Philips M1032A VueLink User manual

Philips M1032A VueLink
External Device
Service Booklet
PHI
Part Number M1032-9000Y 4512 610 18581
Published in October 2006 Edition 22
Y.00.00

Notice
Philips makes no warranty of any kind with regard to this material, including, but not limited to, the implied warranties of merchantability and fitness for a particular purpose. Philips shall not be liable for errors contained herein or for incidental or consequential damages in connection with the furnishing, performance or use of this material.
This document contains proprietary information that is protected by copyright. All rights are reserved. No part of this document may be photocopied, reproduced or translated to another language without prior written consent of Philips Medical Systems. The information contained in this document is subject to change without notice. Philips assumes no responsibility for the use or reliability of its software on equipment that is not furnished by Philips.
Important
United States federal law restricts these devices to sale by or on the order of a physician.
2

Intended Use
Description The Philips M1032A VueLink module is a plug-in module for use with a Philips IntelliVue, V24 or V26 Patient Monitor or a Philips CMS Patient Monitoring System.
The Philips M1032A VueLink module is powered via the patient monitor.
Purpose The Philips M1032A VueLink module transmits information from a connected external device to a Philips IntelliVue, V24 or V26 Patient Monitor or a Philips CMS Patient Monitoring System. The Philips M1032A VueLink module is not a therapeutic device.
Environ- The Philips M1032A VueLink module is intended to be used in a clinical ment environment by trained healthcare professionals. It is not intended for home use.
3

Indications for Use
Condition The use of a Philips M1032A VueLink module is generally indicated when the clinician decides there is a need to include the physiological and device parameters and waves and alarms generated by external devices into the Philips IntelliVue, V24 or V26 Patient Monitor or a Philips CMS Patient Monitoring System.
Part of Body
or Type of
Tissue with
Which the
Device Inter-
acts
The Philips M1032A VueLink module does not contact the body or tissue of the patient. Signals are obtained from external devices.
Frequency
of Use
Physiological Purpose
The Philips M1032A VueLink module is indicated for use when prescribed by a clinician.
The Philips M1032A VueLink module is indicated when the purpose is to gain information for treatment, to assess adequacy of treatment, or to rule out causes of symptoms. The Philips M1032A VueLink module is well suited for patient monitoring.
Patient Pop-
ulation
Prescription Versus Over-the- Counter
Please refer to the documentation describing the external device.
The Philips M1032A VueLink module is a prescription device.
4

Warnings, Cautions and Notes
|
In this guide: |
|
|
|
|
Warning |
• A warning alerts you to a potential serious outcome, adverse event or |
|
safety hazard. |
|
Failure to observe a warning may result in death or serious injury to the |
|
user or patient. |
|
|
|
|
Caution |
A caution alerts you where special care is necessary for the safe and effective |
|
use of the product. |
|
Failure to observe a caution may result in minor or moderate personal injury |
|
or damage to the product or other property, and possibly in a remote risk of |
|
more serious injury. |
|
|
Note |
A note gives special instructions to highlight an operating procedure or practice. |
|
Notes may precede or follow the applicable text. |
|
|
5

Responsibility of the Manufacturer
Philips Medical Systems only considers itself responsible for any effects on safety, reliability and performance of the equipment if:
•assembly operations, extensions, re-adjustments, modifications or repairs are carried out by persons authorized by Philips, and
•the electrical installation of the relevant room complies with national standards, and
•the Philips M1032A VueLink module is used in accordance with the instructions for use contained in this document, and in the relevant chapter of the user documentation for the patient monitor into which the module is plugged (Philips IntelliVue, V24 or V26 Patient Monitor or a Philips CMS Patient Monitoring System).
Warning |
• Failure on the part of the responsible individual hospital or institution |
|
employing the this equipment to implement a satisfactory maintenance |
|
schedule may cause undue equipment failure and possible health |
|
hazards. |
|
The maintenance schedule is defined in the Testing and Maintenance |
|
chapter of the IntelliVue Patient Monitor Service Guide, the Philips CMS |
|
Patient Monitoring System Service Guide, or the Philips V24/V26 |
|
Component Monitoring System Service Guide, corresponding to your |
|
monitor. |
|
|
|
|
Note |
Not all the device drivers are available in all countries. |
|
The Instructions for Use for all drivers are translated into the following languages: |
|
Czech, Danish, Dutch, Estonian, Finnish, French, German, Greek, Hungarian, |
|
Italian, Japanese, Norwegian, Polish, Portuguese, Romanian, Russian, Simplified |
|
Chinese (China), Slovak, Spanish, Swedish, Traditional Chinese (Taiwan), Turkish. |
|
The driver software may not be available in all of these languages. Please refer to the |
|
CMS-PM softserver (http://pww.softserver.anr.ms.philips.com/navigation.asp, |
6

subfolder Vuelink, subfolder Additional Information) for a list of the translated drivers that are available.
Please also check your local regulatory and business requirements for permission to sell.
Philips authorized technical personnel are responsible for the setup, configuration and repair of the VueLink module and all related Philips equipment. The configuration, setup and repair of external devices furnished by manufacturers other than Philips Medical Systems must be carried out by local site personnel, or by representatives of the external device manufacturer.
Philips Medical Systems is not responsible for any problems arising from inaccurate or erroneous data displayed on the Philips patient monitor that is received from external devices furnished by manufacturers other than Philips Medical Systems.
Philips Medical Systems makes every effort to ensure that signal names are maintained between the Philips patient monitor and any interfaced external devices. However, signal names within the Philips patient monitor also need to remain consistent. This means that in some cases the wave and numeric labels used on the Philips monitor may be different from those used on the interfaced external device.
Philips Medical Systems will not be held responsible for any errors in the configuration of the VueLink module for connection to free-analog devices.
If manufacturers other than Philips Medical Systems make changes to any VueLink supported device, Philips Medical Systems will try to ensure that interfacing to that device remains possible. However, Philips Medical Systems reserves the right to discontinue interfacing to any device.
7

Introduction
Who Should
Use This
Guide
How to Use This Booklet
This Service Guide contains technical details for the connection of external devices to the VueLink module. It is not a comprehensive, in-depth explanation of the product architecture or technical implementation.
This guide is for biomedical engineers or technicians responsible for connecting external devices to Philips patient monitoring systems using the VueLink module.
This booklet provides information on the worldwide external devices that can be connected to the VueLink module. It should be printed and inserted into Appendix A of the VueLink Module Handbook, completely replacing the existing worldwide driver information.
The information contained in this document is subject to change without notice.
Philips Medizin Systeme Böblingen GmbH
Hewlett-Packard Str. 2
71034 Böblingen
Germany
© 2006 Philips Medizin Systeme Böblingen GmbH All rights are reserved.
Reproduction in whole or in part is prohibited without the prior written consent of the copyright holder.
8

Connecting your Device to VueLink
Connecting your Device to VueLink
There are two different ways to connect your Device to VueLink:
•Connection via a Supported Device driver. This uses one of the following device outputs:
–Analog
–RS-232 Digital
–Analog/Digital combination
•Connection via a Free Analog Device driver. This only uses device analog outputs.
Supported Device Drivers
Supported Device Drivers are preconfigured in the VueLink module. A dedicated ready-prepared VueLink cable is available for connecting the external device to the module. (Please refer to page A-4 for cable part numbers).
Free Analog Device Drivers
Free Analog Device Drivers must be manually configured into the module at installation. A Free Analog VueLink cable is available for connecting the Free Analog external device to the VueLink module. One end of the cable is unterminated and must be prepared for connection to the external device on-site. (Please refer to page B-4 for cable option part numbers).
9
Connecting your Device to VueLink
10

Contents
Connecting your Device to VueLink . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9
Supported Device Information . . . . . . . . . . . . . . . . . . . . . . 15
Driver Part Numbers for Supported Devices . . . . . . . . . . . . . . . . . . . . . . . . 16
VueLink Cable Overview . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17
Auxiliary Devices . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 23
Abbott Oximetrix 3 - SO2 Monitor. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 23
Critikon Dinamap 1846 / 1846 SX - NBP Monitor . . . . . . . . . . . . . . . . . . . . 25
Danmeter AAI/AEP Monitor. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 27
Nellcor® N-200 - SpO2 Monitor. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 29
Nellcor® N-100C - SpO2 Monitor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31
Ventilators . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 33
Nellcor Puritan-Bennett 740 / 760 / 840 Ventilators. . . . . . . . . . . . . . . . . . . 33 Puritan-Bennett 7200a / 7200ae Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . 36 Dräger Babylog 8000 Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 38 Dräger Evita Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 40 Dräger Evita 2 Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 42 Dräger Evita 2 Dura, Dräger Evita 4 & Dräger Evita XL Ventilators . . . . . . 44 Dräger Graphic Screen for Babylog 8000 . . . . . . . . . . . . . . . . . . . . . . . . . . . 46 Dräger Graphic Screen for Savina . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 48 Dräger Savina . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 50 GE Engström Carestation Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 52 Hamilton Veolar, Veolar FT, and Amadeus Ventilators . . . . . . . . . . . . . . . . . 54 BEAR 1000 Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 56 Infrasonics® Infant Star / ISV Ventilator. . . . . . . . . . . . . . . . . . . . . . . . . . . . . 58 Siemens SCM 990 Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 61 Siemens 900 C/D/E Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 64 Maquet Servo 300/300A Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 66 Maquet SERVO-i Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 68 Maquet SERVO-s Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 70
Gas Analyzers . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 73
HP M1025A/B Gas Analyzer . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 73
Datex Capnomac (II and Ultima) Gas Analyzer . . . . . . . . . . . . . . . . . . . . . . . 75
Dräger Vamos Variable Anaesthetic Gas Monitor. . . . . . . . . . . . . . . . . . . . . 78
11
Ohmeda RGM 5250 Respiratory Gas Monitor . . . . . . . . . . . . . . . . . . . . . . . .80 Ohmeda Rascal II Anesthetic Gas Monitor . . . . . . . . . . . . . . . . . . . . . . . . . . .82
Anesthesia Machines . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .85
Dräger Cato Anesthesia Device. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .85
Dräger Cicero Anesthesia Machine . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .87
Dräger Cicero EM Anesthesia Machine . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .89
Dräger Fabius GS/Tiro Anesthesia Machine. . . . . . . . . . . . . . . . . . . . . . . . . . .91
Dräger Julian Anesthesia Workstation. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .93
Dräger Pallas Anesthesia Workstation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .95
Dräger PM 8050 Anesthesia Machine . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .96
Dräger Primus/Apollo Anesthesia Workstation . . . . . . . . . . . . . . . . . . . . . . .98
Dräger Zeus Anesthesia Device. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .100
GE Aestiva and Avance. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .102
GE Aisys . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .105
North American Dräger Narkomed 2B and Narkomed 2C Anesthesia Systems107
North American Dräger Narkomed 4 Anesthesia System . . . . . . . . . . . . . . .112
North American Dräger Narkomed GS Anesthesia System. . . . . . . . . . . . . .114
North American Dräger Narkomed 6000 Anesthesia System . . . . . . . . . . . .116
Ohmeda 7800/7810 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .118
Ohmeda 7900 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .120
Ohmeda Modulus CD Anesthesia Machine . . . . . . . . . . . . . . . . . . . . . . . . . . .122
Taema Alys Anesthesia Machine. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .125
Auxiliary Plus Devices . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .127
Aspect Medical Systems A-2000™ BIS™ Monitor . . . . . . . . . . . . . . . . . . . . .127 Baxter Explorer Cardiopulmonary Monitor . . . . . . . . . . . . . . . . . . . . . . . . . .129 B|Braun SpaceCom . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .131 Braun FM Device Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .134 Diametrics Medical IRMA SL Series 2000 . . . . . . . . . . . . . . . . . . . . . . . . . . . .136 Diametrics Medical Trendcare Monitor. . . . . . . . . . . . . . . . . . . . . . . . . . . . . .138 Edwards CardiacOutput-Oximetry Monitor Family . . . . . . . . . . . . . . . . . . . .140 Fresenius Vial Base A/DPS/MVP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .144 GE DINAMAP ProCare 100 Series . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .146 i-STAT® 1 Analyzer . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .148 Mortara ELI 100/STM 12-Lead Monitor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .150 Organon Teknika TOF-Watch SX . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .152
Free Analog Device Information . . . . . . . . . . . . . . . . . . . . .155
12
Free Analog Device Information . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 155
Configuring the Free Analog Device . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 156
Dräger Evita Ventilator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 158
Engström ELSA/EAS Anesthesia System . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 166
Nellcor N1000 and N2500 Multi-Function Monitor . . . . . . . . . . . . . . . . . . . 174
Radiometer Transcutaneous pO2/pCO2 Monitor . . . . . . . . . . . . . . . . . . . . . 182
13
14

A1
Supported Device Information
This appendix contains information about the supported devices that can be connected to the VueLink module. The following information is provided for each supported device:
•The part number of the corresponding device driver.
•The configuration settings required on the external device.
•The placement of the output connectors on the external device.
A cable matrix has also been compiled, detailing the cable part numbers for each supported device.
Caution |
Only use cables specified by Philips Medical Systems when connecting |
|
supported devices to the VueLink module. Use of unauthorized or incorrect |
|
cables may result in damage to supported devices and may cause incorrect |
|
data to be displayed on the Philips patient monitor. |
|
|
|
The configuration settings required on the external devices are detailed on the |
|
following pages. Procedures on how to configure the external devices are not |
|
provided. Configuration procedures can be found in the documentation supplied |
|
with the external devices. Philips personnel are only responsible for configuring |
|
Philips manufactured external devices. |
|
|
Note |
The signal labels used on the Philips patient monitor may be different from those |
|
given on the external device. The labels used on the Philips patient monitor are |
|
listed in the Philips M1032A VueLink External Device Instructions for Use. |
|
|
Supported Device Information |
15 |
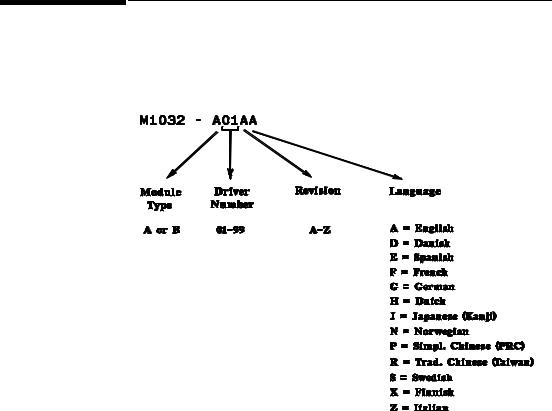
Driver Part Numbers for Supported Devices
Driver Part Numbers for Supported Devices
Each driver is allocated a part number, using the driver key detailed below:
16 |
Supported Device Information |

VueLink Cable Overview
VueLink Cable Overview
3 Separate part numbers are assigned to each VueLink Cable:
M1032A KXX option |
To order a cable with a new module. |
M1182A KXX option |
To order a cable for an existing module or for |
|
upgrades (Sales). |
M1032-XXXXX option |
To order a cable for an existing module from |
|
SMO/SLI. (Support or Immediate Requirement) |
The Supported Device cable options are detailed in the matrix opposite:
VueLink Cable Overview
|
New Module |
Patient Monitor |
SMO/SLI Option |
||||
|
Cable Option |
||||||
|
Option (M1032A #) |
(M1032-) |
|||||
Device |
(M1182A #) |
||||||
|
|
|
|
||||
|
2m |
4m |
2m |
4m |
2m |
4m |
|
|
|
|
|
|
|
|
|
Auxiliary Devices |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Abbott Oximetrix 3 |
K73 |
- |
K73 |
- |
61619 |
61620 |
|
Critikon 1846/1846 SX |
K72 |
- |
K72 |
- |
61613 |
61614 |
|
Danmeter AAI/AEP Monitor |
K56 |
- |
K56 |
- |
61693 |
- |
|
Nellcor N-200 |
K71 |
- |
K71 |
- |
61617 |
61618 |
|
Nellcor N-100C |
K70 |
- |
K70 |
- |
61615 |
61616 |
|
|
|
|
|
|
|
|
|
Ventilators |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Dräger Babylog 8000 |
- |
K2H |
- |
K2H |
- |
61644 |
|
Dräger Evita |
K24 |
K2E |
K24 |
K2E |
61629 |
61630 |
|
Dräger Evita 2 |
- |
K2G |
- |
K2G |
- |
61643 |
|
|
|
|
|
|
|
|
|
Supported Device Information |
17 |
VueLink Cable Overview
VueLink Cable Overview
|
New Module |
Patient Monitor |
SMO/SLI Option |
||||
|
Cable Option |
||||||
|
Option (M1032A #) |
(M1032-) |
|||||
Device |
(M1182A #) |
||||||
|
|
|
|
||||
|
2m |
4m |
2m |
4m |
2m |
4m |
|
Dräger Evita 2 dura |
- |
K2M |
- |
K2M |
- |
61680 |
|
Dräger Evita 4 |
|
|
|
|
|
|
|
Dräger Evita XL Ventilator |
|
|
|
|
|
|
|
Dräger Graphic Screen for |
- |
K29 |
- |
K29 |
- |
61604 |
|
Babylog 8000 |
|
|
|
|
|
|
|
Dräger Graphic Screen for Savina |
- |
K28 |
- |
K28 |
- |
61604 |
|
Dräger Savina |
- |
K27 |
- |
K27 |
- |
61644 |
|
GE Engström Carestation |
- |
K2N |
- |
K2N |
- |
61607 |
|
Hamilton Amadeus |
- |
271 |
- |
271 |
- |
61673 |
|
Hamilton Veolar |
- |
271 |
- |
271 |
- |
61673 |
|
Maquet Servo 300 |
- |
K2F |
- |
K2F |
- |
61642 |
|
|
|
|
|
|
|
|
|
Maquet SERVO-i |
- |
K23 |
- |
K23 |
- |
61696 |
|
Maquet SERVO-s |
- |
K26 |
- |
K26 |
- |
61696 |
|
|
|
|
|
|
|
|
|
Nellcor Puritan Bennett 740 |
- |
K25 |
- |
K25 |
- |
61665 |
|
Nellcor Puritan Bennett 760 |
- |
K25 |
- |
K25 |
- |
61665 |
|
Nellcor Puritan Bennett 840 |
- |
K25 |
- |
K25 |
- |
61665 |
|
Puritan-Bennett 7200a/ae |
K20 |
K2A |
K20 |
K2A |
61621 |
61622 |
|
Siemens 900 C/D/E |
K21 |
K2B |
K21 |
K2B |
61623 |
61624 |
|
Siemens SCM 990 |
K22 |
K2C |
K22 |
K2C |
61625 |
61626 |
|
|
|
|
|
|
|
|
|
BEAR 1000 |
- |
K2K |
- |
K2K |
- |
61657 |
|
|
|
|
|
|
|
|
|
18 |
Supported Device Information |
|
|
|
|
|
VueLink Cable Overview |
||
VueLink Cable Overview |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
New Module |
Patient Monitor |
|
SMO/SLI Option |
|||
|
Cable Option |
|
|||||
|
Option (M1032A #) |
|
(M1032-) |
||||
Device |
(M1182A #) |
|
|||||
|
|
|
|
|
|||
|
2m |
4m |
2m |
4m |
|
2m |
4m |
Infrasonics Infant Star / ISV |
- |
K2J |
- |
K2J |
|
- |
61645 |
|
|
|
|
|
|
|
|
Gas Analyzers |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Datex Capnomac (II & Ultima) |
K41 |
- |
K41 |
- |
|
61633 |
- |
Dräger Vamos |
K4A |
- |
K4A |
- |
|
61695 |
- |
Philips M1025A/B |
K40 |
- |
K40 |
- |
|
61631 |
- |
Ohmeda RGM 5250 (old)1 |
- |
- |
- |
- |
|
61635 |
- |
Ohmeda RGM 5250 (new) |
K43 |
- |
K43 |
- |
|
61636 |
- |
Ohmeda Rascal II |
K45 |
- |
K45 |
- |
|
61664 |
- |
|
|
|
|
|
|
|
|
Anesthesia Machines |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Dräger Cato |
K02 |
- |
K02 |
- |
|
61602 |
- |
Dräger Cicero |
K01 |
- |
K01 |
- |
|
61601 |
- |
Dräger Cicero EM mono |
K10 |
- |
K10 |
- |
|
61675 |
- |
Dräger Cicero EM color |
- |
K12 |
|
K12 |
|
- |
61685 |
Dräger Fabius GS/Tiro |
K49 |
- |
K49 |
- |
|
61700 |
- |
Dräger Julian |
K11 |
- |
K11 |
- |
|
61681 |
- |
Dräger Pallas |
K4C |
- |
K4C |
- |
|
61694 |
- |
Dräger PM 8050 |
270 |
- |
270 |
- |
|
61676 |
- |
Dräger Primus/Apollo |
K48 |
- |
K48 |
- |
|
61694 |
- |
Dräger Zeus |
- |
K4B |
- |
K4B |
|
- |
61666 |
|
|
|
|
|
|
|
|
Supported Device Information |
19 |
VueLink Cable Overview
VueLink Cable Overview
|
New Module |
Patient Monitor |
SMO/SLI Option |
||||
|
Cable Option |
||||||
|
Option (M1032A #) |
(M1032-) |
|||||
Device |
(M1182A #) |
||||||
|
|
|
|
||||
|
2m |
4m |
2m |
4m |
2m |
4m |
|
GE Aestiva |
K15 |
- |
K15 |
- |
61684 |
- |
|
GE Aisys |
K16 |
- |
K16 |
- |
61684 |
- |
|
GE Avance |
K15 |
- |
K15 |
- |
61684 |
- |
|
NAD2 Narkomed 2B/2C |
K05 |
- |
K05 |
- |
61605 |
- |
|
NAD2 Narkomed 4 |
K06 |
- |
K06 |
- |
61605 |
- |
|
NAD2 Narkomed GS |
K47 |
- |
K47 |
- |
61605 |
- |
|
NAD2 Narkomed 6000 |
K46 |
- |
K46 |
- |
61605 |
- |
|
Ohmeda Modulus CD |
K03 |
- |
K03 |
- |
61603 |
- |
|
Ohmeda 7800 |
K07 |
- |
K07 |
- |
61658 |
- |
|
Ohmeda 7810 |
K08 |
- |
K08 |
- |
61659 |
- |
|
Ohmeda 7900 |
K09 |
- |
K09 |
- |
61678 |
- |
|
Taema Alys |
274 |
- |
274 |
- |
61674 |
- |
|
|
|
|
|
|
|
|
|
Auxiliary Plus Devices |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Aspect Medical Systems |
- |
K52 |
- |
K52 |
- |
61687 |
|
A-2000 BIS Monitor |
|
|
|
|
|
|
|
Baxter Explorer |
K74 |
- |
K74 |
- |
61651 |
- |
|
Baxter Vigilance |
- |
- |
- |
- |
61652 |
- |
|
B|Braun SpaceCom |
- |
K59 |
- |
K59 |
- |
61649 |
|
|
|
|
|
|
|
|
|
Braun FM |
- |
275 |
- |
275 |
- |
61691 |
|
|
|
|
|
|
|
|
|
20 |
Supported Device Information |
|
|
|
|
|
VueLink Cable Overview |
||
VueLink Cable Overview |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
New Module |
Patient Monitor |
|
SMO/SLI Option |
|||
|
Cable Option |
|
|||||
|
Option (M1032A #) |
|
(M1032-) |
||||
Device |
(M1182A #) |
|
|||||
|
|
|
|
|
|||
|
2m |
4m |
2m |
4m |
|
2m |
4m |
|
|
|
|
|
|
|
|
Braun FM software revision < 3.0 |
- |
- |
- |
- |
|
- |
61648 |
Diametrics Medical IRMA SL |
- |
K54 |
|
K54 |
|
- |
61688 |
- |
|
||||||
Series 2000 |
|
|
|
|
|
|
|
Diametrics Medical Trendcare |
- |
K55 |
- |
K55 |
|
- |
61689 |
monitor |
|
|
|
|
|
|
|
Edwards CardiacOutput- |
K75 |
- |
K75 |
- |
|
61692 |
- |
Oximetry Monitor Family |
|
|
|
|
|
|
|
Fresenius Vial Base A/DPS/ |
- |
277 |
- |
277 |
|
- |
61682 |
MVP |
|
|
|
|
|
|
|
GE DINAMAP ProCare 100 |
- |
K78 |
- |
K78 |
|
- |
61606 |
Series Patient Monitor |
|
|
|
|
|
|
|
i-STAT® 1 Analyzer |
- |
K58 |
- |
K58 |
|
|
61667 |
Mortara ELI 100/STM |
- |
K76 |
- |
K76 |
|
- |
61653 |
Organon Teknika |
K53 |
- |
K53 |
- |
|
61686 |
- |
TOF-Watch SX |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Free Analog Devices |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Free Analog |
K60 |
K6A |
K60 |
K6A |
|
61611 |
61612 |
|
|
|
|
|
|
|
|
Open Interface Cable (25-pin |
- |
K6B |
- |
K6B |
|
- |
61654 |
digital/analog) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Open Interface Cable (9-pin |
- |
K6C |
- |
K6C |
|
- |
61699 |
digital) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1.Old cable only available for support in case an existing cable fails.
2.NAD = North American Dräger
Supported Device Information |
21 |
VueLink Cable Overview
22 |
Supported Device Information |

A2
Auxiliary Devices
Abbott Oximetrix 3 - SO2 Monitor
Device Driver Name: |
Abbott Oximetrix 3 |
|
Device Driver P/N: |
M1032-A07rl |
|
|
where: |
|
|
r = revision |
|
Supported Devices: |
l = language |
|
Oximetrix (R)3 SO2 Monitor (version 104 and |
||
|
105) |
|
Connection: |
RS-232 Digital (Fixed configuration) |
|
|
Baud Rate: |
1200 |
|
Word Length: |
8 bits |
|
Stop Bits: |
1 |
|
Start Bits: |
1 |
Switch Settings: |
Parity: |
None |
Factory default |
|
|
Auxiliary Devices |
23 |

Abbott Oximetrix 3 - SO2 Monitor
|
Abbott Oximetrix 3 - SO2 Monitor Rear Panel |
|
|
Note |
Please refer to the Philips M1032A VueLink External Device Instructions for Use |
|
for exact details of waves, numerics, INOPs and alarms available from the external |
|
device and via Philips patient monitoring network. |
|
|
24 |
Auxiliary Devices |

Critikon Dinamap 1846 / 1846 SX - NBP Monitor
Critikon Dinamap 1846 / 1846 SX - NBP Monitor
Device Driver Name: |
Critikon 1846 / 1846 SX |
|
Device Driver P/N: |
M1032-A06rl |
|
|
where: |
|
|
r = revision |
|
Supported Devices: |
l = language |
|
Critikon 1846 (with software revision 1846RCM |
||
|
and 1846PRBG) |
|
|
Critikon 1846 SX (with software revision SXRCH |
|
|
and SXPRDH) |
|
Connection: |
RS-232 Digital (Fixed configuration) |
|
|
Baud Rate: |
600 |
|
Word Length: |
8 bits |
|
Stop Bits: |
1 |
Switch Settings: |
Parity: |
None. |
Factory default |
|
|
Auxiliary Devices |
25 |
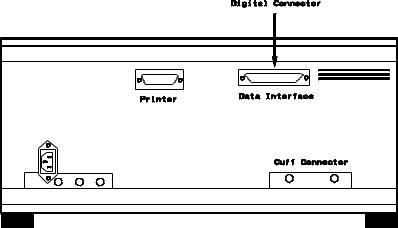
Critikon Dinamap 1846 / 1846 SX - NBP Monitor
|
Critikon Dinamap 1846 / 1846 SX - NBP Monitor Rear Panel |
|
|
Note |
Please refer to the Philips M1032A VueLink External Device Instructions for Use |
|
for exact details of waves, numerics, INOPs and alarms available from the external |
|
device and via Philips patient monitoring network. |
|
|
26 |
Auxiliary Devices |

Danmeter AAI/AEP Monitor
Danmeter AAI/AEP Monitor
Device Driver Name: |
AAI/AEP Monitor |
|
Device Driver P/N: |
M1032-A08rl |
|
|
where: |
|
|
r = revision. |
|
Supported Devices: |
l = language. |
|
Danmeter AAI/AEP Monitor (SBC(software) |
||
|
Version 1.5) |
|
Connection: |
RS-232 Digital (at the rear of the device) |
|
|
Baud Rate: |
9600 (Low1) |
|
Word Length: |
8 bits |
|
Stop Bits: |
1 |
|
Start Bits: |
1 |
|
Parity: |
None |
|
Protocol: |
Device Link (ON1) |
Danmeter AAI/AEP Monitor Rear Panel
1. Must be set and verified according to the document “AAI/AEP Monitor Directions for Use”.
Auxiliary Devices |
27 |

Danmeter AAI/AEP Monitor
Note |
Please refer to the Philips M1032A VueLink External Device Instructions for Use |
|
for exact details of waves, numerics, INOPs and alarms available from the external |
|
device and via Philips patient monitoring network. |
|
|
28 |
Auxiliary Devices |

Nellcor® N-200 - SpO2 Monitor
Nellcor® N-200 - SpO2 Monitor
Device Driver Name: |
Nellcor® N-200 |
|
|
|
Device Driver P/N: |
M1032-A05rl |
|
|
|
|
where: |
|
|
|
|
r = revision |
|
|
|
Supported Devices: |
l = language |
|
|
|
Nellcor® N-200 |
|
|
||
Connection: |
Analog / RS-232 Digital combination |
|||
|
Baud Rate: |
9600 |
|
|
|
Word Length: |
8 bits |
|
|
|
Stop Bits: |
2 |
|
|
|
Parity: |
None |
|
|
Switch Settings: |
Analog: |
1 alarm, 1 wave. |
||
See table below: |
|
|
||
|
|
|
|
|
Name |
|
Number |
Position |
|
|
|
|
|
|
SCALE and OUTPUT VOLT Switches |
|
1 |
|
UP |
|
|
|
|
|
|
|
2 |
|
DOWN |
|
|
|
|
|
DIP Switches |
|
1 |
|
Either |
|
|
|
|
|
|
|
2 |
|
DOWN |
|
|
|
|
|
|
|
3 |
|
DOWN |
|
|
|
|
|
|
|
4 |
|
UP |
|
|
|
|
|
|
|
5 |
|
DOWN |
|
|
|
|
|
|
|
6 |
|
Either |
|
|
|
|
|
|
|
7 |
|
UP |
|
|
|
|
|
|
|
8 |
|
DOWN |
|
|
|
|
|
Auxiliary Devices |
29 |
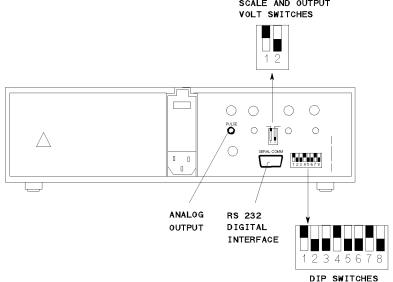
Nellcor® N-200 - SpO2 Monitor
|
Nellcor® N-200 - SpO2 Monitor Rear Panel |
Note |
Please refer to the Philips M1032A VueLink External Device Instructions for Use |
|
for exact details of waves, numerics, INOPs and alarms available from the external |
|
device and via Philips patient monitoring network. |
|
|
30 |
Auxiliary Devices |
 Loading...
Loading...