basic Operation
1-3

The IntelliVue Transceiver
with Smart-Hopping Technology, a cellular infrastructure network. The
transceiver combines traditional transmitter features with two-way
communication capability with the IntelliVue Information Center. The
transceiver is designed to be easy for clinicians to use and comfortable for
patients to wear. Colored labels provide departmental identifiers. The leadsets
are optimized for ambulating patients, with a cable length of 79 cm (30 inches).
Protective covers prevent dirt from accessing unused ECG and SpO
thus simplifying cleaning.
cable ports,
2
The transceiver is available in two models, the ECG only called the IntelliVue
TRx, and the ECG-SPO2 version, called the IntelliVue TRx
+
. The models are
listed below and illustrated on the following pages in this chapter. Subsequent
tables describe the buttons, indicators, labels, ports, safety symbols & other
markings, and auditory information signals of the transceiver respectively.
Transceiver Model
(M4841A) Measurements
IntelliVue TRx ECG
IntelliVue TRx
+
ECG, SpO
2
The transceiver comes with a start-up kit of batteries, electrodes, and pouches.
1-4 Basic Operation
M2601B
IntelliVue TRx
IntelliVue TRx
M4841A
M4841A
+
+
EASI, 3
EASI, 3
The IntelliVue Transceiver
5
5,6
Transceiver
Features
IntelliVue TRx Transceiver - ECG only
Note— The IntelliVue Transceiver and M2601B Transmitter are similar in
appearance. If your hospital uses both, you can distinguish between them by:
• Name on the front of the device
• Label background color (pale gray for transceivers, dark gray for
transmitters)
• Clinician-selectable 5-lead Standard or EASI leads in same device, at the
bedside
• 6-leadset with two V leads for diagnosing multiple cardiac abnormalities,
including wide-QRS complex tachycardias and acute myocardial
ischemia/infarction
• Powered by two AA Alkaline batteries
• Spot-Check SpO
without using any control buttons
2
Basic Operation
1-5

The IntelliVue Transceiver
•FAST-SpO2 (Fourier Artifact Suppression Technology) for improved
motion artifact rejection and low-perfusion performance
• Audio feedback for Spot Check SpO
completion and other common tasks
2
• Simultaneous operation in system with M2601A Transmitter
• Two sizes - smaller ECG only version and larger ECG/SpO
version
2
• Battery gauge on transceiver and, if configured, at Information Center
• Colored labels provide clinical unit identifiers.
• Leadsets are optimized for ambulating patients, with a cable length of 79
cm (30 in).
• Gunk guards prevent debris from accessing unused ECG and SpO
cable
2
ports and the unused TeleMon/Service port, thus simplifying cleaning.
• Pouch with clear front and flap.
Use with
Information
Center
Use with
TeleMon
A02/A03
The bi-directional capability enables remote control from the Information Center
of the following transceiver operations:
• From the Telemetry Setup Window:
–SpO
measurement mode (Spot Check, Continuous, or Off)
2
– Display and storage of real-time pleth wave (enable/disable)
– Volume of audible transceiver information signals
– Find device
– Suppression of SpO
technical alarms during NBP measurement
2
• From the Patient Window
– Standby mode
– Filter bandwidth for ST measurement on/off
– Alarm Pause/Suspend (enable/disable)
• From Unit Settings
– Display of battery gauge (enable/disable)
– 3-wire Lead Selection
The system supports Own Bed Overview, the pairing of a telemetry bed with an
IntelliVue Patient Monitor (Release B or higher) for a single patient. Own Bed
Overview provides the telemetry-monitor data (waveforms, numerics and
alarms) in an integrated form both on the bedside monitor and at the IntelliVue
Information Center.
The transceiver can employ the full functionality of the TeleMon A02/A03
companion monitor, including NBP measurement and local display of alarms.
Connection is made through an interface cable, or tether, at the TeleMon service
1-6 Basic Operation
port. Please refer to the TeleMon A02/A03 Instructions for Use for general
operating instructions and “Transceiver Use with TeleMon A02/A03” on page
1-27 for an operational summary.
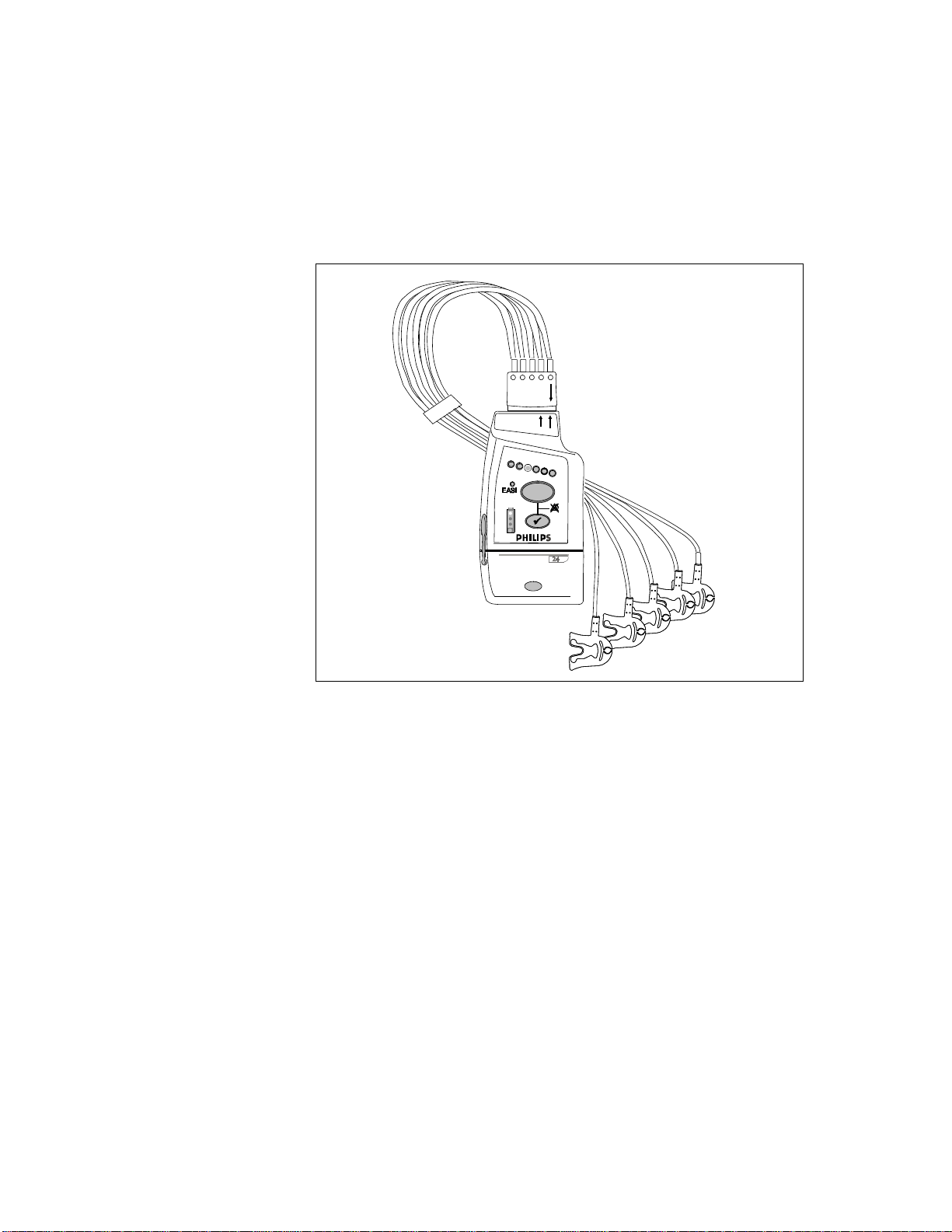
Transceiver Controls - Front
P1
The IntelliVue Transceiver
P2
+
IntelliVue T R x
M4841A
EASI, 3
5,6
I1
I2
B1
I3
I4
B2
P3
B3
The labeled items in the diagram include: Buttons (B1-B3);
Indicators (I1-I4); Labels (L1-L4); Ports (P1-P3). Additional
Labels, and Safety Symbols & Other Markings (S1-S12, appear on the back of the transceiver.
IntelliVue TRx+ Transceiver - Front View
L1
L2
L3
Basic Operation
1-7
The IntelliVue Transceiver
Buttons
+
IntelliVue TRx
M4841A
EASI, 3
5,6
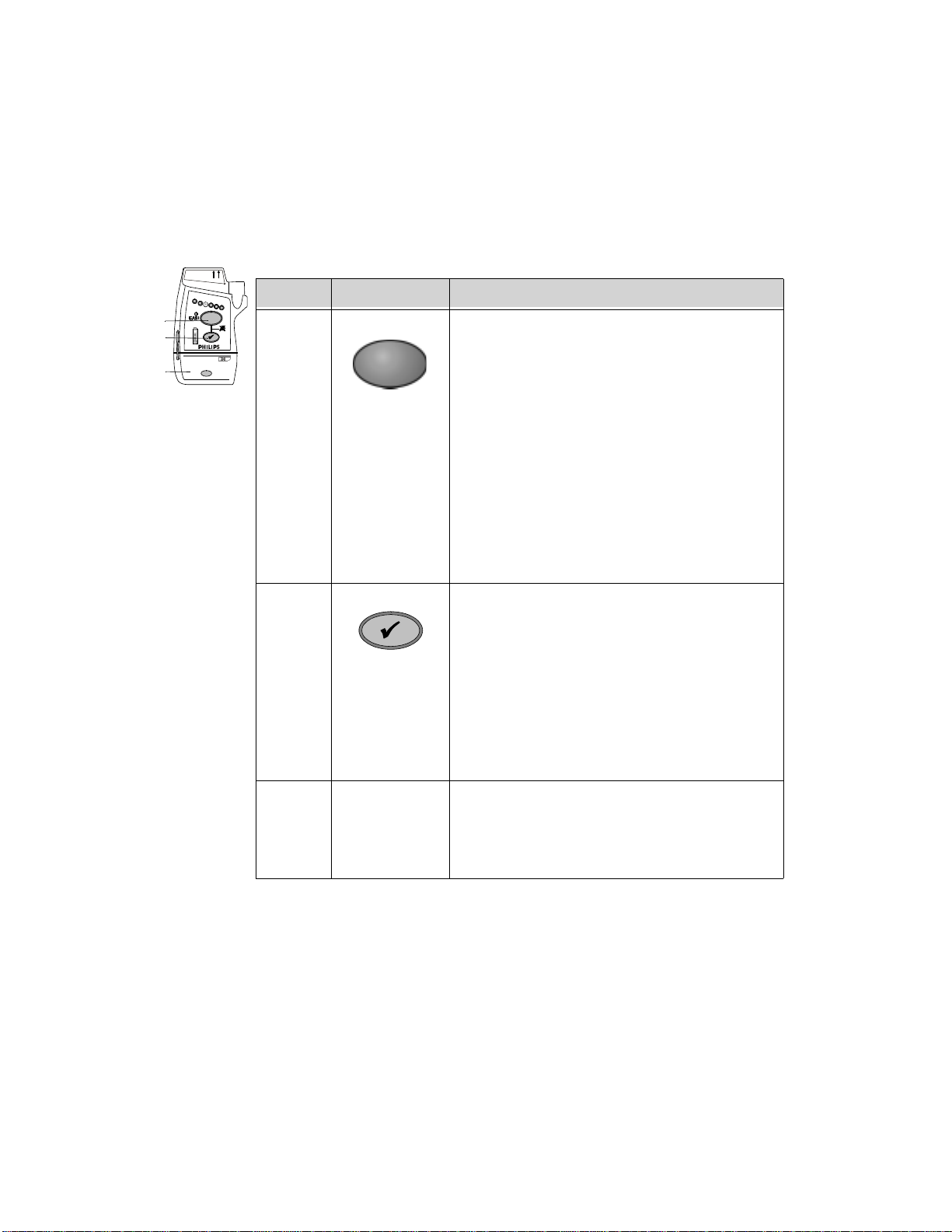
Callout Button Definition
B1
B2
B3
B1 Telemetry Button: Depending on configuration,
B2 Check Button. Initiates a Status Check of the
directs the Information Center to generate a Nurse
Call alarm, remote recording, Nurse Call alarm
and recording, or none. See “Patient-Configurable
Settings in Telemetry Setup” on page 6-3.
Note—Delayed recordings generated by the
Telemetry button are stored in Alarm Review at
the Information Center.
When pressed simultaneously with the Check
button, turns Alarm Suspend/Pause on/off (not
when tethered to TeleMon). See “Suspending/
Pausing Alarms” on page 2-2.
Transceiver. See “Status Check” on page 1-18.
1-8 Basic Operation
When pressed simultaneously with the Telemetry
Button, turns Alarm Suspend/Pause on/off (not
when tethered to TeleMon). See “Suspending/
Pausing Alarms” on page 2-2.
Silences the Find Device tone.See “Telemetry
Controls in the Patient Window” on page 6-2.
B3 Power On/Off Battery Compartment. Insertion of batteries
turns transceiver power on; removal of batteries
turns power off. See “Turning the Transceiver
On” on page 1-15.
I1
I2
I3
I4
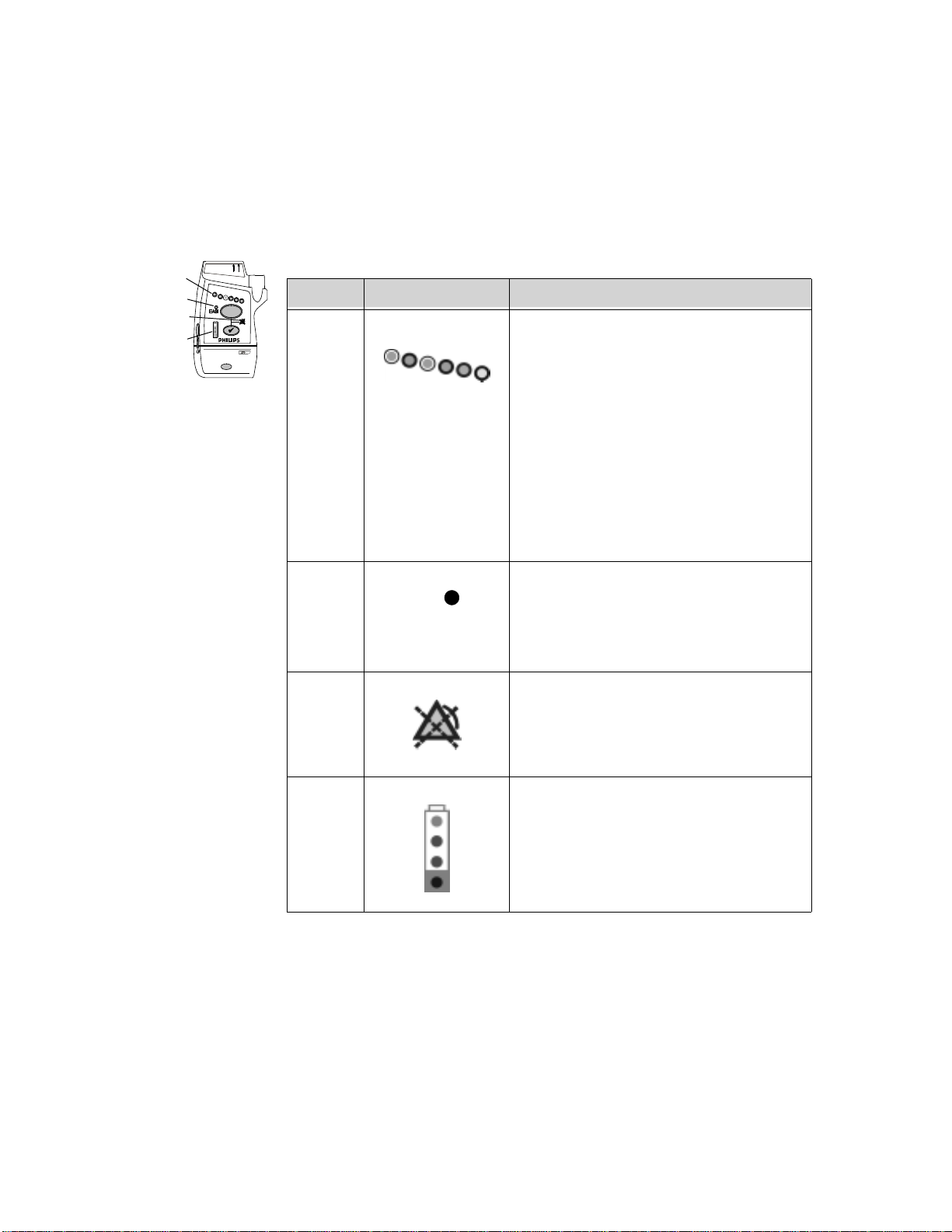
Indicators
+
IntelliVue TRx
M4841A
EASI, 3
5,6
The IntelliVue Transceiver
Callout Indicator Definition
I1 Lead Indicator.
Illuminates momentarily during leadset
insertion to indicate attached leads.
Illluminates when Check button is pressed to
indicate attached leads.
During a Leads Off condition, illuminates to
indicate the lead(s) that need to be reapplied.
Momentarily illuminates three alternate
lights, indicating the transceiver has no
Equipment Label assigned.
I2 EASI Indicator. Illuminates momentarily
upon insertion of leadset in EASI position.
EASI
Illuminates when Check button is pressed if
EASI is in use.
I3 Alarms Suspend/Pause Indicator.
Illuminates during 3 minute alarm pause
initiated at transceiver or Information Center.
I4 Battery Gauge. Illuminates when the EHck
button is pressed to indicate the amount of
power remaining in the batteries. Valid only
for recommended battery type. See
“Checking the Battery Power Level” on page
1-22.
Basic Operation
1-9

The IntelliVue Transceiver
Labels
IntelliVue TRx
M4841A
+
EASI, 3
L1
5,6
Callout Label Definition
L2
L3
L1 Leadset Insertion Guide. Assist in aligning
IntelliVue TRx
M4841A
EASI, 3
the ECG cable for different leadsets. See
“Connecting the ECG Cable” on page 3-19.
5,6
Note—If your unit uses only one monitoring
configuration, the transceiver may have
special "lock out" plugs that allow only one
way to insert the leadset.
L2 Device Identification Label. Identifies the
device within the IntelliVue Wireless
Network.
L3 Unit Identification Label. Uses one of
seven color-coded labels to identify a clinical
unit.
Ports
IntelliVue TRx
M4841A
P2
+
EASI, 3
5,6
Callout Definition
Note— Ports can be covered with protective covers (gunk guards) when not in
P1
P3
use. See “Gunk Guards” on page -4.
1-10 Basic Operation
P1 ECG Leadset Port. Connection for 3-wire or 5-wire leadset.
P2 SpO
Sensor Port. Connection for SpO2 sensor. (IntelliVue TRx+
2
only)
P3 TeleMon/Service Port. Connection for cable to TeleMon
Companion Monitor or to Service Tool.
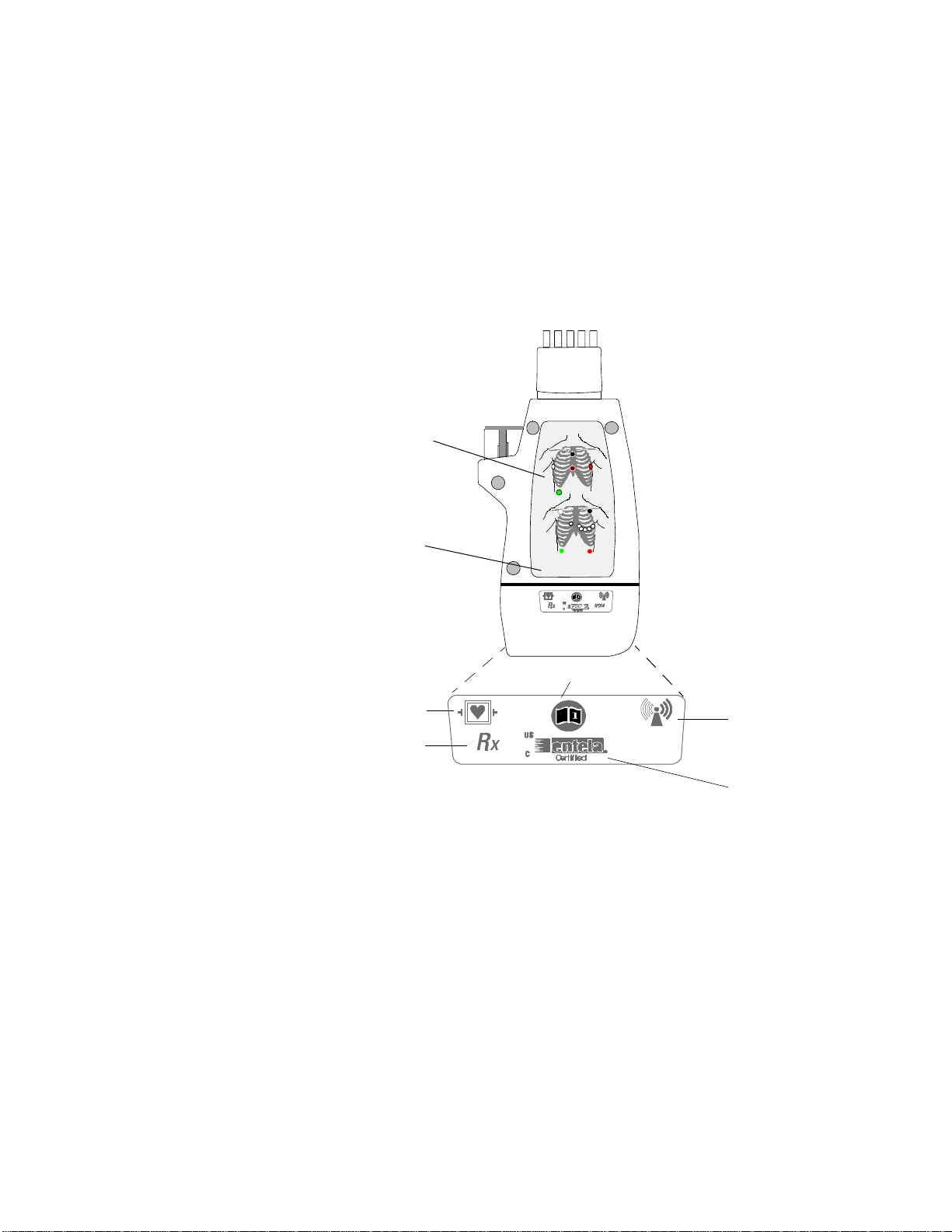
Transceiver Controls - Back
L4
S1
EASI
EASI
EASI
I
S
E
1
FCCID: XXXXXXXX
S6
The IntelliVue Transceiver
A
2
6
5
3
4
S7-S11 not shown
(inside battery
compartment)
S2
S3
IntelliVue TRx+ Transceiver - Back View
Basic Operation
S4
S5
1-11
The IntelliVue Transceiver
Labels
L4
EASI
EASI
I
Safety
Symbols &
Other Marks
S
A
E
1
2
6
345
Callout Definition
L4 Electrode Placement Diagrams (See “Positioning ECG
Electrodes” on page 3-8.)
Callout Label Definition
S1
FCCID: XXXXXXXX
Federal Communications Commission
(FCC) (PTT) label
S2 Patient connections are protected against
defibrillation (DEFIBRILLATIONPROOF) and are a TYPE CF APPLIED
PART.
S3 Prescription device.
R
x
1-12 Basic Operation
S4 Non-Ionizing Radiation. Interference to
electronic equipment may occur in the
vicinity of devices marked with this
symbol.
S5
Complies with all applicable Canadian and
American standards.
The IntelliVue Transceiver
Callout Label Definition
S6 Follow operating instructions.
i
S7 Philips Catalog Number
REF
S8 Serial Number (inside battery
compartment). Needed to identify the
SN
S9 MAC Address of device
equipment during a call to the Response
Center.
MAC
Auditory
Information
Signals
S10 Date of manufacture
S11 Battery Polarity
The transceiver produces auditory feedback to inform you of measurement and
battery conditions. Adjustable sounds can be set to 5 different volume levels or
turned off per patient at the Information Center (see “Patient-Configurable
Settings in Telemetry Setup” on page 6-3). Adjustable sounds include Check
Basic Operation
1-13

The IntelliVue Transceiver
button Standby functions, SpO2 measurement complete, outside of coverage
area warning, and the pulse detection tone.
Auditory Information
Signal
Definition
Single Tone Self Test passed
SpO
Spot Check measurement successfulnoyes
2
Single Tone, low pitch Pulse detection successful (when locally
initiated)
Double Tone Self Test failed
SpO
Spot Check measurement failed
2
Double Tone repeated
Out of range yes
every 5 seconds
Continuous Double
Find Device no
Tone, two pitches
Single Tone (when
Check button pressed)
Double Tone (when
Check button pressed)
Transceiver is associated with sector at
Information Center (after Standby).
Transceiver not associated with sector at
Information Center (after Standby).
Volume/Mute
Adjustable
yes
no
yes
yes
yes
Double Tone and all
indicators flashing
Fast Double Tone and
alternate Leads Off
indicators flashing
1-14 Basic Operation
No equipment label is assigned from
Information Center. No monitoring.
Equipment label is received from
Information Center and is awaiting local
acknowledgment by Check button press.
no
no

Transceiver
Safety
Information
Turning the Transceiver On
WarningWarning
If another radio medical device is operating at the same frequency as an
IntelliVue Transceiver, it is possible that either device will not function
properly.
WarningWarning
Although the transceiver is shielded against Electromagnetic Interference
(EMI), avoid the use of other electrically radiating devices in close
proximity to the transceiver because they might interfere with transceiver
operation.
WarningWarning
Place the transceiver in a pouch or over clothing, or both, during patient
use. The transceiver should not touch the patient’s skin during use.
Turning the Transceiver On
WarningWarning
Arrhythmia relearning is initiated whenever the transceiver is powered
down for one minute or longer. Be sure to check your patient’s arrhythmia
annotation for accuracy whenever relearn has occurred.
The transceiver is powered by two AA alkaline batteries. To turn the transceiver
on, insert both batteries. Remove the batteries to turn the power off.
The configuration data set by the Service Provider prior to transceiver use is
retained after battery removal.
Basic Operation
1-15

Turning the Transceiver On
When the transceiver is turned on, all indicators illuminate briefly and a
sequence of sounds indicates the instrument is ready for use. You should hear a
single beep indicating that the self test was passed, followed by a series of
double beeps while the transceiver attempts to associate with the Information
Center. The cessation of sounds indicates a successful association. If you hear a
single double beep or any other sound sequence, the automatic self-test of the
device has not passed, or there is another problem. Contact your Service
Provider.
Sounds at successful start-up
Self-test
insert batteries
Turning the
Transceiver
Off
Auto Shutoff Automatic Shutoff causes the transceiver to stop broadcasting a radio signal if
Turn off the transceiver by removing the batteries. A NO SIGNAL technical
alarm will be in effect at the Information Center until the device is turned on or
until Standby is initiated.
Telemetry monitoring can be turned off in the following ways:
• Manually, by activating Monitoring Standby at the Information Center
(see “Standby Mode” on page 2-5).
• Automatically, if Transceiver RF Auto Shutoff is enabled and there is no
ECG signal for 10 minutes.
Note—Turning off telemetry monitoring does not turn off the transceiver.
there is no ECG signal for 10 minutes. This prevents interference with other
transceivers in use. The technical alarm text at the Information Center is
Transmitter Off. To conserve battery power, remove batteries.
1 beep (pass)
Transceiver looking
for Info Center
beep beep repeated
every 3 seconds
Connected
no beep
1-16 Basic Operation

Testing intelliVue Transceiver Functionality
To restart monitoring, insert batteries if necessary, attach leads to the patient and
press the Check button to verify association with the Information Center.
Testing intelliVue Transceiver Functionality
There are two tests of IntelliVue Transceiver functionality:
• Self Test -performed automatically each time the transceiver is turned on
• Status Check - initiated manually by the clinician.
Self Test
WarningWarning
Do not use the transceiver for patient monitoring if it fails the Power On
Self Test.
In Case of
Failure
A self test of the transceiver functions is automatically performed each time that
the transceiver is turned on (that is, batteries are inserted).
Self Test
Status
Passed Single beep All indicators illuminate for 3 seconds
Failed Double beep One or more indicators do not light up.
If any portion of the self test fails, the transceiver will attempt to report the
failure to the monitoring system. In case of failure, use another transceiver, and
contact your Service Provider.
Auditory
Signal (if
configured on)
Visual Indicators
Basic Operation
1-17

Testing intelliVue Transceiver Functionality
Status
Check
You can check the status of the transceiver indicators at any time.
To initiate a Status Check, use the following instructions.
Step Action
1Press the
The following indicators should illuminate for as long as the Check
button is depressed.
2 If one or more of the expected indicators do not light up, check the
following:
If there is still a problem, contact your Service Provider for
assistance.
Check button.
• Battery gauge
• Type of leadset
• EASI (if in use)
• Lead block insertion. Make sure the leadset is correctly
inserted in the transceiver and the orange line at the base of
the cable is not visible (see “Connecting the ECG Cable” on
page 3-19).
• Power and position of batteries (see “Checking the Battery
Power Level” on page 1-22)
• Lead positions and connections (see “Verifying Electrode
Connections” on page 3-22)
1-18 Basic Operation

Battery Information
Battery
Safety
Information
WarningWarning
Use Duracell MN 1500 AA 1.5V Alkaline batteries to ensure specified
performance. Outdated, mismatched or poor-quality batteries can give
unacceptable performance (e.g., insufficient Battery-Low warning time).
The use of fresh high-quality alkaline batteries is strongly recommended.
Batteries should be removed from the transceiver at the end of the battery’s
useful life to prevent leakage.
If battery leakage should occur, use caution in removing the battery. The
leaked substance may cause eye or skin irritation. Avoid contact with skin.
Clean the battery compartment according to instructions in “Chapter 7.
Maintenance & Troubleshooting”. Wash hands.
Certain failure conditions, such as short circuits, can cause a battery to
overheat during normal use. High temperatures can cause burns to the
patient and/or user. If the transceiver becomes hot to the touch, place it
aside until it cools. Then remove the batteries and discard them. Have the
transceiver operation checked by your Service Provider to identify the
cause of overheating.
Battery Information
The battery door must be closed during defibrillation.
If you receive a BATTERY LOW alarm, the batteries must be promptly
replaced. A "Battery Low" condition that is not corrected will result in a
transceiver shutdown and cessation of monitoring.
Basic Operation
1-19

Battery Information
Disposal of
Batteries
Caution
The batteries must be removed if a transceiver will be stored for an extended
period of time.
Important—When disposing of batteries, follow local laws for proper disposal.
Dispose of batteries in approved containers. If local regulations require you to
recycle batteries, recycle batteries in accordance with those regulations.
Battery Life Battery life is dependent upon:
• Condition of the batteries
• Parameters being monitored - ECG only, ECG and Spot Check SpO
ECG and Continuous SpO
By observing the following guidelines, you can optimize battery life in the
transceiver:
• REMOVE the batteries when the transceiver is not in use.
• Disconnect the SpO
disconnected, the SpO
an extender cable will continue to drain power from the SpO
.
2
extender cable (if used). (When the SpO2 sensor is
2
functionality is automatically powered down, but
2
2
electronics.)
2
, or
Inserting/
Removing
Batteries
Arrhythmia relearning is initiated whenever the transceiver is powered
down for one minute or longer. Be sure to check your patient’s arrhythmia
annotation for accuracy whenever relearn has occurred.
Remove the batteries before storing a transceiver for an extended period of time.
1-20 Basic Operation
WarningWarning
Caution

Battery Information
The battery compartment is located at the bottom of the transceiver behind a
swinging door. It accommodates a pair of AA 1.5V Alkaline batteries. Only this
type of disposable battery shall be used.
Important—Do not use rechargeable batteries. Use of this type of battery will
adversely affect:
• Battery gauge performance
• Battery low warnings
• Battery life performance
Insert batteries into the transceiver using the following procedure.
Step Action
1 Open the battery compartment by swinging the compartment door
2 Insert two AA 1.5V Alkaline batteries, matching the polarity with
o
90
counterclockwise into an open hinged position.
the +/- indications inside the compartment.
+
+
Note— Both batteries are inserted with the + polarity in the same
direction.
Basic Operation
1-21

Battery Information
Step Action
3 Close the battery compartment door.
4 Listen for the start-up sounds.
Watch for the indicators on the front of the transceiver to illuminate
briefly.
5 Connect/reconnect the patient cables to the transceiver.
Removing the
Batteries
Checking
the Battery
Power Level
Batteries should be changed in sets, that is, if you change one battery, change
them both. To remove the batteries, simply open the battery compartment door
and push from the opening at the back of the compartment to pop the batteries
out. Transceiver settings (ECG leadset type, SpO
retained indefinitely when the batteries are removed.
If you remove good batteries to turn off the transceiver, keep them together as a
set for later re-use so that both batteries will have the same level of power
remaining.
Batteries should be removed when the transceiver is not in use or is being stored.
DO NOT "STORE" BATTERIES BY LEAVING THEM IN THE
INCORRECT POLARITY POSITION IN THE TRANSCEIVER.
Be careful not to short circuit the batteries. Short circuits are caused when a
piece of metal touches both the positive and negative terminals simultaneously
(for example, by carrying batteries in a pocket with loose change). More than a
momentary short circuit will generally reduce the battery life. In case of a short
circuit, discard both batteries in a pair, or just the shorted one if the batteries are
new.
When the Check button is pressed, the battery gauge on the transceiver indicates
the battery power level. It is reliable only when specified batteries (i.e., AA 1.5V
Alkaline) are used. The battery gauge is also displayed in the Patient Sector at
the Information Center to enable you to closely monitor battery status, for
example, at change of shift.
mode, volume, etc.) are
2
1-22 Basic Operation

Battery Information
Important— In the following table, battery life times are based on Duracell MN
1500 batteries. Battery life for other brands may be different.
Approximate
Battery Gauge
Approximate Battery
Life Remaining
Operating
Time
Functionality
Remaining
4 green indicators > 75% > 34.7 hours Normal operation
3 green indicators > 50% > 23.1 hours Normal operation
2 green indicators > 25% > 11.6 hours Normal operation
1 green indicator 25% to Low Battery level > 15 minutes Normal operation
1 red indicator Battery Low level to
Replace Battery level
no indicator Replace Battery level
(Check batteries for
correct polarity)
To check the power level:
Step Action
1Press the
Check button to determine the level.
2 If no indicators flash:
1. Check that the batteries are inserted properly.
2. Replace both batteries.
3. If there are still no indicators on the battery gauge, contact
your Service Provider.
If the indicators illuminate but do not behave as described above,
the transceiver has malfunctioned. Contact your Service Provider.
< 15 minutes Sp O
disabled
2
none Transceiver shutdown/
RF shutdown
Basic Operation
1-23

Briefing the Patient
Briefing the Patient
WarningWarning
Patients should be instructed not to open the battery cover while the
transceiver is in use.
If the Telemetry button has been configured to generate a Nurse Call, remote
recording, or both, instruct the patient to use the button when needed.
Note—If desired, you can turn off patient use of the button at the Information
Center. See “Patient-Configurable Settings in Telemetry Setup” on page 6-3.
Pouch Use The transceiver is not designed for direct contact with the patient’s skin. During
normal use, the transceiver should be worn over clothing, in a pocket or,
preferably, in a pouch. The carrying pouch is an appropriate means for holding
the transceiver.
1-24 Basic Operation

Securing
the Pouch
Briefing the Patient
Step Action
1 Secure the pouch on the patient with upper ties around the patient’s
head and arm, and lower ties around the patient’s lower torso.
WarningWarning
To avoid strangulation, do not tie a pouch solely around the
patient’s neck.
2 Insert the transceiver into the pouch with lead wires and SpO2
sensor cable, if used, exiting from the same side.
Important—Do not coil the cables inside the pouch. They are part of
the wireless system, and need to be freely exposed.
Basic Operation
1-25

Briefing the Patient
Step Action
3 Fold the flap down and snap closed.
4 Check that the patient is comfortable wearing the pouch with
transceiver.
Showering
Signal quality and leads off detection may be compromised when showering
due to significant patient movement. Appropriate clinical precautions must
be taken.
The transceiver can be used to monitor a patient in the shower, but only when
placed inside a Philips carrying pouch with the flap closed and the snaps
secured. The combination of the transceiver and pouch will withstand showering
for up to 10 minutes.
1-26 Basic Operation
WarningWarning

Transceiver Use with TeleMon A02/A03
Drying the
Transceiver
after
Showering
Accidental
Wetting
After showering, perform the following steps to continue monitoring:
1. Pat dry the leadset connections at the electrodes.
2. Wipe the lead wires with care.
3. If wet, dry the outside of the transceiver with a non-lint producing cloth.
4. If wet, wipe the inside of the battery compartment dry. Dry the batteries.
5. If wet, disconnect the ECG lead block and shake out any water. Dry the
connector pin area with a cotton swab.
Note—The transceiver should not be used for monitoring if the battery
compartment is wet. Remove the batteries and wipe the compartment dry before
continued monitoring use.
If the transceiver is accidentally immersed in liquid for up to 5 minutes, no
damage to the device and no electrical safety issues for the patient will result.
Remove the device, dry it off, and follow the procedure for cleaning/disinfection
or cleaning/cross-infection prevention under “Troubleshooting” on page 7-15 as
appropriate.
Transceiver Use with TeleMon A02/A03
When tethered to TeleMon, the following transceiver operational behavior will
be in effect.
Alarms
• 3 minute alarm pause/suspend can be initiated only from TeleMon, not
from the transceiver or from the Information Center. The Alarm Suspend/
Pause indicator on the transceiver will accurately reflect the current state
of alarm pause.
• Standby mode is not available.
ECG Operation
• Vb, the second V-lead in 6-wire lead-set, is not supported.
• After a change in leadset, the TeleMon returns to the default ECG
settings, and arrhythmia relearn occurs automatically. Be sure to check the
monitoring leads after you switch leads.
SpO2 Operation
Basic Operation
1-27

Transceiver Use with TeleMon A02/A03
•SpO2 is always in continuous mode.
• Changes in SpO
disconnected from TeleMon. Mode settings are defined in the following
table:
Mode at TeleMon Mode at Disconnected Transceiver
• After a change in SpO2 sensor, TeleMon returns to the default ECG
settings, and arrhythmia relearn occurs automatically. Be sure to check the
monitoring leads after you change the sensor.
Transceiver Operational Controls & Indicators
• When you press the Telemetry button, a Nurse Call alarm, recording,
alarm and recording, or neither will be generated, depending on the
configuration at the Information Center.
• When you press the Check button, the transceiver battery gauge indicates
full power, regardless of actual battery strength.
• When you press the Telemetry and Check buttons together, there is no
action; a 3-minute alarm pause/suspend can be initiated only from
TeleMon. The alarm pause indicator on the transceiver will accurately
reflect the current state of alarm pause.
• When the transceiver is connected to TeleMon, the battery gauge on the
transceiver and displayed at the Information Center always indicates "full"
regardless of actual battery power. To update the gauge, disconnect the
transceiver and wait several seconds for the updated battery strength to be
displayed.
Defibrillation
• In the event of patient defibrillation, it may take several seconds for the
ECG trace to reappear on the screen.
mode do not take effect until after the transceiver is
2
Continuous Continuous
5-min. Continuous
1-min. Continuous
Manual Spot Check
1-28 Basic Operation

2
Alarms
This chapter describes how to pause/suspend alarms temporarily. It also lists
Physiologic (Patient) Alarms and Technical (Inoperative Conditions) Alarms.
Both types of alarms are listed alphabetically.
• Alarm Indicators. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-2
• Suspending/Pausing Alarms. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-2
• Standby Mode. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-5
• Alarm Behavior with Own Bed Overview . . . . . . . . . . . . . . . . . . . . . . 2-7
• Physiologic Alarms. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2-9
• Technical Alarms (INOPs). . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .2-14
Introduction
Alarms 2-1

Alarm Indicators
Alarm Indicators
A description of visual and auditory information signals for patient and technical
alarms on the Information Center is located in the IntelliVue Information Center
Instructions for Use and the Information Center Online Help.The Information
Center documentation also includes the default alarm settings and physiological
alarm limit ranges
Testing
Alarm
Indicators
The visual alarm information signal on the transceiver is the Alarms Suspend
icon. During self test, the Alarm Suspend indicator illuminates, and a single tone
indicates association with the Information Center. These positive test results
indicate that the Alarm Suspend icon on the transceiver is functioning correctly
(see “Status Check” on page 1-18).
Suspending/Pausing Alarms
WarningWarning
If the Alarms Suspend indicator on the transceiver remains illuminated
after the button combination to unsuspend alarms is pressed, a the
transceiver malfunction may have occurred. Alarms resume automatically
after the 3 minute suspension period, or you can resume them manually at
the Information Center. The transceiver should be replaced, and the
malfunctioning unit should be sent to your Service Provider.
All alarms for a patient can be suspended/paused from the Information Center,
from the TeleMon Companion Monitor, or, depending on transceiver
configuration, from the transceiver itself. The Alarm Suspend/Pause duration is
fixed at 3-minutes. Alarms automatically resume after 3 minutes, and can be
reactivated manually earlier.
2-2 Alarms

Suspending/Pausing Alarms
If connected to TeleMon, alarms can be suspended only from TeleMon, and not
from the Information Center, and the Alarms Suspend icon on the transceiver is
lit (see “Transceiver Controls - Front” on page 1-7), and an ALARMS
SUSPENDED message appears at TeleMon and the Information Center.
Important—Patient monitoring (display of patient waveforms and numerics)
continues for the duration of Alarm Suspend/Pause.
Step
Action
1 To activate Alarm Pause/Suspend at the transceiver, press the
Telemetry
and Check buttons simultaneously.
While alarms are suspended:
• The transceiver illuminates the Alarms Suspend icon.
• The message ALARMS SUSPENDED (or ALARMS
PAUSED) is displayed in the Patient Sector at the
Information Center.
2 For instructions on how to pause/suspend alarms from the
Information Center, see the IntelliVue Information Center
Instructions for Use and the Information Center Online Help.
Alarms
2-3

Suspending/Pausing Alarms
Resuming/
Unsuspending
Alarms
Alarms will be resumed automatically after 3 minutes. You can cancel alarm
suspend manually before the 3-minute period has expired from the transceiver
(see following directions) or from the Information Center.
Step Action
1Press the
Telemetry and Check buttons simultaneously.
• Auditory alarm indication at the transceiver is switched on.
• The Alarm Suspend icon is turned off.
• The message ALARMS SUSPENDED (or ALARMS
PAUSED) is removed from the display.
2 Alternately, you can unsuspend alarms at the Information Center.
See IntelliVue Information Center Instructions for Use or Online
Help for directions.
2-4 Alarms

Standby Mode
Standby Mode
Standby mode is useful when the transceiver is temporarily removed from a
patient or when a patient is intentionally moved out of range of an Access Point.
Standby suspends monitoring so you won’t get any patient alarms or waveforms.
It also disables the out of range audio signal. Patient data and current settings are
preserved during the Standby period.
Standby also serves to turn telemetry monitoring off when a patient is
discharged. Patient data is not erased and unit default settings are not in effect
until the discharge operation is performed. A bed should be put into Infinite
standby until a new patient is connected to the transceiver. Monitoring can then
be restarted by a click of the Resume Monitoring button.
Note—To discharge a patient:
1. Disconnect the patient.
2. Remove batteries from the transceiver.
3. Put bed in Standby.
You place a patient in standby mode at the Information Center. You can select
the duration of the standby period (Infinite, 4, 3, 2, or 1 hours, 30, 20, or 10
minutes). The approximate time of resumption of monitoring will be di splayed.
To restart monitoring, click on
Resume Monitoring in the Patient Sector.
Initiating
Standby
Note—Unless you are discharging a patient, keep the leads attached until the
patient is in Standby. If you remove the leads before putting a patient into
Standby, you’ll get an ECG LEADS OFF technical alarm, as well as reminders,
if configured.
Step Action
1 In the Patient Window or All Controls at the Information Center,
select the
Standby button.
2 Select the patient destination from the pre-defined list.
Alarms
2-5

Standby Mode
Step Action
3 Depending on your equipment configuration, select the duration of
the standby period (30, 20 or 10 minutes; 2, 3 or 4 hours; or
infinite).
Resuming
Monitoring
4 Select the
Suspend Monitoring button. This suspends all
monitoring and displays the following messages in the Patient
Sector: PATIENT LOCATION:xxx (for example, X-ray) and
TELEMETRY STANDBY. The approximate time of resumption is
also displayed.
Step Action
1 If the standby period has expired when the patient returns to
the unit, monitoring will resume automatically.
To verify the resumption of monitoring, press the Check button.
You should hear a single beep.
2 If the standby period has not expired when the patient returns
to the unit, monitoring must be reactivated manually.
Either click Resume Monitoring at the Information Center or
press the Check button on the transceiver to establish
reassociation. The audible tone (single beep) at the transceiver
verifies that monitoring has resumed.
Note—When you take an EASI patient out of Standby, the lead settings will be
reset to the Information Center’s default lead settings.
2-6 Alarms

Alarm Behavior with Own Bed Overview
Alarm Behavior with Own Bed Overview
Both the IntelliVue Patient Monitor and the telemetry system source alarms. The
following tables summarize alarm behavior when Own Bed Overview is used.
For detailed information, see the IntelliVue Patient Monitor Instructions for Use
and the IntelliVue Information Center Instructions for Use.
Alarm Pause/Suspend
When alarms are paused/suspended, the messages and types of alarms affected
depend on where the pause/suspend was initiated.
If alarms are
paused/
suspended
from....
Information
Center
IntelliVue
Patient Monitor
these alarms are
paused/
suspended
both bedside and
telemetry
measurements
bedside
measurements only
and this message appears
Information Center: ALARMS
PAUSED or ALARMS
SUSPENDED
Bedside: ALARMS OFF in
Overview window and ALARMS
PAUSED or ALARMS OFF on the
monitor (depending on
configuration)
Information Center: BED
ALARMS PAUSED or BED
ALARMS SUSPEND (depending on
configuration)
Bedside: ALARMS PAUSED or
ALARMS OFF
Alarms
2-7

Alarm Behavior with Own Bed Overview
Alarm Silence
When an active alarm is silenced, the types of alarms that are silenced depend on
the alarm source and where the silence was initiated.
Alarm
Source
Where
Silenced
Effect at
Paired
Bedside
Bedside alarm Bedside Alarm is
silenced
Effect at
Information
Center
Bedside alarm is
silenced. There is
no effect on
telemetry alarms
Telemetry
alarm
Bedside No effect on
telemetry
No effect on
telemetry alarms
alarms
Bedside and/or
telemetry alarm
Overview
Silence Control
Bedside or
telemetry alarm
is silenced
Bedside alarm is
silenced (if
Silence
Overview
Alarms is
configured)
Bedside and/or
telemetry alarm
Note—If tethered to TeleMon, silencing an active alarm at TeleMon silences the
Information
Center
Bedside or
telemetry alarm
is silenced
Bedside or
telemetry alarm
is silenced
alarm at TeleMon only. It has no effect on the paired bedside monitor or the
Information Center.
2-8 Alarms

Physiologic Alarms
Alarm/INOPs at the Information Center
The alarms and INOPs that are displayed, recorded and stored at the Information
Center depend on the type of alarm.
Type of Alarm/INOP Effect at Information Center
Physiologic Alarms
Physiologic alarms indicate a life-threatening situation or a less urgent situation
such as heart rate beyond limits. There are no physiologic alarm signals
generated by the transceiver. All physiologic alarms are generated at the
IntelliVue Information Center, and all alarm signals must be acknowledged
at the Information Center.
All ECG telemetry alarms and
INOPs
Note—ECG is generated from
telemetry when paired.
Bedside ECG INOPs and RESP
INOPs
Bedside non-ECG alarms and
non-ECG INOPs
Displayed, recorded (if
configured), and stored
Ignored. Not displayed, recorded,
or stored
Displayed, recorded (if
configured), and stored
Arrhythmia alarm chaining and other aspects of alarm behavior, such as alarm
levels, setting alarm limits, customizing arrhythmia alarm settings on a per
patient basis, switching individual measurement alarms on/off, and reviewing
alarm messages, are described in IntelliVue Information Center Instructions for
Use.
There are two levels of arrhythmia analysis available at the Information Center:
Basic and Enhanced. Enhanced analysis includes Basic alarms.
Alarms
2-9

Physiologic Alarms
In the table, Red (***) alarms are listed alphabetically, followed by the Yellow
(**) alarms, the Yellow (*) arrhythmia alarms.
Physiologic (Patient) Alarms
Alarm Text Priority Condition Source
***ASYSTOLE Red Asystole. No QRS for 4 consecutive
seconds
*** BRADY yyy < xxx Red Extreme Bradycardia. HR less than
extreme Brady HR Limit
*** DESAT Red Very Low SpO
Saturation. SpO2 value
2
below desaturation limit (10% or 10
"points" below low limit).
*** EXTREME BRADY Red Extrem e Bradycardia. HR less than
extreme Brady HR Limit
*** EXTREME TACHY Red Extreme Tachycardia. HR greater than
extreme Tachy HR Limit
*** TACHY yyy > xxx Red Extreme Tachycardia. HR greater than
extreme Tachy HR Limit
ST/AR
Basic &
Enhanced
Arrhythmia
ST/AR
Enhanced
Arrhythmia
SpO
2
ST/AR
Basic &
Enhanced
Arrhythmia
ST/AR
Basic &
Enhanced
Arrhythmia
ST/AR
Enhanced
Arrhythmia
*** VFIB/TACH Red Ventricular Fibrillation. Fibrillatory
waveform for 4 consecutive seconds
2-10 Alarms
ST/AR
Basic &
Enhanced
Arrhythmia

Physiologic Alarms
Physiologic (Patient) Alarms
Alarm Text Priority Condition Source
*** VTACH Red Ventricular Tachycardia. Sustained run of
PVCs accompanied by a high heart rate
** NURSE CALL Yellow Patient or nurse button press on the
transceiver (when configured for Nurse
Call operation)
Also initiated if the installation includes a
paging system and if the Information
Center is configured for paging upon
receipt of Nurse Call signal.
** SpO
HIGH Yellow High SpO2. SpO2 value greater than high
2
SpO
Limit.
2
** SpO2 LOW Yellow Low SpO2. SpO2 value less than low
SpO
Limit.
2
** SpO2 yyy > xxx or Yellow High SpO2. SpO2 value greater than high
SpO
Limit.
2
** SpO2 yyy < xxx or Yellow Low SpO2. SpO2 value less than low
SpO
Limit.
2
* HR HIGH Yellow High Heart Rate. HR greater than high
HR Limit
ST/AR
Basic &
Enhanced
Arrhythmia
Clinicianinitiated at
transceiver
or by
paging
subsystem
SpO
2
SpO
2
SpO
2
SpO
2
ST/AR
Basic &
Enhanced
Arrhythmia
* HR LOW Yellow Low Heart Rate. HR less than low HR
Limit
ST/AR
Basic &
Enhanced
Arrhythmia
Alarms
2-11

Physiologic Alarms
Physiologic (Patient) Alarms
Alarm Text Priority Condition Source
* HR yyy > xxx Yellow High Heart Rate. HR greater than high
HR Limit
ST/AR
Enhanced
Arrhythmia
* HR yyy < xxx Yellow Low Heart Rate. HR less than low HR
Limit
ST/AR
Enhanced
Arrhythmia
* IRREGULAR HR Yellow Irregular Heart Rate. Constantly irregular
HR.
ST/AR
Enhanced
Arrhythmia
* MISSED BEAT Yellow Missed Beat. Beat omitted ST/AR
Enhanced
Arrhythmia
* MULTI ST Lx, Ly Yellow Multi ST Leads exceeding Limit (EASI
mode or when selected). Two ST leads
ST/AR
Arrhythmia
(Lx=Lead X and Ly=Lead Y) exceed
alarm limit elevation or depression for >
60 seconds
* MULTIFORM PVCs Yellow Multiform PVCs ST/AR
Enhanced
Arrhythmia
* NON-SUSTAIN VT Yellow Non-Sustained VT. Non-sustained
Ventricular Tachycardia
* PACER NOT CAPT Yellow Pacer Not Capture. Missed beat with pace
pulse (paced patient).
2-12 Alarms
ST/AR
Enhanced
Arrhythmia
ST/AR
Basic &
Enhanced
Arrhythmia

Physiologic Alarms
Physiologic (Patient) Alarms
Alarm Text Priority Condition Source
* PACER NOT PACE Yellow Pacer Not Pacing. Missed beat without
pace pulse (paced patient).
ST/AR
Basic &
Enhanced
Arrhythmia
* PAIR PVCs Yellow Pair of PVCs. ST/AR
Enhanced
Arrhythmia
* PAUSE Yellow Pause. No QRS for more than x seconds ST/AR
Enhanced
Arrhythmia
* PVC > xx/min Yellow PVCs > xx/min. PVCs greater than Rate
Limit
ST/AR
Basic &
Enhanced
Arrhythmia
* R-ON-T PVCs Yellow R-on-T PVCs ST/AR
Enhanced
Arrhythmia
* RUN PVCs Yellow Run PVCs. Run of PVCs length >= 2 ST/AR
Enhanced
Arrhythmia
* ST lead > xxx Yellow STx > Elevation limit. ST segment is
elevated
* ST lead < xxx Yellow STx < Depression limit. ST segment is
depressed.
* SVT Yellow Supra Ventricular Tachycardia. SVT for >
15 seconds
ST/AR
Arrhythmia
ST/AR
Arrhythmia
ST/AR
Enhanced
Arrhythmia
Alarms
2-13

Technical Alarms (INOPs)
Physiologic (Patient) Alarms
Alarm Text Priority Condition Source
* VENT BIGEMINY Yellow Ventricular Bigeminy. Predominant
Bigeminy rhythm present.
* VENT RHYTHM Yellow Ventricular Rhythm. Ventricular rhythm
present.
* VENT TRIGEMINY Yellow Ventricular Trigeminy. Predominant
Trigeminy rhythm present.
Notes: xxx = limit that was exceeded; yyy = current value.
* NBP alarms are processed independently at the Information Center.
The alarm delay from TeleMon to the Information Center is not more than 10 seconds.
ST/AR
Enhanced
Arrhythmia
ST/AR
Enhanced
Arrhythmia
ST/AR
Enhanced
Arrhythmia
Technical Alarms (INOPs)
Technical Alarms, or INOPs, are sourced at the transceiver, the ST/AR
algorithm running at the Information Center, or TeleMon Companion Monitor,
and identify inoperative conditions (that is conditions where the system cannot
measure or detect alarm conditions reliably). There are three levels of Technical
Alarms:
2-14 Alarms
• Severe - Monitoring and alarms disabled. Audible tone at the Information
Center. Must be acknowledged by a clinician.
• Hard - Monitoring and alarms are disabled. Audible tone at the
Information Center.
• Soft - Monitoring and alarms remain active. No audible tones are
generated.

Technical Alarms (INOPs)
Technical Alarms (Inoperative Conditions) (in alphabetic order)
Alarm Text Priority Condition What to do
BATTERY LOW Soft Power is low. Replace batteries promptly to
avoid transceiver shutdown
and cessation of monitoring.
CANNOT ANALYZE
ECG
Hard Arrhythmia algorithm
cannot reliably analyze the
ECG data on any
monitored leads.
CANNOT ANALYZE ST Soft ST algorithm cannot
reliably generate any valid
ST values on any
monitored lead.
CHARGE MON BAT Hard TeleMon battery charge is
less than or equal to 25%.
CUFF NOT DEFLATED Severe Cuff pressure is greater
than a specified safety limit
for a period of time.
Assess the lead selections,
initiate relearn, and validate
analyzed rhythm.
Review the ECG signal
quality and correct if
necessary. Reposition the Is
and J points.
Connect TeleMon to AC
power and charge battery, or
insert battery with charge
greater than 25%.
Remove cuff from patient
and disconnect from tubing.
Gently expel any air. If water
in cuff, replace cuff.
Reconnect, and reapply cuff
to patient.
Note—If the alarms are
suspended, this INOP will
unsuspends them at the
Information Center and
TeleMon. To clear the INOP
message, initiate a new NBP
measurement.
Alarms
2-15

Technical Alarms (INOPs)
Technical Alarms (Inoperative Conditions) (in alphabetic order)
Alarm Text Priority Condition What to do
ECG EQUIPMENT MALF Hard Failure of the ECG
equipment or failure to
calibrate ECG.
Replace the transceiver or
calibrate ECG with Service
Tool.
ECG LEADS OFF Hard Multiple leads off Reattach ECG leads to
patient.
<electrode> LEAD OFF Hard Primary lead is off.
Note—If primary lead is
Reattach ECG lead to
patient.
MCL, lead will be
identified as V in INOP
text.
<electrode> LEAD OFF Soft Non-primary lead is off. Reattach ECG lead to
patient.
INVALID LEADSET Hard Bad lead selection switches
in the transceiver
Replace transceiver case.
Use supported leadset.
Contact Service.
NO SIGNAL Hard Patient is out of range,
radio board has failed, ICN
connection failure, or no
batteries in transmitter.
Make sure that the
transceiver is in own care-
group range and has good
batteries. Replace the
transceiver if Power On Self
Test fails, and notify Service
Provider.
REPLACE BATTERY Hard,
Latched
Message
remains
until
acknowle
dged by
clinician.
2-16 Alarms
Dead battery. No
monitoring is occurring.
Replace batteries.

Technical Alarms (INOPs)
Technical Alarms (Inoperative Conditions) (in alphabetic order)
Alarm Text Priority Condition What to do
SOME ECG ALRMS OFF Soft Some yellow arrhythmia
alarms have been turned off
for this patient.
T EQUIP MALF Hard Malfunction in the SpO2
SpO
2
equipment
SpO
T ERRATIC Hard Erratic SpO2
2
measurements, often due to
a faulty sensor or invalid
SpO
measurements, or
2
incorrect transducer
position
SpO
T EXTD UPDATE
2
Numeric is replaced by a
-?-.
Soft The update period of
displayed values is
extended due to an NBP
measurement on the same
limb or an excessively
noisy signal.
SpO
T INTERFERENCE Hard Level of ambient light or
2
level of electrical
interference are so high that
the SpO
measure SpO
sensor cannot
2
and pulse
2
rate.
For information only.
SpO2 board needs to be
replaced. Call Service.
Repeat measurement,
reposition sensor on patient,
or finally, replace sensor.
If NBP is not active, check
the sensor placement.
Reposition the sensor on
patient, or replace sensor.
Reduce ambient light to
sensor or electrical noise
sources.
SpO
T LOW PERFUSION Soft Accuracy may be reduced
2
due to low perfusion. Data
displayed with ?. Warm the
site.
SpO
T NO SENSOR Hard No sensor attached to SpO2
2
device
Increase perfusion. Change
sensor site. Avoid site distal
to BP cuff or intra-arterial
line.
Attach SpO2 sensor.
Alarms
2-17

Technical Alarms (INOPs)
Technical Alarms (Inoperative Conditions) (in alphabetic order)
Alarm Text Priority Condition What to do
SpO2T NOISY SIGNAL Hard Excessive patient
movements or electrical
Reduce movement or
electrical noise sources.
interference are causing
irregular pulse patterns
SpO
T NON-PULSATILE Hard Pulse is too weak or not
2
detectable
Check connection to patient.
Change sensor site. Avoid
site distal to BP cuff or intra-
arterial line.
SpO
T SENSOR MALF Hard Malfunction of the SpO2
2
Replace sensor.
sensor/adapter-cable
TELEMETRY STANDBY Soft Information Center standby
mode timer is active, or
Cancelled when patient is
removed from Standby.
patient was not returned to
telemetry coverage area.
There is no data from bed.
TRANSMITTER MALF Hard Transceiver malfunction Replace and notify Service
Provider.
TRANSMITTER OFF Hard RF shut off after 10
minutes of leads off
WEAK SIGNAL Soft Patient is at outer rang e of
the radio coverage area.
Telemetry pack is receiving
a weak signal with high
data loss from the AP.
Reattach ECG leads to
patient.
Return patient to the radio
coverage area. If patient is in
close proximity to AP,
replace telemetry pack. If
condition exists for multiple
devices in a specific area, the
AP in that area is suspect.
Contact Service.
2-18 Alarms

3
ECG Monitoring
This chapter covers the specifics of ECG measurement. It includes the
following sections:
• ECG Safety Information. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-2
• Measuring ECG . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-3
• Positioning ECG Electrodes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-8
• Connecting the ECG Cable. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-19
• Verifying Electrode Connections . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-22
• Monitoring during Leads Off . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3-23
• Optimizing ECG Measurement Performance. . . . . . . . . . . . . . . . . . . 3-25
Introduction
ECG Monitoring 3-1

ECG Safety Information
ECG Safety Information
WarningWarning
For ALL Patients:
Always confirm Information Center observations with clinical observation
of the patient before administering interventions.
Every lead must be secured to an electrode on the patient.
Conductive parts of electrodes must not contact earth or other conductive
parts.
Philips recommends that you change the lead label only to reflect the
physical placement of electrodes. This will ensure a match between the
monitored lead and the label, and prevent any possible confusion.
When switching from EASI to standard monitoring, there is a loss of data
for 30 seconds.
For PACED Patients:
The output power of the transceiver and other sources of radio frequency
energy, when used in the proximity of a pacemaker, can be sufficient to
interfere with pacemaker performance. Due to the shielding effects of the
body, internal pacemakers are somewhat less vulnerable than external
pacemakers. However, caution should be exercised when monitoring any
paced patient.
3-2 ECG Monitoring
In order to minimize the possibility of interference, position electrodes,
electrode wires, and the transceiver as far away from the pacemaker as
possible.
Consult the pacemaker manufacturer for information on the RF
susceptibility of their products and the use of their products with the
Philips IntelliVue Telemetry System. See the IntelliVue Information Center
Instructions for Use for additional information on monitoring paced
patients.

Measuring ECG
The electrocardiogram (ECG) measures the electrical activity of the heart and
displays it on the Information Center as a waveform and a numeric.
There is no cardiotach within the transceiver; cardiotach analysis resides in the
arrhythmia algorithm at the Information Center. Arrhythmia analysis is always
turned on for telemetry patients. Arrhythmia analysis is either basic or enhanced,
depending on the product configuration.
Measuring ECG
EASI ECG EASI
conventional 12-lead ECGs. As the 12-lead ECG derived with EASI is not
exactly identical to the 12-lead conventional ECG obtained from an
electrocardiograph, it should not be used for diagnostic interpretations.
ECG
Leadsets
The IntelliVue Transceiver supports 3-, 5-, and 6-wire leadsets. It detects the
inserted leadset type and automatically determines the ECG measurement and
transmitted leads. The Leadset Insertion Guide on the device will assist you in
ensuring the correct measurement during transceiver usage (see “Connecting the
ECG Cable” on page 3-19). The 5-wire leadset can be used for either standard or
EASI electrode configurations. The leadsets are compatible with the 5- and 3wire leadsets used with the IntelliVue family of monitors and with M2601B
transmitters.
The electrode placements for the illustrations in this chapter use the AAMI
labels and colors and are summarized in the following table.
Leadset Electrode Color (AAMI) Electrode Location
3-wire Black
®
derived 12-lead ECGs and their measurements are approximations to
LA
White
Red
RA
LL
ECG Monitoring
3-3

Measuring ECG
Leadset Electrode Color (AAMI) Electrode Location
ECG Leads
Monitored
5-wire
(Standard mode)
5-wire (EASI
mode)
6-wire Black
Depending on the leadset connected to the transceiver, a different set of
viewable leads are available at the Information Center. The transceiver can
Black
White
Red
Green
Brown
Black
White
Red
Green
Brown
White
Red
Brown
Green
Brown/White
LA
RA
LL
RL
V
S
I
A
RL
E
LA
RA
LL
Va
RL
Vb
3-4 ECG Monitoring

Measuring ECG
source up to four raw ECG waves. The transceiver automatically recognizes the
leadset connected.
these leads can be selected at the
If you are using ...
Information Center
3-wire I, II, III
If lead selection is enabled, sourced waves are
received as:
• Channel 1 - I
• Channel 2 = II
• Channel 3 = III
If lead selection is disabled, the sourced wave is
II.
Default is II.
5-wire (Standard mode) I, II, III, aVR, aVL, aVF, MCL and V
Sourced waves are received as:
• Channel 1 - I
• Channel 2 = II
• Channel 3 = III
Defaults are II, V, III.
5-wire (EASI mode) I, II, III, aVR, aVL, aVF, V1, V2, V3, V4, V5,
V6
In EASI mode, the sourced waves are received
as:
• Channel 1 = Vector 1 (A-I)
• Channel 2 = Vector 2 (A-S)
• Channel 3 = Vector 3 (E-S)
Note—Arrhythmia monitoring is performed only
on the primary and secondary leads selected at
the Information Center, although you can view
and perform ST analysis on all 12 EASI derived
leads.
ECG Monitoring
3-5

Measuring ECG
If you are using ...
these leads can be selected at the
Information Center
6-wire
(not supported in TeleMon)
I, II, III, aVR, aVL, aVF, V1, V2, V3, V4, V5,
V6, V7, V8, V9, V
, V4R, V5R.
3R
Sourced waves are received as:
• Channel 1 = II
• Channel 2 = III
• Channel 3 = Va
• Channel 4 = Vb
The two chest leads, Va and Vb, can be placed
on the patient in any of the V lead positions (V1
through V9, V3R, V4R, V5R). Lead assignment
is available at the Information Center. When
unassigned, the chest leads use the defaults.
Defaults are II, III, Va=V2, Vb=V5.
Note—When display of the pleth wave is
enabled at the Information Center, the second
chest lead (Vb) is not available for monitoring
due to bandwidth limit. That is, the lead label
assigned to Vb cannot be selected for Va even
though Vb does not appear to be used.
Reconstructed
Leads
3-6 ECG Monitoring
Reconstruction of leads from the sourced wave is defined by the calculations in
the following table. EASI reconstructed leads are a linear combination of all
three raw EASI leads. Default labels/leads are shown in bold text.
ECG Lead
Clinical Calculations
in terms of electrodes
3-Lead
5-Lead
Standard
6-Lead
II ILA-RA

ECG Lead
Measuring ECG
5-Lead
3-Lead
Standard
6-Lead
II II II
III
- MCL - Va-LA, C=Va (see Note)
- aVR aVR RA-(LA+LL)/2
- aVL aVL LA-(RA+LL)/2
- aVF aVF LL-(LA+RA)/2
-
III III
V
Va C-(RA+LA+LL)/3, where
Vb Vb-(RA+LA+LL)/3, where Vb
Clinical Calculations
in terms of electrodes
LL-RA
LL-LA
C-(RA+LA+LL)/3, where C=V
V2 position
Va=
=
V5
ECG Monitoring
3-7

Positioning ECG Electrodes
Positioning ECG Electrodes
WarningWarning
Do not mix and match electrodes of different types. In particular, do not use
electrodes of dissimilar metals. This helps ensure optimal signal quality.
WarningWarning
When you are connecting the electrodes or the patient cable, make sure that
the connectors never come into contact with other conductive parts, or with
earth. In particular, make sure that all of the ECG electrodes are attached
to the patient, to prevent them from contacting conductive parts or earth.
WarningWarning
Non-manufacturer supplied accessories and supplies can corrupt the
performance of the equipment. Use only AAMI EC-12 compliant electrodes
with this device. Use of electrodes that are non-compliant may provide
erroneous results.
3-8 ECG Monitoring
Caution
To protect the transceiver from damage during defibrillation, to ensure accurate
ECG information, and to provide protection against signal noise and other
interference, use only ECG electrodes and cables specified by Philips.
To make it possible to compare measured ECG signals, the electrodes (or
leadsets) are placed in standardized positions, forming so-called "leads". To
obtain ECG signals optimized for use in diagnosis and patient management in
different care environments, different leadsets in varying lead placements can be
used. You can use either standard lead placements or EASI lead placements with
the transceiver.

Positioning ECG Electrodes
When placing electrodes on the patient, choose a flat, non-muscular site where
the signal will not be impacted by either movement or bones. Correct lead
placement is always important for accurate diagnosis. Especially in the
precordial leads, which are close the heart, QRS morphology can be greatly
altered if an electrode if moved away from its correct location.
In addition to correct positioning of the electrodes, optimal skin preparation
prior to electrode placement will help ensure a clear signal for diagnosis.
Step
Action
1 Prepare the patient’s skin. Good electrode-to-skin contact is
important for a good ECG signal, as the skin is a poor conductor of
electricity.
• Select sites with intact skin, without impairment of any kind.
• Clip or shave hair from the site as necessary.
• Wash site with soap and water, leaving no soap residue.
Note--Philips does not recommend using ether or pure
alcohol, because they dry the skin and increase the resistance.
• Dry thoroughly.
• Use ECG skin preparation paper (abrasive) to remove dead
skin cells and to improve the conductivity of the electrode
site.
ECG Monitoring
3-9

Positioning ECG Electrodes
Electrode
Placement
Step
Diagrams for 5-lead standard and EASI electrode placement are located on the
back of the transceiver. Additional lead placement information is available in the
Online Help in the IntelliVue Information Center.
Action
2 Check electrodes for moist gel, and attach to the clips. If you are not
using pre-gelled electrodes, apply electrode gel to the electrodes
before placement.
Note—Gel must be moist to provide a good signal.
3 Place the electrodes on the patient according to the lead placement
you have chosen (see Electrode Placement following). Place the
edge down, then "roll down" the rest of the pad. Press firmly around
the adhesive edge toward the center.
Note—When placing electrodes, choose a flat, non-muscular site
where the signal will not be interfered with by either movement or
bones. Correct lead placement is always important for accurate
measurement, especially in the precordial leads, which are close to
the heart. QRS morphology can be greatly altered if an electrode is
moved away from its correct location.
Philips recommends that electrodes be changed every 24 hours.
3-10 ECG Monitoring

Locating the
Fourth
Intercostal
Space
Positioning ECG Electrodes
Angle of Lewis
For accurate chest electrode placement and measurement, it is important to
locate the fourth intercostal space. This can be done using the Angle of Lewis.
1. Locate the second intercostal space by first palpating the Angle of Lewis
(the bony protuberance where the body of the sternum joins the
manubrium). This rise in the sternum is where the second rib is attached,
and the space just below this is the second intercostal space.
2. Palpate and count down the chest until you locate the fourth intercostal
space.
ECG Monitoring
3-11

Positioning ECG Electrodes
3-Lead
Placement
RA LA
LL
3-12 ECG Monitoring
Lead Placement
RA directly below the clavicle and near the right
shoulder
LA directly below the clavicle and near the left
shoulder
LL on the left lower abdomen

5-Lead
Placement
(Standard
Mode)
RA LA
Lead Placement
V1
V2
V3
Positioning ECG Electrodes
V4V5V6
LLRL
RA directly below the clavicle and near the right
shoulder
LA directly below the clavicle and near the left
shoulder
LL on the left lower abdomen
RL on the right lower abdomen
V on the chest, the position depends on your
required lead selection. The default position is V2.
V1 on the fourth intercostal space at the right sternal
border
ECG Monitoring
3-13

Positioning ECG Electrodes
Lead Placement
V2 on the fourth intercostal space at the left sternal
border
V3 midway between the V2 and V4 electrode
positions
V4 on the fifth intercostal space at the left
midclavicular line
V5 on the left anterior axillary line, horizontal with
the V4 electrode position
V6 on the left anterior axillary line, horizontal with
the V4 electrode position
3-14 ECG Monitoring

5-Lead
Placement
(EASI Mode)
Positioning ECG Electrodes
WarningWarning
EASI derived 12-lead ECGs and their measurements are approximations to
conventional 12-lead ECGs. As the 12-lead ECG derived with EASI is not
exactly identical to the 12-lead conventional ECG obtained from an
electrocardiograph, it should not be used for diagnostic interpretations.
EASI lead placement is supported for adult patients only
S
AI
E
Lead
E V on the lower sternum at the level of the
A LL on the left midaxillary line at the same
Corresponds to
Standard Lead
Placement
fifth intercostal space
level as the E electrode
ECG Monitoring
3-15

Positioning ECG Electrodes
6-Lead
Placement
Lead
Corresponds to
Standard Lead
Placement
S LA on the upper sternum
I RA on the right midaxillary line at the same
level as the E electrode
N Reference can be anywhere, usually below the sixth
rib on the right hip
Make sure that the D and E electrodes line up vertically on the sternum,
Note—
and that the I, E and A electrodes align horizontally.
A 6-lead placement uses the same four limb leads as 5-lead standard placement,
and two precordial leads - referred to at the Information Center as Va and Vb.
The default position of Va - the brown lead - is at the V2 position.
The default position for Vb - the brown/white lead - is at the V5 position.
Your unit may use other precordial leads for Va and Vb. In that situation, you
need to assign those new positions in the Patient Window at the Information
Center. The Va and Vb default positions can be changed in the configuration at
the Information Center on a per patient basis.
3-16 ECG Monitoring

Lead Placement
V1
V2
V3
Positioning ECG Electrodes
LARA
V4V5V6
LLRL
RA directly below the clavicle and near the right
shoulder
LA directly below the clavicle and near the left
shoulder
RL on the right lower abdomen
LL on the left lower abdomen
Va on the chest, the position depends on your
required lead selection. The Philips default
position is V2. For other positions, relabel the lead
at the Information Center.
ECG Monitoring
3-17

Positioning ECG Electrodes
Lead Placement
Vb on the chest, the position depends on your
required lead selection. The Philips default
position is V5. For other positions, relabel the lead
at the Information Center.
V1 on the fourth intercostal space at the right sternal
border
V2 on the fourth intercostal space at the left sternal
border
V3 midway between the V2 and V4 electrode
positions
V4 on the fifth intercostal space at the left
midclavicular line
V5 on the left anterior axillary line, horizontal with
the V4 electrode position
3-18 ECG Monitoring
V6 on the left anterior axillary line, horizontal with
the V4 electrode position

Connecting the ECG Cable
Note—Your transceiver may have alignment guides to assist you in leadset
insertion.
Connecting the ECG Cable
Step
Action
1 Match the arrow on the ECG cable with the arrow on the Lead
Insertion Guide according to the lead type you have chosen, and
insert the ECG cable into the transceiver.
Important—Make sure that the cable is pushed completely into
the transceiver. When correctly inserted, the orange line at the
base of the cable is not visible.
3-wire
Match the arrow on the cable
with the left arrow (labeled
EASI, 3) on the Leadset Insertion Guide.
IntelliVue TRx
M4841A
+
EASI, 3
5.6
Note—Leadset is keyed for
only one insertion position.
ECG Monitoring
3-19

Connecting the ECG Cable
Step
Action
5-wire Standard
+
IntelliVue TRx
M4841A
EASI, 3
5-wire EASI
Match the arrow on the cable
with the right arrow (labeled 5,
6) on the Leadset Insertion
Guide.
5.6
Match the arrow on the cable
with the left arrow (labeled
EASI, 3) on the Leadset Insertion Guide.
3-20 ECG Monitoring
+
IntelliVue TRx
M4841A
EASI, 3
5.6
Note—If you are using a 5-wire leadset in both Standard and EASI
modes, insert the protective plug into the open lead port with the
wide lip facing the leads. Then, press down firmly so that the plug is
securely under the leadset and the orange line is not visible.

Connecting the ECG Cable
Step
Action
6-wire
Match the arrow on the cable
with the right arrow (labeled
5,6) on the Leadset Insertion
Guide.
+
IntelliVue TRx
M4841A
EASI, 3
5.6
2 Check that the correct Electrode Indicators are lit for the leadset you
are using, and that the EASI indicator light is illuminated
appropriately. See “Verifying Electrode Connections” on page 3-22.
• When using 5-lead standard, the EASI indicator does not
illuminate.
• When using EASI, the EASI indicator illuminates
momentarily.
Cable
Disconnection
3 For 3-lead, select the lead label(s) at the Patient Window.
When disconnecting the leadset from the transceiver, grasp the leadset firmly
and pull free. Do not pull on the lead wires.
ECG Monitoring
3-21

Verifying Electrode Connections
Verifying Electrode Connections
The electrode indicators enable you to verify that the leads are available for the
desired monitoring. Each electrode is color-coded. Pressing and holding the
Check button enables you to view the leadset status. During routine use of the
transceiver for monitoring, all lead indicators are off.
To verify electrode connections, use the following procedure:
Step
Action
1 Press and hold the Check button for 2 seconds
2 Expected Response:
• If 3-wire cable is attached: Red, White and Black indicators
illuminate, then all turn off.
• If 5-wire cable in Standard mode is attached: Red, White,
Black, Green & Brown indicators illuminate, then all turn off.
• If 5-wire cable in EASI mode is attached: Red, White, Black,
Green & Brown indicators illuminate, then all turn off. The
EASI indicator also illuminates briefly.
• If 6-wire cable is attached: Red, White, Black, Green, Solid
Brown and Brown/White indicators illuminate, then all turn
off.
• If no leadset is attached: all indicators are off.
3 Unexpected Response:
Any other response indicates a problem with the transceiver. Check
the leadset connection and/or use a new leadset. If the problem is
not corrected, contact your Service Provider.
3-22 ECG Monitoring

During routine monitoring, the electrode indicators also notify you if one or
more leads are not functioning. When a LEADS OFF condition occurs, the
transceiver automatically illuminates the indicator corresponding to the missing
lead.
Monitoring during Leads Off
ECG Fallback and Extended monitoring states are supported for the transceiver
when the primary and/or secondary leads are in a Leads Off INOP condition.
Both these states are entered into after 10 seconds of Leads Off in an attempt to
maintain monitoring and arrhythmia analysis.
Monitoring during Leads Off
ECG
Fallback
Multilead
Analysis
Single Lead
Analysis
Fallback for
EASI
ECG Fallback occurs when the primary lead is in Leads Off for 10 seconds and
a secondary lead is available. ECG Fallback must be configured on by your
Service Provider.
If there is a LEADS OFF technical alarm in the primary lead for > 10 seconds,
the active secondary lead becomes the primary lead. The arrhythmia algorithm
switches the leads on the display, but relearn does not occur. When the Leads
Off condition is corrected, the leads are switched back to their original state.
For single lead analysis, if there are two leads available, the secondary lead is
made the primary lead until the Leads Off condition is corrected. The arrhythmia
algorithm performs a relearn using the available lead.
If one of the derived EASI leads is in a technical alarm condition, a flat line is
displayed. After 10 seconds, the directly acquired EASI AI, AS, or ES lead,
depending on which is available, is displayed with the label "ECG". Arrhythmia
relearn is performed with transition to or from EASI Fallback monitoring using
the available lead(s).
ECG Monitoring
3-23

Monitoring during Leads Off
Extended
Monitoring
Extended monitoring occurs when both the primary and secondary leads are in
Leads Off for 10 seconds and another lead is available. It becomes the primary
lead and the arrhythmia algorithm performs a relearn.
Extended Monitoring applies if:
• Telemetry is configured for Extended Monitoring ON.
• The leas set provides more than two leads (e.g., when using a 5-wire
leadset ).The leadset must provide more than two leads, and Extended
monitoring must be configured on by your Service Provider.
Relearning Whenever there is a Leads Off condition, the arrhythmia algorithm performs a
Relearn, using the available leads.
WarningWarning
Since Relearn happens automatically, if learning takes place during
ventricular rhythm, the ectopics can be incorrectly learned as the normal
QRS complex. This can result in missed detection of subsequent events of
V-Tach and V-Fib. For this reason, you should:
1. Respond promptly to any technical alarm.
2. Ensure that the arrhythmia algorithm is labeling beats correctly.
Using EASI Leads to T roubleshoot
If there is artifact in the ECG waves or a CANNOT ANALYZE ECG technical
alarm condition is in effect, you can use the three EASI leads to troubleshoot:
1. Click
2. The three directly acquired EASI leads will be displayed so that you can
3-24 ECG Monitoring
12-Lead ECG on the Patient Window, then on 3 EASI Leads.
determine which electrodes are causing the problem and need to be
replaced.

Optimizing ECG Measurement Performance
Optimizing ECG Measurement Performance
While telemetry monitoring offers many advantages, it can be a challenge. The
reliability and quality of the signal transmission through the air and hospital
walls is governed by a number of variables which can be difficult to control. A
telemetry system cannot be as dependable as a hardwired bedside monitor that
transmits its signal through a wire.
The effect of interference on the Philips IntelliVue Telemetry System ranges
from a momentary loss of ECG to complete inoperability, depending on the
situation. The strength, frequency, and proximity of the source of interference to
the transceiver devices or the access point system are factors that determine the
degree of severity. In cases where the source of interference is known - for
example, cellular phones, magnetic equipment such as MRI, other radio or
motorized equipment - removing or moving away from the source of
interference will increase the equipment's dependability.
WarningWarning
Philips IntelliVue Telemetry System should not be used for primary
monitoring in applications where the momentary loss of the ECG is
unacceptable.
The
Telemetry
Signal
In this section, we'll investigate some of the problems affecting ECG signal
clarity and when possible, show you how you can greatly enhance performance.
Important—Philips IntelliVue Telemetry System also emits radio frequencies
that can affect the operation of other devices. Contact the manufacturer of other
equipment for possible susceptibility to these frequencies.
The transceiver worn by the patient acquires the patient's physiological data,
amplifies and digitizes it, detects pace pulses and broadcasts this information via
radio waves to the Philips IntelliVue Telemetry System. Since the signal passes
through the air, it is susceptible to interference from many sources.
ECG Monitoring
3-25

Optimizing ECG Measurement Performance
Trouble- shooting Signal Disturbances
Dropouts Because Philips IntelliVue Telemetry System is a wireless system, under certain
conditions RF (Radio Frequency) “dropouts” can occur. Dropouts result from a
weak signal or RF interference. There will be signal drops to the bottom of
channel for a minimum of 200 ms to indicate to the clinical user that it is a nonphysiological event. If dropouts are frequent enough to affect the heart rate
count, the CANNOT ANALYZE ECG or CANNOT ANALYZE ST technical
alarm occurs. The following recording strip is an example of dropouts.
If frequent dropouts are occurring, the following section describes some steps
you can take to improve performance.
Signal
Strength
3-26 ECG Monitoring
The Philips IntelliVue Telemetry System is custom designed for your unit, so
reliable signal reception is only possible where there are receiving access points.
When the signal is too low, the following technical alarms occur:
To correct, first check the location of the patient. If the patient is not in the
coverage area, do one of the following.
• CANNOT ANALYZE ECG
• CANNOT ANALYZE ST
• WEAK SIGNAL
• NO SIGNAL
• Return the patient to the specified coverage area.
• Put telemetry in Standby Mode. See “Standby Mode” on page 2-5.
• If the patient is in the coverage area and is stationary, try moving the
location of the transceiver from its original location by about 15 cm (6
inches).

Optimizing ECG Measurement Performance
Radio
Frequency
Interference
Muscle and
Movement
Artifact
Problem Cause Remedy
60-Cycle (AC) Interference Poor electrode placement.
Radio frequency (RF) interference is caused by anything that intrudes into the
transmitted electrical signal, such as paging transmitters. You are probably
familiar with electrical interference in our homes and cars when it causes signal
loss or static with cell phones. These same types of interference can occur with
the transmitted telemetry signal. Even though the Philips IntelliVue Telemetry
System is designed to resist these effects, interference can occasionally be seen
in the form of “dropouts”. To improve performance, the source of the
interference must be identified and eliminated.
Muscle and movement artifact differ from radio frequency interference since
you can prevent much of the occurrence. Noise on the ECG signal can be caused
by many sources, such as interference from other electrical equipment, muscle
artifact and respiratory variation. It is up to the clinician to use certain
techniques to minimize these types of noise.
Use the following table to help you troubleshoot the most common sources of
ECG noise.
Troubleshooting Common Causes of ECG Noise
Possible non-grounded
instrument near patient
Apply fresh electrodes after
recommended skin preparation.
Disconnect electrical appliances near
patient (one at a time) by pulling wall
plugs, to determine faulty grounding.
Have engineering check grounding.
Muscle Artifact Tense, uncomfortable
patient.
Poor electrode placement.
Tremors.
Diaphoresis
Make sure patient is comfortable.
Check that electrodes are applied on
flat non-muscular areas of the torso;
apply fresh electrodes after
recommended skin preparation if
necessary (see “Positioning ECG
Electrodes” on page 3-8).
ECG Monitoring
3-27

Optimizing ECG Measurement Performance
Troubleshooting Common Causes of ECG Noise
Problem Cause Remedy
Irregular Baseline Poor electrical contact.
Respiratory interference.
Faulty electrodes.
Dry electrodes.
Baseline Wander Movement of patient.
Improperly applied
electrodes.
Respiratory interference.
Poor Electrode Contact Loose electrodes.
Defective cables.
Leadset not firmly
connected.
Apply fresh electrodes after
recommended skin preparation if
necessary (see “Positioning ECG
Electrodes” on page 3-8).
Move electrodes away from areas with
greatest movement during respiration.
Make sure patient is comfortable.
Apply fresh electrodes after
recommended skin preparation if
necessary (see “Positioning ECG
Electrodes” on page 3-8). Check that
patient cable is not pulling electrodes.
Move electrodes away from areas with
greatest movement during respiration.
Apply fresh electrodes after
recommended skin preparation if
necessary (see “Positioning ECG
Electrodes” on page 3-8).
Replace cables.
3-28 ECG Monitoring

4
ST/AR Arrhythmia & ST Segment
Monitoring
This chapter describes the ST/AR algorithms used for telemetry at the Philips
Information Center. It includes the following sections:
• ST/AR Arrhythmia Algorithm . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-2
• ST/AR provides Heart Rate and PVC Rate numerics and alarm detection
for the conditions listed in the following table. There are two detection
levels: Basic and Enhanced. Enhanced includes the Basic alarms. . . . 4-4
• The Measurement . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-6
• Adjusting ST Measurement Points. . . . . . . . . . . . . . . . . . . . . . . . . . . . 4-9
• Establishing ST Reference Beats (Baseline) . . . . . . . . . . . . . . . . . . . 4-11
• ST Alarm Settings. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-11
• ST Alarm Settings. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .4-11
Introduction
ST/AR Arrhythmia & ST Segment Monitoring 4-1

ST/AR Arrhythmia Algorithm
ST/AR Arrhythmia Algorithm
Safety
Information
WarningWarning
FOR ALL PATIENTS
During complete heart block or pacemaker failure (to pace or capture), tall
P-waves (greater than 1/5 of the average R-wave height) can be erroneously
counted by the arrhythmia algorithm, resulting in missed detection of
cardiac arrest.
Learning/Relearning
- Learning: If you initiate learning during ventricular rhythm, the ectopics
can be incorrectly learned as the normal QRS complex. This can result in
missed detection of subsequent events of V-Tach and V-Fib.
- Relearning: When using EASI ECG monitoring, Relearn happens
automatically when there is a LEADS OFF technical alarm. If learning
takes place during ventricular rhythm, the ectopics can be incorrectly
learned as the normal QRS complex. This can result in missed detection of
subsequent events of V-Tach and V-Fib. Be sure to check the beat labels
and initiate a relearn to correct.
1. Respond to the technical alarm [for example, reconnect the electrode(s)].
2. Ensure that the arrhythmia algorithm is labeling beats correctly.
4-2 ST/AR Arrhythmia & ST Segment Monitoring

ST/AR Arrhythmia Algorithm
WarningWarning
FOR PACED PATIENTS
It is possible that pacemaker pulses will not be detected when the ECG
analog output of a defibrillator or telemetry unit is plugged into a bedside
monitor. This can result in the arrhythmia algorithm’s failure to detect
pacemaker non-capture or asystole.
Some pace pulses can be difficult to reject. When this happens, the pulses
are counted as a QRS complex, and could result in an incorrect HR and
failure to detect cardiac arrest or some arrhythmias. Keep pacemaker
patients under close observation.
-- During complete heart block or pacemaker failure (to pace or capture),
tall P-waves (greater than 1/5 of the average R-wave height) can be
erroneously counted by the arrhythmia algorithm, resulting in missed
detection of cardiac arrest.
-- When arrhythmia monitoring paced patients who exhibit only intrinsic
rhythm, the monitor can erroneously count pace pulses as QRS complexes
when the algorithm first encounters them, resulting in missed detection of
cardiac arrest.
ST/AR
Arrhythmia
Analysis
For patients who exhibit intrinsic rhythm only, the risk of missing cardiac
arrest can be reduced by monitoring these patients with the low heart rate
limit at or slightly above the basic/demand pacemaker rate. A low heart
rate alarm alarms you when the patient begins pacing. Proper detection
and classification of the paced rhythm can then be determined.
-- When an external pacemaker is being used on a patient, arrhythmia
monitoring is severely compromised due to the high energy level in the
pacer pulse. This can result in the arrhythmia algorithm’s failure to detect
pacemaker non-capture or asystole.
For information on arrhythmia detection, refer to the following documentation:
• ST/AR Algorithm - Arrhythmia Monitoring Application Note (4522 981
ST/AR Arrhythmia & ST Segment Monitoring
4-3

ST/AR Arrhythmia Algorithm
• IntelliVue Information Center Instructions for Use and Online Help
The intended use of the ST/AR basic arrhythmia analysis algorithm is to monitor
the patient’s ECG for heart rate and ventricular arrhythmias and to produce
events/alarms simultaneously for one or more ECG leads. The arrhythmia
algorithm is effective when monitoring both paced and non-paced patients in a
clinical environment.
IntelliVue Telemetry does not have a dedicated cardiotach. Instead, the
arrhythmia cardiotach at the Information Center is used. Therefore, the ST/AR
Arrhythmia algorithm is always on for all IntelliVue Telemetry patients, and
cannot be turned off.
ST/AR provides Heart Rate and PVC Rate numerics and alarm detection for the
conditions listed in the following table. There are two detection levels: Basic and
Enhanced. Enhanced includes the Basic alarms.
Asystole Tachy yyy > xxx Vent Bigeminy
93051)
Basic & Enhanced Arrhythmia Detection
Basic Arrhythmia
Detection
Enhanced Arrhythmia Detection
Vent Fib/Tach Brady yyy < xxx Vent Trigeminy
V tach Non-Sustain VT Multiform PVCs
Extreme Brady Vent Rhythm HR yyy > xxx
Extreme Tachy Run PVCs HR yyy < xxx
High HR Pair PVCs Irregular HR
Low HR Pause
PVCs > 30/min Missed Beat
Pacer Not Capturing SVT
Pacer Not Pacing R-on-T PVCs
4-4 ST/AR Arrhythmia & ST Segment Monitoring

ST/AR Arrhythmia Algorithm
Beat classification determined by the ST/AR algorithm is shown on the primary
delayed wave in the Arrhythmia Analysis window at the Information Center. To
access this window, select Arrhythmia Analysis from the Patient Window.
The annotation requires clinical validation of the analyzed heart rhythm. If the
analysis is inaccurate, perform a relearn of the rhythm.
Annotation Beat Classification Color
A Artifact Blue
I Inoperative Red
L Learning Red
M Missed Beat Red
N Normal Blue
P Paced Blue
S Supraventricular Premature Blue
V Ventricular Premature Red
? Questionable Red
‘ Pacer Mark Blue
When monitoring is initiated, when the Wave 1 lead is changed, or if Relearn is
selected, a question mark (?) is displayed next to HR and the annotation “L”
appears on the annotated wave until the HR is calculated and the rhythm is
learned.
ST/AR Arrhythmia & ST Segment Monitoring
4-5

ST/AR ST Segment Algorithm
ST/AR ST Segment Algorithm
WarningWarning
This device provides ST level change information; the clinical significance
of the ST level change information should be determined by a physician.
The ST/AR ST algorithm at the Information Center (not available for M3153A)
monitors ST segment elevation or depression for each available telemetry ECG
lead and produces events/alarms simultaneously. ST values update with every
measurement period and enunciate, depending upon the severity of the change,
events and alarms as they are detected.
The ST/AR ST algorithm is approved for use only with non-paced and atriallypaced adult telemetry-monitored patients. With EASI monitoring, ST analysis is
performed on up to 12 leads, and an additional value of ST index is calculated
and displayed (see “EASI ST Analysis” on page 4-8). Assessment of EASIderived 12-lead ST measurement is recommended for adult patients that meet
the following parameters:
• Ages: 33-82 years
• Heights: 147 to 185 cm (58 to 73 in)
• Weights: 53 to 118 kg (117 to 261 lbs)
• Height to Weight Ratios: 1.41 to 2.99 cm/kg (0.25 to 0.54 in/lb)
All ST analysis and ST alarms for telemetry patients are performed by the
Information Center.
For additional information on ST monitoring, refer to the following
documentation:
• ST/AR Algorithm - ST Segment Monitoring Application Note (4522 981
92851)
• Information Center Online Instructions for Use and Online Help
The
Measurement
4-6 ST/AR Arrhythmia & ST Segment Monitoring
The ST measurement for each beat complex is the vertical difference between
two measurement points. The isoelectric point provides the baseline for the

ST/AR ST Segment Algorithm
measurement and the ST point provides the other measurement point. It is
positioned with reference to the J-point.
R-WAVE PEAK
AT 0 MSEC
P
ISO ELECTRIC
POINT
DEFAULT =
-80 MSEC
J POINT
Q
S
MEASUREMENT
DEFAULT =
J+60 MSEC
ST
POINT
T
DIFFERENCE =
ST VALUE
ST/AR Arrhythmia & ST Segment Monitoring
4-7

ST/AR ST Segment Algorithm
Algorithm
Processing
Displayed
ST Data
ST analysis analyzes ECG signals to classify the heart beats. Only beats
classified as normal or Supraventricular (atrially paced) are used to calculate ST
elevations and depressions.
The ST/AR ST algorithm processing includes special ST filtering, beat selection
and statistical analysis, calculation of ST segment elevations and depressions,
and lead reconstruction and wave generation.
When ST analysis is being performed on two leads, the averaged derived and
reconstructed ST waves and associated ST segment values are given for up to
six leads, depending on the type of patient cable:
• 3-wire: one lead
• 5-wire: up to two leads if monitoring a chest and a limb lead
• 5-wire: up to six leads if monitoring two limb leads with the Philips
Transmitter (without EASI monitoring)
• 5- wire: up to 12 leads if monitoring using EASI
Note—No ST analysis is done on a patient if an electrode falls off.
ST data displays as values in the Patient Sector and Patient Window. A positive
value indicates ST segment elevation; a negative value indicates ST segment
depression. You can view ST data in ST Review, Trend Review, and Event
Review windows.
EASI ST
Analysis
4-8 ST/AR Arrhythmia & ST Segment Monitoring
The Information Center generated ST values presented in the patient sector and
Patient Window for EASI derived leads is STindx (ST Index). STindx is a
summation of three ST segment measurements, using the leads that can indicate
ST segment changes in the different locations of the heart:
• anterior lead V2
• lateral lead V5
• inferior lead aVF
Caution
Be sure not to duplicate the lead labels. This can result in incorrect ST values
being displayed for those leads.

ST
Operation
ST/AR ST Segment Algorithm
Turning ST
Monitoring On/
Off
Adjusting ST
Measurement
Points
The ST Setup Window allows you to turn ST monitoring on or off for all
available ECG leads.
To turn ST monitoring on at the Information Center, perform the following
steps:
Step
You would turn ST monitoring off if:
• You are unable to get any lead that is not noisy.
• Arrhythmias such as atrial fib/flutter cause irregular baseline.
• The patient is continuously ventricularly paced.
• The patient has left bundle branch block.
The ST Setup Window enables you to adjust the ST measurement points to
ensure accurate data.
There are three measurement cursors:
Action
1 From the Patient Window, click the All Controls button.
2 From the All Controls Window, click the
3 From the ST Setup Window, click
ST Setup button.
ST On.
• The ISO measurement cursor positions the isoelectric point in relation to
the R-wave peak.
• The J-point cursor positions the J-point in relation to the R-wave peak.
The purpose of the J-point is to correctly position the ST measurement
point.
• The ST measurement cursor positions the ST point a fixed distance from
the J point.
Note—The ST measurement points may need to be re-adjusted if the patient's
heart rate or ECG morphology changes significantly.
ST/AR Arrhythmia & ST Segment Monitoring
4-9

ST/AR ST Segment Algorithm
Perform the following steps at the Information Center to adjust the ST
measurement points:
Step
Action
1 Access the ST Setup window by clicking on the All Controls button
in the Patient Window, then clicking on the ST Setup button.
2 If you need to adjust the ISO (isoelectric) point, place the cursor
over the ISO button to access the adjustment arrows. Then use the
arrows to position the bar in the middle of the flattest part of the
baseline (between the P and Q waves or in front of the P wave).
R
T
S
ISO
point
P
Q
4-10 ST/AR Arrhythmia & ST Segment Monitoring

ST/AR ST Segment Algorithm
Step
Action
3 Adjust the J point, if necessary, by placing the cursor over the J-
point button to access the adjustment arrows. Then use the arrows to
position the bar at the end of the QRS complex and the beginning of
the ST segment.
R
P
SQJ point
T
4 Adjust the ST point, if necessary, by using the J point as an
“anchor” and placing the bar at the midpoint of the ST segment.
Choices are J+0, J+20, J+40, J+60, or J+80.
R
P
Q
T
S
ST point
Establishing
ST Reference
Beats
(Baseline)
ST Alarm
Settings
After adjusting the measurement points, you can establish baseline reference
beats for all available leads in the ST Review window at the Philips Information
Center. Reference beats enable you to compare waveform changes, for example
from admission, or prior to or after treatment. The reference continues to be
saved beyond the 24 hour review window, but you can update it to any beat
within the last 24 hours. Please refer to the Philips Information Center
Instructions for Use18
or on-line Help for directions.
All Philips Information Center alarm settings (limits and on/off status) have unit
default settings. The Philips Information Center however, lets you set the high
and low ST alarm limits for individual patients based on:
ST/AR Arrhythmia & ST Segment Monitoring
4-11

ST/AR ST Segment Algorithm
• Your assessment of the patient's clinical condition.
• Unit protocols.
• Physician orders or medication specified limits.
You can make the following adjustments to ST alarm limits to accommodate the
clinical condition of individual patients:
• Turn all alarms off/on.
• Adjust the alarm limits:
You adjust the ST alarm limits in the ST Alarms Window. Each ST parameter
has its own alarm limit. The alarm is triggered when the ST value exceeds its
alarm limit for more than 1 minute. The alarm will be a yellow alarm.
When more than one ST parameter is in alarm, only one alarm message displays.
For multilead alarms when using an EASI transmitter, an alarm is generated if
two or more ST leads exceed the alarm limits. The default setting is +/-1.0. The
alarm message indicates the two leads that are in greatest violation of the limits,
for example, “**MULTI ST AVR, V6”. If another lead becomes deviant, the
message changes but it is considered the same alarm (no new alarm sounds and
it is not listed as a new event).
– to specific high and low limits
– to Smart Limits (see the Philips IntelliVue Information Center
Instructions for Use for information on Smart Limits)
– back to unit default settings.
See “Physiologic Alarms” on page 2-9 for a list of all ST alarms.
See IntelliVue Information Center Instructions for Use for specifics on alarm
management and behavior.
Adjusting ST
Alarms
Make adjustments to ST alarms on the ST Alarms window at the Information
Center.
Step
Action
1 From the Patient Window, select the All Controls button.
4-12 ST/AR Arrhythmia & ST Segment Monitoring
 Loading...
Loading...